Abstract
Introduction
Mild traumatic brain injury (mTBI) is a major public health concern, particularly in children, representing 25% of all TBI cases in Europe, 1 and one of the leading causes of pediatric emergency department (PED) visits. 2 Children are particularly vulnerable to mTBI, and their developing brains may exhibit different injury mechanisms and recovery kinetics compared to adults. The management challenges in pediatric mTBI are distinct, 3 as clinicians strive to avoid unnecessary radiation while ensuring accurate diagnosis.
Diagnosis of mTBI relies primarily on self-reported symptoms and signs collected during clinical assessments. 4 To detect potential intracranial injuries (ICI), imaging techniques such as computed tomography (CT) scans are employed;5,6 however, their use is often limited in children due to concerns about radiation exposure. 7 Consequently, clinicians frequently opt for monitoring the child’s symptoms over time rather than immediate imaging.3,8 While this observational approach aims to minimize unnecessary risks, it often prolongs the child’s stay in the PED, creating additional stress for both the patient and their family and contributing to overcrowding. At present, clinicians lack reliable bedside tools to confidently discharge children with mTBI during the acute phase of management. 9
mTBI can affect multiple brain structures, enhancing neuroinflammatory cascade and damaging the blood–brain barrier. 10 All these injuries can lead to the leakage of proteins into the bloodstream.11,12 Blood-based biomarkers have gained attention as a potential fast and cost-effective method for determining whether a patient with mTBI should undergo a head CT scan. Given the complexity and variability of mTBI, relying on a single biomarker for diagnosis may lack sufficient sensitivity and specificity. As a result, combining biomarkers into panels appeared to be more effective for better detecting diverse types of brain injuries.13,14 While combinations of biomarkers have been investigated in adult mTBI,15,16 their performance in the pediatric population remains to be explored.
In adults, glial fibrillary acidic protein (GFAP) is a well-established protein released by astrocytes and has been extensively studied in the context of mTBI. Notably, its combination with ubiquitin C-terminal hydrolase L1 (UCHL-1) has been explored to differentiate between patients with ICI on CT (CT+) and patients without (CT−) following mTBI. The combination of GFAP and UCHL1 showed a sensitivity of 97%, a negative predictive value (NPV) of 99%, and a specificity of 36% in mTBI patients.16,17 Another combination including GFAP has been tested in adult mTBI patients alongside heart fatty acid binding protein (HFABP). HFABP, a capillary-binding protein indicative of vascular brain injury, has been found elevated in adult mTBI patients with CT+ results. 18 The GFAP and HFABP combination improved specificity to 46% while maintaining 100% sensitivity. 15
The S100 calcium-binding protein B (S100b) is another protein associated with astrocyte damage and has been extensively studied in the context of mTBI.19,20 It has been included in both the Scandinavian
21
and French
22
guidelines for adults, based on its demonstrated ability to identify one-third of patients for whom unnecessary CT scans could have been avoided.23,24 Additional blood-based biomarkers that may be released from different brain cell components or cell types have also been investigated in mTBI.
25
They have been shown to help differentiate between CT+ and CT− patients.26,27 This includes proteins such as cytokines (interleukins) related to the neuro-inflammation triggered by the trauma; proteins involved in the neuro-cardiac axis, such as the
In children, S100b is to date the most described blood biomarker in mTBI patients, also showing a reduction of one-third of unnecessary CT scans.20,28,29 S100b is known to be age-related, with significantly higher physiological expression observed in the healthy pediatric population during the first three years of life. 30 In a randomized clinical trial with pediatric mTBI patients, S100b biomonitoring led to a reduction in both CT utilization and in-hospital observation time. 31 More recently, the combination of GFAP and UCHL1 has been evaluated in children, 32 demonstrating within 3 h post mTBI a sensitivity of 100% and a specificity of 67% in identifying clinically important TBI.
