Abstract
Introduction
In recent years, volatile organic compounds (VOCs) produced by the petrochemical, organic chemistry, painting, and other industries have caused environmental damage by partially destroying the ozone layer, thus posing great risks to human health (Mo et al., 2009). In conventional chemical facilities, adsorption has been widely used in the treatment of low concentrations of VOCs (Shen et al., 2017). Activated carbons (ACs) with their large specific surface area, dense pore structure, and excellent adsorption performance have become a popular choice of adsorbent for removal of VOCs. The superior adsorption performance of ACs is mainly a result of their unique internal structure and surface chemical properties. ACs can be modified to improve their structural properties and thus promote their adsorption performance. The methods of modifying the structural properties of ACs include mainly physical methods (Álvarez-Torrellas et al., 2017), chemical methods (Cao et al., 2014), and the combination of the two methods (Bhatnagar et al., 2013; Rivera-Utrilla et al., 2011). Modification enables the specific surface area of ACs to be greatly enlarged by forming an elaborate pore structure. Surface chemical modification is able to enhance the adsorption performance of ACs by loading functional groups corresponding to the targeted adsorbates, thus promoting chemical reactions. Chemical modification methods include oxidative modification (Zhou et al., 2009), reduction modification (Nakagawa et al., 2007), load metal modification (Mahmoud et al., 2015), and so on. Whether for physical or chemical modification, the traditional methods are based on conventional techniques for establishing contact between reacting phases. The time required for these modification methods is often several tens of hours or more because of the use of conventional immersion contact. To overcome the problem of long modification time, as well as the poor effect of modification, suitable strategies to accelerate modification and significantly improve the efficiency of absorption are urgently needed. One promising form of equipment to strengthen the effect of modification is a high-gravity rotating packed bed (RPB) with ACs as the filler layer.
The RPB is a kind of equipment used to study the physical and chemical changes occurring under high gravity, which is induced by the high-speed rotation of the packing layer. The RPB enhances the effect of mass transfer between substances by increasing the area of contact of a two-phase system. This recently developed mass transfer equipment has been widely used in distillation (Sudhoff et al., 2015), absorption (Tan and Chen, 2006; Zhang et al., 2016), desorption (Tan and Lee, 2008), and extraction (Modak et al., 2016). The technology has been applied by several groups to improve adsorption, based on the better mass transfer effect between the two phases enabled by RPB. Chiang et al. (2012) used an RPB to adsorb hydrophobic VOCs, showing that it is possible to adsorb such VOCs with silicone oil and that an RPB enhances the adsorption effect. The rate of adsorption can reach more than 98% with a contact time of just seconds. Therefore, the use of high-gravity technology in modifying ACs is feasible in principle.
In outline, the RPB uses an adsorbent as a filler to strengthen the mass transfer effect between the adsorbent and a gas or liquid phase. In this way, the adsorbent undergoes compound (i.e. both physical and chemical) thermal modification in the RPB to effectively improve its physical structure and increase the content of surface functional groups. This in turn increases the adsorption performance of the adsorbent (ACs in the present case).
As a highly efficient and inexpensive solid adsorbent, ACs are widely used in the adsorption of gas in industry. Toluene is a toxic and environmentally harmful gas produced by industrial activity. The adsorption of toluene gas by ACs is an economically viable method for its removal. In this study, ACs are modified by an RPB and by traditional modification equipment. The effect of modification by the two kinds of equipment is compared, and the adsorption performance of the modified ACs is investigated.
Experimental
Pretreat of ACs
The method for preparing the initial ACs was as follows: the ACs (with a weight of 100 g, and particle size of 100–200 mesh) were boiled for 30 min in a resistance furnace and washed for 10 min in an ultrasonic washer. After that, the ACs were dried for 12 h at a temperature of 383 K in a vacuum oven, which yielded the pretreated ACs.