In addition, a recent study in pediatric TBI patients revealed altered cytokine profiles following mTBI, highlighting the effect of neuro-inflammation triggered by the trauma. 33
Based on these results, this study aimed to explore a selection of blood biomarkers in combination to enhance diagnostic accuracy in safely ruling out children without ICI after mTBI.
Materials and Methods
Study population
Children aged 0 to 16 years with an mTBI occurring within 24 h prior to their presentation at one of the participating PEDs were included. Five hospitals in Switzerland were part of the study (Geneva University Hospitals, Fribourg Hospital HFR, Neuchâtel Hospital [RHNE], University Children’s Hospital Zurich, and Inselspital, Bern University Hospital). Institutional review board approval was received; the study was conducted in accordance with Good Clinical Practice guidelines and provisions of the Declaration of Helsinki and was registered at www.clinicaltrials.gov: NCT06233851.
Informed consent was obtained from the parents or legal guardians of the children, as well as from the children themselves if they were 14 years of age or older. mTBI was defined by one of the following conditions: 3 Head trauma with (1) a Glasgow Coma Scale (GCS) score of 14; or (2) a GCS score of 15 with at least one of the following symptoms: loss of consciousness, post-traumatic amnesia (PTA), persistent headaches, irritability, three or more episodes of vomiting, confusion, dizziness or vertigo, post-traumatic seizure, or transient neurological abnormalities; or (3) evidence of a basilar skull fracture; or (4) severe mechanism of injury, such as a traffic accident or a fall from a height greater than 0.9 m in children under 2 years old, or over 1.5 m in children 2 years or older.
Exclusion criteria included participation in another clinical study involving pharmacological treatment, recent alcohol consumption or psychoactive substance use, a traumatic brain injury (TBI) within the past month, seizures within the last month, a diagnosis of Down syndrome, acute encephalopathy, encephalitis, meningitis, or refusal to provide consent.
A group of healthy children was also recruited by study nurses throughout the study period with obtained informed consent. Inclusion criteria were any child aged 16 or younger with a scheduled blood sample in the ambulatory care unit and without TBI. Exclusion criteria were the same as those defined for the TBI group.
Intervention and data collection
After informed consent was obtained, a blood sample was drawn as soon as possible, but no later than 24 h after the trauma. All participating hospitals followed the same protocol for blood collection, and a standardized centrifugation protocol was employed by each hospital laboratory to prepare frozen serum aliquots.
The study did not interfere with any medical decision-making. Study data were collected and managed using REDCap electronic data capture tools hosted at Hôpitaux Universitaires de Genève (HUG).34,35 Clinical records included sex, age, GCS score, causes of injury, history of coagulation disorders with medication intake prior to trauma, TBI-associated symptoms, simple skull fractures, presence of extracranial injuries (ECIs) such as other body fractures or organ injuries, time between trauma and blood sampling, physician decision regarding patient management (observation without CT scan or undergoing CT scan), effective time in observation in the PED for symptom monitoring, neurosurgery and intubation if needed, and results of imaging. The value of C-reactive protein, when available, was also recorded as a marker of inflammation and infection to ensure that such conditions did not influence our biomarker assessments.
Blood-Based biomarker analysis
Serum samples were obtained by centrifugation and stored locally at −80°C before being sent in batches to the University of Geneva for analysis. IL6, NfL, NTproBNP, GFAP, IL10, S100b, and HFABP concentrations were measured using enzyme-linked immunoassay: R-plex Human NfL (F217X), NTproBNP (F214I), GFAP (F211M), S100b (F212E), and FABP3/HFABP (F214T) antibody sets; and V-plex Proinflammatory Panel 1 (Human) with IL6 and IL10 antibody sets, on the MesoQuickPlex SQ120 reader (Meso Scale Diagnostics, Rockville, MD, USA). Lower limit of detection was, respectively, 5.5 pg/mL with a calibration range of 12.21–50,000 pg/mL for NfL; 0.30 pg/mL and 0.12–500 pg/mL for NTproBNP; 63 pg/mL and 122–500,000 pg/mL for GFAP; 1.6 pg/mL and 1.22–5,000 pg/mL for S100b; 90 pg/mL and 24.41–100,000 pg/mL for HFABP; 0.06 pg/mL and 0.06–488 pg/mL for IL6; and finally 0.04 pg/mL and 0.04–233 pg/mL for IL10. The lower limit of quantification (LLoQ) was determined as the minimum concentration with a coefficient of variation (CV) under 20% and a recovery rate between 80% and 120%. Duplicate control serum samples were analyzed on each plate, ensuring that intra- and inter-plate CVs remain below 20%. All kits were utilized following the manufacturers’ guidelines.
Outcome measures
The main outcome measure was the identification of ICI. To assess ICI, the study followed the Pediatric Emergency Care Applied Research Network (PECARN) criteria, 3 which include intracranial hemorrhage or contusion, cerebral edema, traumatic infarction, diffuse axonal or shearing injuries, sigmoid sinus thrombosis, midline shift, signs of brain herniation, skull diastasis, pneumocephalus, or a skull fracture depressed by at least the thickness of the skull table. A single pediatric radiologist (CH), who was blinded to both clinical details and biomarker results, reviewed all CT scans. Cases with any of these criteria were classified as CT+, while those without were considered CT−. For non-scanned mTBI patients, any secondary presentation to the PED due to symptom deterioration was recorded (patients’ hospital records remained accessible for review throughout the entire study period). In the absence of such presentations, patients were considered to have no ICI and were grouped with the CT-patients.
Statistical analysis
Statistical analysis was performed using R (http://www.rproject.org, version 4.4.1) in RStudio (http://www.rstudio.com, version 2024.09.0). Biomarker concentrations were normalized using their medians as correction factors. Patients were dichotomized into two groups: (1) patients with ICI on CT (=CT+); and (2) patients without ICI on CT or without CT scan but with in-hospital-observation (=CT− and Obs.). Differences between groups were established using the nonparametric Mann–Whitney
A separate analysis limited to only CT-scanned patients was also performed (Supplementary Data).
Results
Clinical parameters
A total of 419 mTBI children were included between October 2020 and March 2024 (Supplementary Fig. S1). Among these, 18 (4%) patients had an ICI on CT scan (CT+), 79 patients (19%) had a CT scan without ICI (CT−), and the remaining 322 patients (77%) were kept for observation without imaging (in-hospital-observation). None of the last group developed a subsequent ICI within the entire study period. Both the CT− and in-hospital-observation without imaging patients were grouped together (CT− and Obs.), and their clinical parameters were compared with the CT+ group (Table 1).
Clinical Parameters of mTBI Patients with or without ICI

Severe mechanism of injury, such as a traffic accident or a fall from a height greater than 0.9 m in children under 2 years old or over 1.5 m in children 2 years or older.
CT−, negative result on CT scan; CT+, positive result on CT scan (defined by PECARN criteria); GCS, Glasgow Coma Scale; ICI, intracranial injuries; Min, minimum; Max, maximum; mTBI, Mild traumatic brain injury; Obs., patients kept in observation without CT scan; PECARN, Pediatric Emergency Care Applied Research Network; SD, standard deviation; yo, years old.
The age of included children ranged from 1 month to 16 years in the CT− and Obs. group and from 9 months to 15 years in the CT+ group. The mean age across both groups was 8 years. Most patients had a GCS of 15 with associated symptoms (86% and 66% for the CT− and Obs. and CT+ groups, respectively). Signs of basilar skull fracture and severe mechanism of injury were reported in both groups, but significantly more in CT+ patients (33.3% and 77.8%, respectively). There were no significant differences in the reported symptoms at admission between the compared groups. However, the two most prevalent associated symptoms were PTA (28.9%) and persistent headaches (26.2%) in the CT− and Obs. group and persistent headaches (33.3%) and more than three episodes of vomiting (22.2%) in the CT+ group (Table 1). Extracranial injuries (ECI) were significantly more reported in the CT+ group and mainly corresponded to fractures of the facial bones. The presence of simple skull fractures seen on CT was also significantly higher in the CT+ patient group (
Out of the 18 patients with a CT+ result (as defined by the PECARN criteria), 14 (78%) had intracranial hemorrhage on their CT images. Most of these hemorrhages (67%) were located in the subdural region. Details of the specific ICI observed in CT+ patients are presented in Table 2.