Preparation of modified ACs
Thermal compound modification of ACs
The pretreated ACs were placed in a vacuum tube, 99% nitrogen (at a flow rate of 15 L/h) was passed from the side of the pipe, and the ACs were heat-treated for 60 min in the resulting tube furnace with the temperature controlled at 573–873 K. The resulting sample was denoted AC-N. The sample AC-N was placed in 200 mL of a 1 mol·L−1 solution of nitric acid (HNO3) as a modifying solution, treated in a constant-temperature magnetic stirrer for 24 h at a temperature of 343 K, neutralized with deionized water after filtering, and dried in a vacuum oven for 24 h at a temperature of 378 K. The resulting sample was denoted AC-HNO.
Thermal compound modification under high gravity
The thermal compound modification of the ACs in a high-gravity environment is shown in Figure 1. An RPB was packed with 100 g of ACs as the filler layer, and operated at a speed of 900 r/min for 300 min with 99% nitrogen (at a temperature of 573–1173 K) passed from the inlet port. The resulting ACs were denoted AC-RPB-N. Both the original ACs and AC-RPB-N were then modified in an RPB with 1 mol·L−1 HNO3 at a temperature of 343 K, flow rate of 40 L · min−1, and rotational speed of 36.3 Hz. Following this treatment, both samples were dried in a vacuum oven at 378 K for 24 h after rinsing with deionized water, and denoted AC-RPB-HNO and AC-RPB-N-HNO, respectively.
The experimental of modification flowing chart. 1, nitrogen; 2, valve; 3, flow meter; 4, gas heater; 5, Liquid distributor; 6, rotated packed bed; 7, pump; 8, reservoir.
Characterization of ACs
The physical parameters of the ACs, including specific surface area and pore size distribution (microcellular, mesoporous, and macroporous), were determined using the low-temperature nitrogen adsorption method with an automatic physical-chemical adsorption analyzer. The surface chemical groups of the ACs were analyzed using Fourier transform infrared (FTIR) spectroscopy with a wavelength scanning range from 4000 to 400 cm−1. The content of each chemical group was measured using Boehm titration. The morphology of the ACs was observed by scanning electron microscopy (SEM).
Experimental study of adsorption performance