PECARN Criteria for Positive CT Findings (List of ICIs)

CT, computed tomography; ICI, intracranial injuries; PECARN, Pediatric Emergency Care Applied Research Network.
Clinical parameters focusing solely on CT-scanned children are presented in Supplementary Table S1.
Individuals’ biomarker performances
The mean with standard deviation of all biomarkers, as well as the median with interquartile range and minimum and maximum values, are reported in Table 3. The median time between head trauma and blood sampling was 8 h for CT+ patients and 6 h for the other mTBI patients, with no significant difference (
Blood Biomarker Concentrations in mTBI Patients with or without ICI

CT, computed tomography ; GFAP, glial fibrillary acidic protein; HFABP, heart fatty acid binding protein ; ICI, intracranial injuries ; IQR, interquartile range; Min, minimum; Max, maximum; mTBI, Mild traumatic brain injury; NfL; neurofilament light chain protein; NTproBNP,
Blood concentration of IL6, NfL, GFAP, and S100b was significantly increased in CT+ patients compared with CT− and in-hospital-observation patients (Table 3).
The diagnostic performance of each individual biomarker is presented with ROC curves and their area under the curve in Figure 1A. At 100% SE, IL6 yielded the best SP of 47.6% [95% IC: 42.9–52.4%], followed by NfL with 29.4% [95% IC: 25.0–33.8%], GFAP with 25.7% [95% IC: 21.5–29.9%], and NTproBNP with 24.4% [95% IC: 20.3–28.6%]. IL10, S100b, and HFABP were not able to reach more than 20% of SP at 100% of SE (Fig. 1A and Table 4).

Biomarkers ROC curves discriminating mTBI patients with and without ICI (CT+ vs CT− and Obs.).
Biomarkers Performances Discriminating mTBI Patients with and without ICI (CT+ vs CT− and Observation) Individuals’ Biomarkers
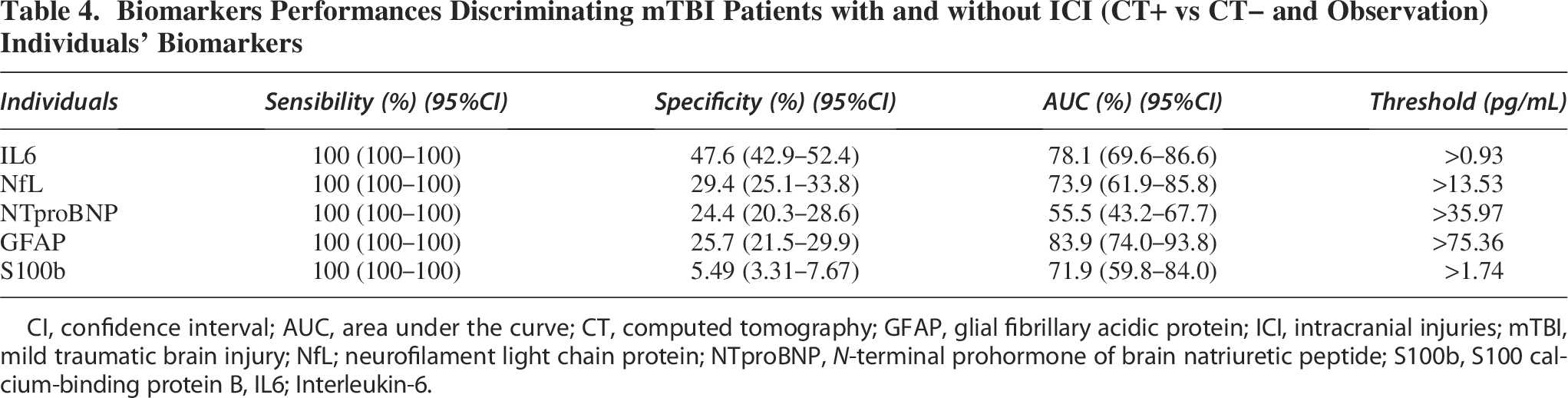
CI, confidence interval; AUC, area under the curve; CT, computed tomography; GFAP, glial fibrillary acidic protein; ICI, intracranial injuries; mTBI, mild traumatic brain injury; NfL; neurofilament light chain protein; NTproBNP,
S100b testing is recommended within 3 h following trauma. Among patients sampled within this time frame, only 5 were CT+ (and 92 were CT− or observed). Under this condition, at 100% SE, S100b yielded a SP of 53.26% [95% IC: 43.33–63.19%]. The diagnostic performance of S100b under this condition is reported in Supplementary Table S4.
In the analysis focusing solely on CT-scanned patients, NfL, GFAP, and S100b remained significantly increased in CT+ patients (
Combinations of biomarkers performances
Each possible duplex combination of biomarkers was tested for their capacity to rule out, setting the SE at 100%. IL6 was present in each of the three best duplex combinations (Fig. 1B and Table 5). In association with either NfL, NTproBNP, or GFAP, IL6 was found to safely identify CT− and in-hospital-observation patients with more than 55% SP. The duplex IL6 + NfL showed the best performance with a SP reaching 61% [95% IC: 56%–65%], followed by IL6 + NTproBNP (60% [95% IC: 55%–65%]) and IL6 + GFAP (57% [95% IC: 53%–62%]). Their predictive value was positive if at least two biomarker values exceed their respective threshold. ROC curves of the three best duplex, compared with individual proteins, are presented in Figure 1B. The other duplex combinations were below 50% SP when SE was set at 100%.
Biomarkers Performances Discriminating mTBI Patients with and without ICI (CT+ vs CT− and Observation) The Three Best Duplex Combinations

CI, confidence interval; AUC, area under the curve; CT, computed tomography; GFAP, glial fibrillary acidic protein; ICI, intracranial injuries; mTBI, mild traumatic brain injury; NfL; neurofilament light chain protein; NTproBNP,
A confusion matrix was built based on the prediction rules defined by the IL6 + NfL combinations (Table 6) and allowed to truly identify all CT+ (100% NPV) and truly discharged 33 CT− (out of 79) and 212 (out of 322) observed without imaging patients.
Confusion Matrix with IL6 + NfL Predictions

Obs., observation. CT, computed tomography; ICI, intracranial injuries; mTBI, Mild traumatic brain injury; NfL, Neurofilament Light chain protein; IL6, Interleukin-6.
In the restricted analysis of solely CT-scanned patient, the diagnostic performance of the duplex, including IL6 with either NfL, NTproBNP, or GFAP, was also improved (Supplementary Table S3b).
Age-Correlation in the healthy population
The age-correlation of each biomarker was investigated in a healthy group of 99 children, aged 1 month to 16 years (mean age 8 years). Blood biomarker concentrations in this group are available in Supplementary Table S5. Spearman correlation analysis revealed that GFAP, IL10, and S100b were negatively and significantly correlated with age. NfL and HFABP were also significantly age-correlated but with a low correlation coefficient (Spearman coef. of −0.22); and neither NTproBNP nor IL6 were age-correlated (Table 7).