The ability of the ACs for adsorbing toluene was studied in an adsorption column. The dosage of ACs was 10 g and the adsorbate, toluene gas, was produced by a gas generator. The data on the adsorption performance of the ACs under different conditions were obtained through determining the concentration of toluene before and after adsorption. A flow chart of the experimental adsorption apparatus is shown in Figure 2. The toluene gas from the gas generator was mixed with nitrogen, which was then adsorbed in the thermostatic adsorption column. The toluene content before and after adsorption was measured by an online FID detector and the rate of toluene flow was controlled by a flow meter. Adsorption equilibrium was considered to have been reached when the outgassing and inlet concentration were consistent for a period of 30 min. The adsorption gas was emptied through the exhaust gas outlet.
The experimental of adsorption flowing chart. 1, nitrogen; 2, dryer; 3, valve; 4, gas generator of VOCs; 5, flow meter; 6, adsorption column; 7, thermostatic chamber; 8, exhaust treatment; 9, online detector of VOCs. VOCs: volatile organic compounds.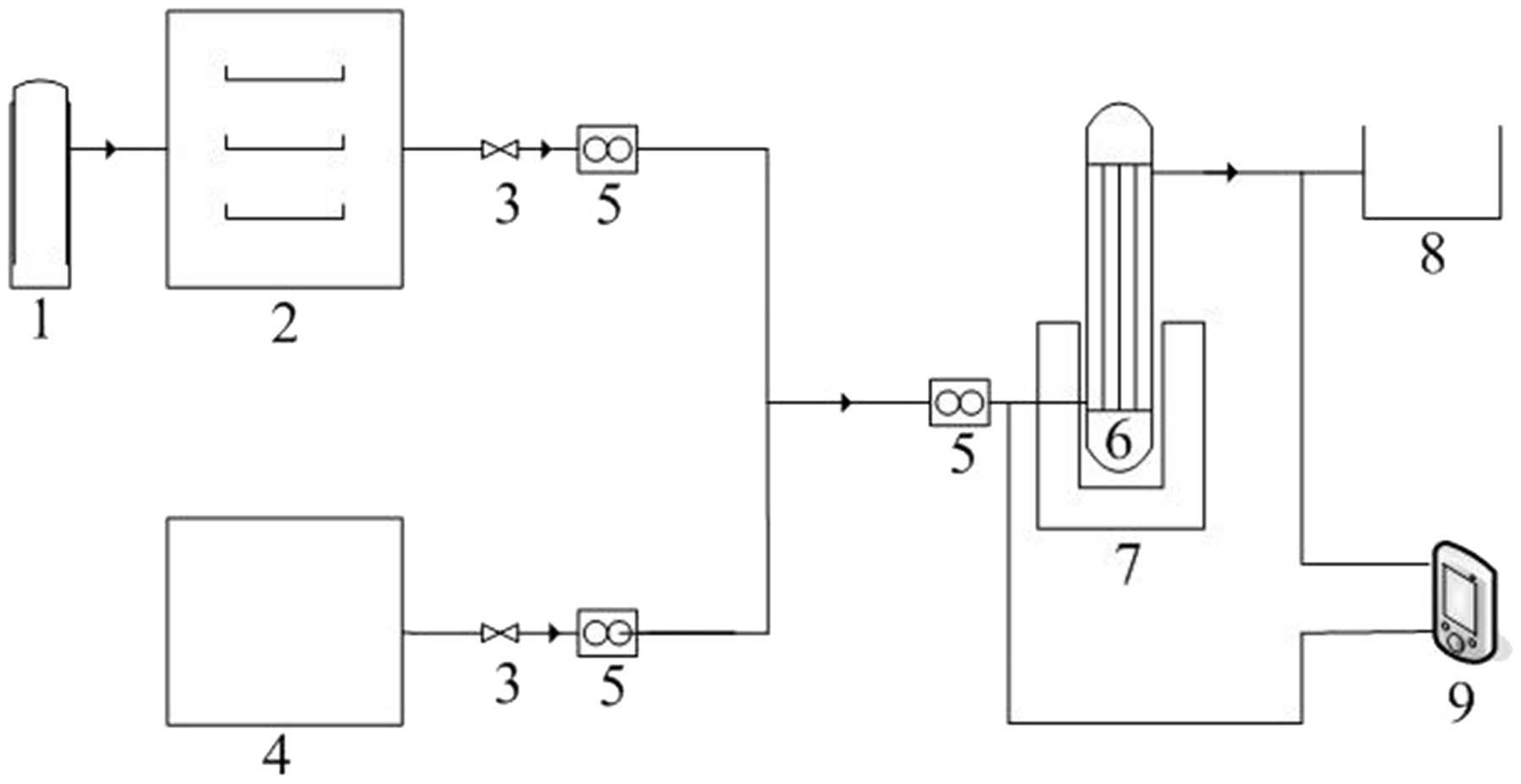
Results and discussion
Analysis of ACs’ surface functional groups
The FTIR spectra are shown in Figure 3. The modified ACs clearly show additional absorption peaks compared with the original ACs. The spectrum of the original ACs is similar to that of the nitrogen-modified ACs. Neither shows a peak at 1050 cm−1, which would indicate –C–O–C– groups located on or slightly below the surface of the ACs. The HNO3-modified ACs have a characteristic peak at 1600 cm−1, indicating the presence of C = O bonds, which may represent acidic groups such as carboxyl or lactonic groups (Radkevich et al., 2008). This confirms that acidic oxygen-containing groups were formed in the HNO3 modification process. All four kinds of ACs show an absorption peak at 3400 cm−1, which arises from the presence of hydroxyl groups. Comparing the two HNO3-modified ACs, the absorption peak areas are obviously larger in the RPB case, indicating the greater content of the corresponding groups.
The FTIR spectrum of different ACs. FTIR: Fourier transform infrared; ACs: activated carbons.
The content of group on ACs.

ACs: activated carbons; RPB: rotating packed bed.
Characterization of the physical structure of ACs
The pore size parameter of ACs.

ACs: activated carbon; RPB: rotating packed bed.

Aperture and micropore distribution of differ ACs. ACs: activated carbons.