Age-Correlation in the Healthy Population (

GFAP, glial fibrillary acidic protein; HFABP, heart fatty acid binding protein; NfL; neurofilament light chain protein; NTproBNP,
Discussion
This study explored both the individual and combined performances of seven blood biomarkers in distinguishing children with and without ICI presenting in a PED with mTBI. Among the 419 children included, 23% underwent CT scans, of whom 19% were found to have an ICI (representing 4% of the total cohort). These proportions are consistent with reported prevalence in the literature2,3,40–43 and underscore the relevance of finding reliable biomarkers to reduce unnecessary CT scans and minimize prolonged hospital stays for children without ICI.
The measurement of IL6 within 24 h of trauma successfully identified 47% of children without ICI while maintaining 100% SE in detecting those with ICI. These results confirm prior findings from intermediate analyses in this cohort. 36 But it also highlights the limitation of using a single biomarker, due to the complex physiopathology of mTBI.
Combining biomarkers aimed to increase diagnostic SP while maintaining high SE to safely discharge patients without ICI. Notably, the combination of IL6 with either NfL, NTproBNP, or GFAP significantly improved SP, with the IL6 + NfL panel reaching 61% SP and 100% SE. Retrospectively, in our cohort, without missing any patients with ICI, this combination would have allowed the safe discharge of 33 out of 79 children who underwent CT scans without having ICI and 212 out of 322 children who were kept under observation for symptom monitoring in the PED. These findings represent a significant clinical benefit by reducing 42% of unnecessary CT scans and decreasing 67% of observations in PED, with a 100% NPV. The rate of false positives in current clinical practice is inherently high, given that most children with mTBI do not have ICI. The IL6 + NfL panel, despite a SP of 61%, represents a significant improvement in patient management by providing an additional tool to support clinical decision-making.
Among previously studied biomarkers, S100b has been shown to identify 34% of CT− patients with 100% SE. Its short half-life in blood has led to recommendations for a maximum delay of 3–6 h for sampling after head trauma.20,22,28,44 In our study, S100b exhibited lower SP (below 20%) when assessed across the full cohort. Even when selecting only patients sampled within 6 h post-trauma, IL6 outperformed S100b, reinforcing IL6’s potential as a more robust marker in both early and late trauma windows. 36 This suggests that IL6 could be particularly valuable in real-world settings, where many patients present to the hospital more than 6 h after injury. In our cohort, patients arriving at the PED later than 6 h after trauma represented one quarter and should not be ignored.
Interestingly, although NfL is a promising biomarker in neurodegenerative diseases due to its high neuroSP, previous studies in adults have reported that it does not outperform the well-established S100b in ruling out ICIs. 45 The different results observed in our study may be partly explained by differences in the population studied. In elderly individuals, elevated blood NfL levels might be influenced by age-related processes and the possible presence of neurodegenerative conditions or neurological sequelae from past diseases. In the general pediatric population, such confounding factors are not observed. In addition, our study’s inclusion window of up to 24 h post trauma may have captured the more linear and gradual rise in NfL levels, related to its longer half-life compared with other biomarkers.
NTproBNP, mostly known for cardiac diseases, was found promising in duplex with IL6 in our study. Alone, NTproBNP is not sensitive or specific enough to detect ICI after mTBI. But its elevation in a subset of patients might reflect a distinct physiological response. NTproBNP is a cardiac stress biomarker, observed to be elevated in various neurological conditions, possibly due to neuro-cardiac axis following brain injury.
Importantly, IL6 also benefits from being independent of age, making it a versatile decision-making tool across pediatric populations. While both S100b and GFAP are known to be age-dependent, requiring adjustments for use in young children, IL6, along with NTproBNP, can be applied without age stratification, further enhancing its clinical utility.
Given that IL6 is a cytokine released during neuroinflammatory responses, its elevated level post-trauma is unsurprising, although cytokines are not currently used clinically in this context. Furthermore, its combination with brain-specific proteins such as NfL, GFAP, or NTproBNP enhances its SP, suggesting that pairing systemic inflammatory markers with proteins reflecting brain injury could yield a powerful diagnostic tool. 25 In cases of multiple traumas, where IL6 might be elevated due to injuries outside the brain, combining it with brain-specific markers could help isolate brain traumas. Furthermore, in our previous work, 36 we found no significant differences in IL6 performance whether we included only patient with isolated mTBI or those with additional ECIs.