The SEM photographs of the ACs are shown in Figure 5. The modified ACs have a greater number of narrow channels compared with the originals. These channels are formed during the high-temperature nitrogen modification of the ACs, in which the decomposition of impurities leads to opening of a part of the pores on the surface, and the surface condensation on the ACs are dispersed into fine channels. The channels of AC-RPB-N are richer than those of AC-N, because the RPB increases the probability of contact between N2 and the ACs. As a result of the accelerated ACs–N2 contact rate in a rotating environment, greater perforation occurred. The number of surface channels of the ACs was increased by modification, which in turn increased their pore volume and specific surface area.
SEM photographs of AC, AC-N, AC-RPB-N. SEM: scanning electron microscopy; AC: activated carbon; RPB: rotating packed bed.
In the high-gravity physical modification process, nitrogen gas distribution is no longer a single flow from top to bottom, but is transported to the ACs by convection in all directions. Upon contact with the ACs, the nitrogen carves out new micropores and dredges the existing adsorption channels. Therefore, it increases their surface area and pore capacity. A simplified schematic is shown in Figure 6.
The schematic of nitrogen and nitric acid modification in normal and high-gravity environment.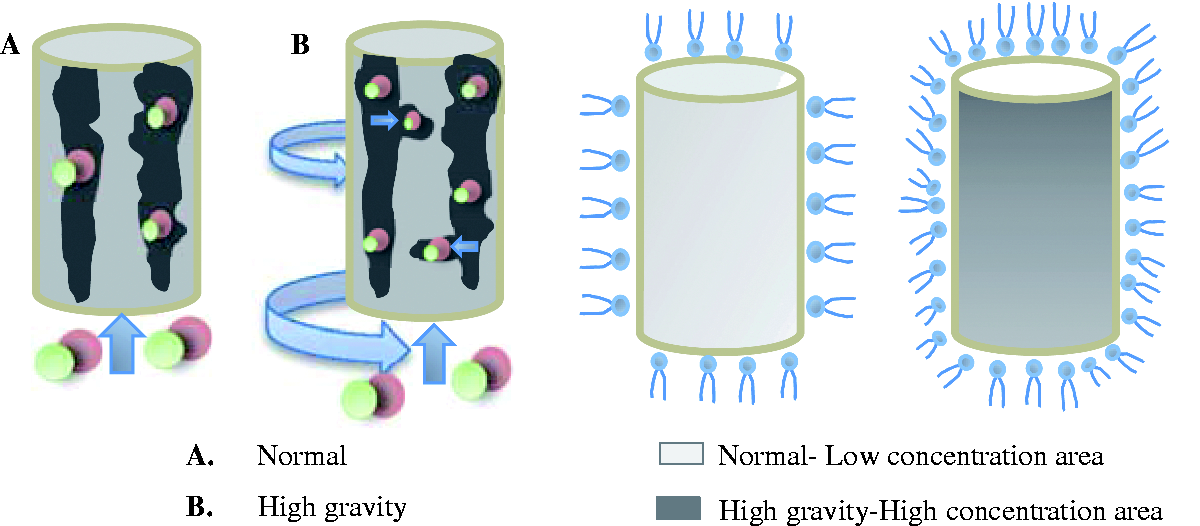
Comparison of adsorption performance
The breakthrough curves and adsorption isotherm curves of toluene on the different ACs are shown in Figures 7 and 8. The rate of adsorption on the different ACs from high to low follows the order: AC-RPB-N-HNO > AC-RPB-HNO > AC-HNO > AC-RPB-N > AC-N > AC. The larger the adsorption rate, the longer the penetration time, which in turn is related to the complex pore size distribution of the ACs and the amount of surface functional groups. The micropore volumes of the ACs are enlarged in high-temperature nitrogen, which increases the internal pore volume of the channels of the ACs. In the high-gravity nitrogen environment, the internal structure of the original ACs, which is unfavorable for absorption of toluene, is redistributed and split into a new spatial configuration, which further increases the breakthrough time. The breakthrough time of the ACs treated by HNO3 modification in a high-gravity environment is greater than that of the ACs with HNO3 soaking. This is because HNO3 is dispersed into fine droplets in the RPB, which increases its contact probability with the surface and interior of the ACs, which in turn enhances the loading of oxygen-containing functional groups on the ACs and increases their adsorption efficacy. In high gravity, the ACs treated by compound modification have a long breakthrough time and large adsorption rate, because physical modification increases the specific surface area and enlarges the pore volume distribution while chemical modification increases the number of surface oxygen groups loaded by RPB. In turn, these characteristics enhance the adsorption performance.