The three proteins IL6, NfL, and NTproBNP are already widely available in routine clinical analysis for other conditions, making them readily applicable in clinical practice once their utility in mTBI is validated. GFAP is also available on established diagnostic platforms, which could expedite the integration of these biomarker panels into clinical guidelines for mTBI management.
Strengths and limitations
This study’s key strengths include its large cohort size, making it one of the largest studies of pediatric mTBI biomarkers to date, and its comprehensive biomarker analysis within 24 h of injury. The inclusion of both scanned and non-scanned patients enhances the generalizability of the findings, particularly in a pediatric population where radiation exposure is a critical concern and imaging rate is lower than in adult mTBI care.
Our study highlights the need to address not only CT-scanned patients but also the high majority (77% in this cohort) of mTBI patients kept under observation without imaging. Reducing observation times for these patients could significantly alleviate stress for families and reduce hospital costs.
Blood-based proteins circulating in the bloodstream are biomarkers easily collected and analyzed in laboratory hospitals. The use of blood-based brain biomarkers offers a noninvasive way to diagnose and monitor neurological conditions such as TBI and are less invasive compared with cerebrospinal fluid biomarkers. They are more convenient for repeated measurements and suitable for routine screening or monitoring.
However, this study also has limitations. The cohort contained relatively few CT+ patients, reflecting the fact that most children with mTBI do not have ICI. While this is consistent with clinical practice, it limits the power of our analysis to fully explore the diagnostic performance for more severe injuries. Indeed, future mTBI biomarker studies should focus on detecting clinically important traumatic brain injuries (ciTBI) rather than just ICI. To clearly incorporate blood-biomarker measurements into the widely used PECARN 3 decision-making algorithm, further studies should be based on the same inclusion criteria. However, our study differs in two points: (1) Patient population: PECARN recommendations are specifically tailored for patients with GCS of 15, while our study also included children with a GCS of 14. (2) Outcome definition: PECARN aims to rule out ciTBI, whereas our study focused on ICI as identified on CT (given the low prevalence of ciTBI and the large sample size required to study it reliably).
Given these differences, a direct comparison or combined predictive analysis was not feasible within the scope of our current study. Nevertheless, with the promising performance of IL6 in combination with either NfL, NTproBNP, or GFAP highlighted in our findings, we believe future prospective studies should be designed specifically to address the incremental value of blood biomarkers with established clinical decision rules. Developmental changes in children might influence biomarker variability. Therefore, a stratified age group comparison to further evaluate the correlation between age and biomarker levels would be necessary to adequately explore these developmental influences. However, our sample size was insufficient to perform these analyses with statistical robustness.
In addition, we only evaluated combinations of two biomarkers due to statistical limitations related to sample size. This prevented us from exploring more complex multi-biomarkers or clinical panels that could potentially yield even higher SP. However, given that IL6 and its combinations showed robust performance, expanding this work to include larger cohorts and more biomarkers should be a priority.
Lastly, while we used a research-based platform for measuring biomarker levels, routine clinical validation is needed to establish practical cutoff values for each biomarker. This limitation can be addressed in future studies, as the biomarkers assessed are already available on validated clinical platforms. In addition, we were not able to test UCHL1, a potentially relevant TBI marker recommended in combination with GFAP in adults, due to the lack of a reliable immunoassay for its measurement in our laboratory.
Ultimately, mTBI is not always mild. It presents in various ways, with diverse recovery trajectories. Researchers are working to refine its classification by identifying endopheno-types.46,47 In addition, combined blood biomarkers may contribute to a biological signature of the trauma.