The breakthrough curves of ACs. ACs: activated carbons. Adsorption isothermal curve of ACs. ACs: activated carbons.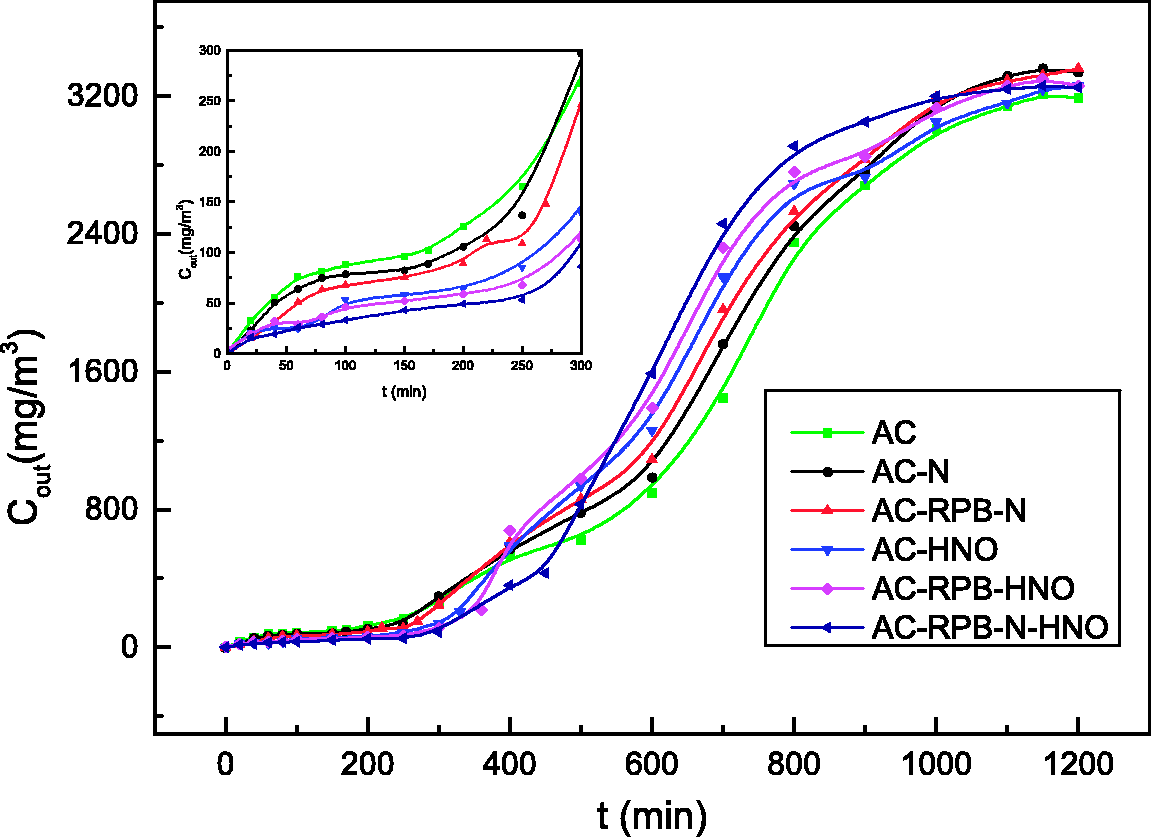
The adsorption of toluene on ACs.

ACs: activated carbon; RPB: rotating packed bed.
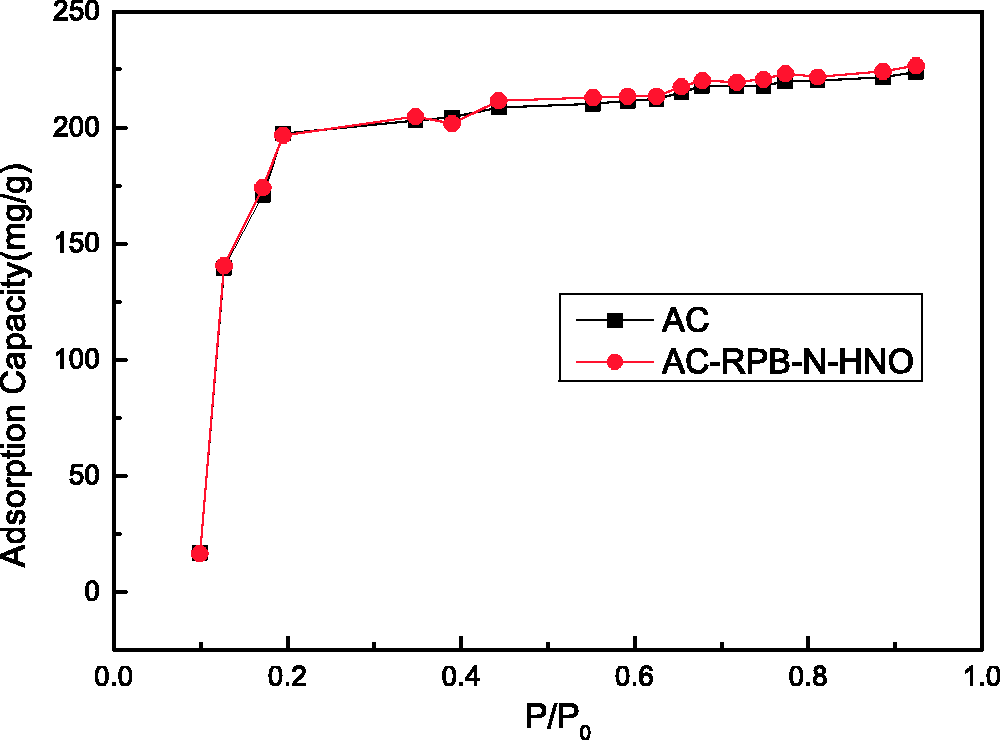
The adsorption isotherm for the adsorption of toluene by AC is type I, which indicates the monolayer adsorption mechanism (Rouquérol et al., 1994). The microporous structure of ACs plays a leading role in the process of adsorption. The adsorptivity of AC following compound modification by RPB is higher than that of the unmodified AC. Adsorption by AC involves micropore filling at the initial relative pressure, but transitions to multilayer adsorption on the pores’ surface under relatively high pressure.
The parameter of Langmuir and Freundlich isotherms equations.

AC: activated carbon; RPB: rotating packed bed; q: equilibrium adsorption capacity (mg·g−1); qm: adsorption capacity (mg·g−1); p: equilibrium pressure (Pa); b: Langmuir constant; K: Freundlich constant; c: equilibrium concentration (mg·m−3).
Within the experimental concentration range, the fitted correlation coefficient R2 of the Freundlich isothermal adsorption model is greater than the Langmuir coefficient, which illustrates that the Freundlich model can better describe the adsorption of toluene on the ACs. For AC-RPB-N-HNO, the value of n in the Freundlich model is less than for the unmodified ACs. The performance of the compound-modified ACs for adsorption of toluene is superior to that of the unmodified ACs. The ACs were thermally compound modified in RPB, and their physical and chemical properties were changed. Modification of ACs with N2 allows N2 to enter the ACs’ internal channels. Through this process of high-temperature inert-gas purging, the ACs’ internal channels become smooth and the blocked pores are opened, thereby increasing the micropore distribution and specific surface area. In the high-gravity environment, HNO3 is dispersed as fine droplets. Modification of ACs with HNO3 greatly increases the mass transfer area between ACs and HNO3, and thus enhances the loading of oxygen-containing functional groups.
The fitting of the Freundlich adsorption model is shown in Figure 9. The adsorption of toluene on ACs is a combined interaction involving both chemical and physical adsorption (Kim et al., 2006). The adsorptivity of the compound-modified ACs is greater than that of the non-modified ACs in the high-gravity environment.