Conclusions
This study confirms, in a larger pediatric cohort, the diagnostic potential of seven blood-based proteins to safely identify children without ICI after an mTBI. Among these, the combination of IL6 with either NfL, NTproBNP, or GFAP demonstrated strong diagnostic value, suggesting its potential integration as a clinical decision-making tool for the management of mTBI in children. Specifically, the combination of IL6 and NfL was able to safely rule out ICI in 61% of cases with 100% NPV, significantly reducing the need for unnecessary CT scans and shortening the length of stay in PEDs. These findings highlight the promising role of biomarker panels in optimizing pediatric mTBI management, though further validation in external cohorts is needed to confirm their broader applicability.
Authors’ Contributions
Methodology: A.C.C., V.P., J.C.S., S.M., and C.K. Software: A.C.C. Validation: A.C.C., J.C.S., and S.M. Formal analysis: L.G. Investigation: A.C.C. Resources: S.M., F.M., F.S., F.R., V.W., M.S., C.R.S., and C.H. Data curation: A.C.C. and V.P. Writing—original draft preparation: A.C.C. Writing—review and editing: all authors. Supervision: J.C.S. and S.M. Funding acquisition: J.C.S. and S.M. Conceptualization: A.C.C., V.P., J.C.S., S.M., and C.K. Data curation: A.C.C. and V.P. Formal analysis: A.C.C. Funding acquisition: J.C.S. and S.M. Investigation: A.C.C. and L.G. Methodology: A.C.C., V.P., J.C.S., S.M., and C.K. Project administration: A.C.C., V.P., J.C.S., S.M., and C.K. Resources: S.M., F.M., F.S., F.R., V.W., M.S., C.R.S., C.H., J.C.S., and V.P. Software: A.C.C. Supervision: J.C.S. and S.M. Validation: A.C.C., J.C.S., and S.M. Writing—original draft preparation: A.C.C. Writing—review and editing: all authors. All authors have read and agreed to the published version of the article.
Footnotes
Acknowledgments
The authors especially thank patients and their families for their participation in this study. The authors thank all the members of the t-BIOMAP study, clinicians, research nurses, radiologists, neuropsychologist, hospital laboratories, and case managers. The authors thank the Platform of Pediatric Clinical Research of the HUG for their invaluable help.
Author Disclosure Statement
The authors declare no conflicts of interest.
Funding Information
This research was funded by a private grant from Geneva University Hospitals (HUG) for its first year of recruitment.
Transparency,Rigor,and Reproducibility Statement
The study was registered at clinicaltrials.gov (NCT06233851), after the study began. The analysis plan was not formally pre-registered, but the team member with primary responsibility for the analysis (lead author) certifies that the analysis plan was prespecified. The study was set in a discovery-validation context aiming to evaluate ciTBI prevalence in children with mTBI, with a sample size estimation of 600, including controls and TBI patients, grounded in prior research and expected ciTBI event rates. Human participants were unable to guess the results of their fluid biomarker measurements. Handling of biofluid samples, measurements, quality control decisions, and analyses were performed by investigators blinded to relevant characteristics of the participants. Samples were acquired within a maximum of 24 h after the trauma, using a dry tube for serum. Samples were processed using centrifugation at 3,000 rpm for 10 min at room temperature. Samples were stored at −80°C prior to analysis; freeze-thaw cycles were performed once prior to analysis. All samples were analyzed at the same time in a single batch, with same lots of reagents. Quantitative spike-recovery in serum was performed by using a control serum measured in duplicate. The assay CV was below 20%. All equipment and analytical reagents used to perform measurements on the fluid biomarkers are widely available from MSD (Meso Scale Diagnostics, Rockville, MD, USA). The key inclusion criteria and primary clinical outcome measure are established standards in the field. Recruitment is completed, with a commitment to open access publication and future research permissions from a majority of participants, underscoring the study’s dedication to advancing the field responsibly. The study adheres to open access publication standards under Mary Ann Liebert Inc.’s appropriate license.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Geneva (CCER-ID: 2020-01533 on the 12 August 2020).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