Freundlich fitting figure. AC: activated carbon.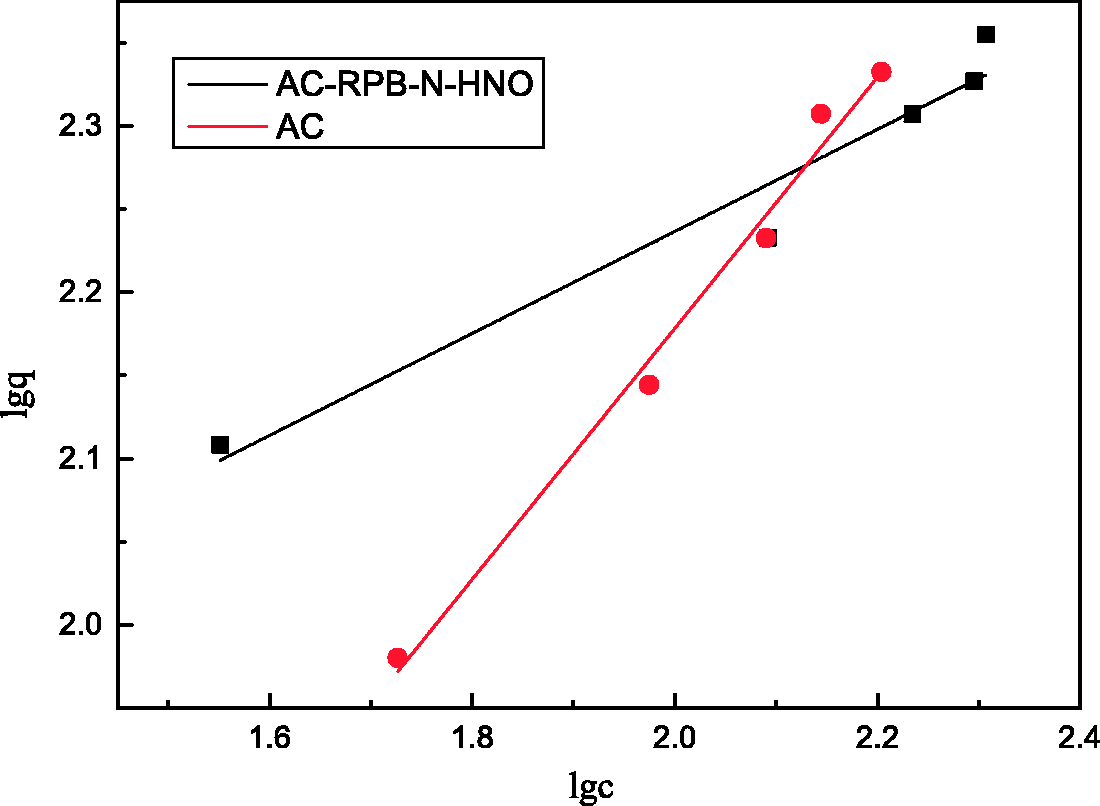
Coverage area and surface coverage of toluene on different ACs.

ACs: activated carbons; RPB: rotating packed bed.
Fraction of the surface covered

Conclusions
The structural characteristics and adsorption properties of ACs following physical, chemical, and compound modification in conventional and high-gravity environments have been studied. The specific surface area, pore structure, and functional-group content of ACs treated by high-temperature nitrogen and nitric acid modification were characterized, and these species were then applied in the adsorption of toluene.
The chemical modification of the ACs resulted in the loading of acidic oxygen-containing functional groups. The content of these groups in AC-RPB-HNO was 1.5 times greater than that of AC-HNO, suggesting that AC-RPB-HNO should be more able to adsorb toluene, a polar gas. The physical modification of the ACs at high temperature (473–573 K) resulted in a smaller average pore size (0.524 nm) and larger specific surface area (871.50 m2/g). These superior pore structures also contributed to the adsorption of toluene gas. The adsorption of toluene on the different ACs was studied. The ACs treated by high-gravity compound modification had the greatest adsorption rate and capacity (267.57 mg/g), the latter value being 3.2 times that of the unmodified ACs. It was also found that the microporous structure played a leading role in the process of the ACs’ adsorption of toluene. The excellent adsorptivity of the ACs resulted from the compound modification, which increased the amount of effective adsorption sites. The surface coverage of toluene on AC-RPB-N-HNO was 51.67%. Therefore, compound modification of ACs under high gravity can greatly improve the adsorption performance.
