Abstract
Introduction
Peripheral neuropathies encompass a wide range of disorders affecting the peripheral nervous system. The symptoms of peripheral neuropathy vary according to the types and components of nerve fibres involved. Sensory symptoms include burning sensation, tingling, pins and needles, shock-like feelings, hyperalgesia and allodynia. As the sensory neuropathy progresses, it results in unstable gait and, in worst scenarios, foot ulcers, leading to amputation. Motor symptoms include distal weakness or atrophy. Autonomic symptoms may present as orthostatic hypotension, constipation or diarrhoea, difficulty in micturition or erectile dysfunction. 1
Due to diverse clinical presentations involving sensory, motor and autonomic systems, a systematic approach is important for diagnosis and management. The diagnosis of peripheral neuropathy is established clinically. Nerve conduction studies (NCS) play a pivotal role in the management of peripheral neuropathy. They are of prime importance in differentiating axonal and demyelinating neuropathy, and they also provide confirmatory diagnosis of distal sensory polyneuropathy. However, the widespread usage of NCS is limited due to its availability and they are also of less value in diagnosing small fibre neuropathy. 2
A simple and effective bedside tool that utilises clinical skills is always sought after for diagnosing peripheral neuropathy. A majority of the assessment scales for polyneuropathy were primarily focused on detecting the changes induced by impaired glycaemic control. 3 The Toronto Clinical Neuropathy Score (TCNS) is one of the valid and reliable scales for diagnosing and staging diabetic polyneuropathy and was later found to be useful in nondiabetic polyneuropathy. 4 The modified Toronto Clinical Neuropathy Score (mTCNS) was derived from the parent score, the TCNS. It includes both the symptom and sensory components. Another striking feature of this scale is that it assesses the dorsal column pathway (light touch, vibration, position sense), anterior spinothalamic tract (pin prick) and lateral spinothalamic tract (temperature). Thus, the mTCNS scale assesses both small fibre and large fibre neuropathy. 5
The gravity of peripheral neuropathy among people older than 55 years is high, and the estimated prevalence is nearly 8%, almost a fourfold increase from the general population. 6 Diabetes mellitus is the most common cause of peripheral neuropathy, and almost 50% of the patients with diabetes experience signs and symptoms of peripheral neuropathy. The prevalence of diabetic neuropathy among the Indian population ranges between 19.1% and 29.2%. 7
Published guidelines for treating diabetic peripheral neuropathy recommend tricyclic antidepressants, the serotonin-norepinephrine reuptake inhibitor duloxetine, and the anticonvulsants pregabalin and gabapentin as the first-line treatment. However, pregabalin and duloxetine are the only approved drugs for diabetic peripheral neuropathy. Despite these recommendations, comorbidities, contraindications and the use of concomitant medications with potential interactions pose a significant challenge in treating neuropathy. 8
Till date, most drug trials have focused on the adult population. Most of the clinical trials focus on pain and use the Verbal Rating Scale (VRS) for pain as the primary end point to assess the efficacy. A holistic end point to evaluate the efficacy of drugs in peripheral neuropathy is still lacking in the majority of the clinical trials. Also, we have limited information on the efficacy of and safety data on drugs used in neuropathic pain in the elderly. Considering the magnitude of peripheral neuropathy in the geriatric subset and the relative scarcity of pharmacological information on drugs used for this entity, we plan to conduct a study on geriatric patients suffering from peripheral neuropathy.
Methods
Study Design
The trial was a single centre, randomised, open-label, active-comparator pilot study with four treatment arms. The Institute Ethics Committee of All India Institute of Medical Sciences Raipur approved the study with reference to AIIMSRPR/IEC/2020/645 dated 13/11/2020. The study was also registered with the Clinical Trial Registry of India with reference to CTRI/2021/02/030935 dated 02-02-2021.
Participants
Eligible participants were patients of either sex attending the General Medicine Outpatient Department, aged 60 years or older, with peripheral neuropathy confirmed by the mTCNS (score > 11). The diagnosis was clinical. All participants provided written informed consent. Inclusion criteria were as follows: patients of either sex aged ≥60 years with mTCNS > 11 and not on any interventional drugs for the past eight weeks. Exclusion criteria included the following: age < 60years, patients with score ≤ 11 in mTCNS, patients not willing to give written informed consent, acute serious illness (like myocardial infarction, stroke, sepsis or electrolyte disturbances), known case of any malignancy, history of alcohol consumption (>100 gm/day) for more than three years, serum aminotransferase (ALT) > three times the upper limit of normal level, chronic kidney disease or estimated glomerular filtration rate (eGFR) < 45 ml/min/1.73 m2, vitamin B12 deficiency, history of raised intra-ocular pressure/acute angle closure glaucoma, history of any major psychiatric disorders, history of treatment with monoamine oxidase inhibitors in the past 14 days, and history suggestive of lower urinary tract symptoms or benign prostatic hyperplasia.
Randomisation and Allocation Concealment
The trial examined four treatment pathways: amitriptyline 10 mg, duloxetine 30 mg, gabapentin 300 mg and pregabalin 75 mg for 8 weeks. All the drugs were given once at bedtime. Randomisation was done by generating random numbers using Microsoft Excel 2016. Allocation concealment was done using sealed opaque envelop technique. The random numbers generated were marked on the outside of each envelope. Drugs were appropriately coded and were concealed within the envelope. Randomisation and allocation concealment was done by a person who was not part of the study.
Procedures
All patients were screened with routine blood investigations, that is, complete blood count, liver and renal function tests, glycaemic profile, vitamin B12 levels and a 12-lead electrocardiogram (ECG). After successful screening, randomisation was performed and trial medications were prescribed. Each treatment pathway consists of monotherapy for eight weeks. At the end of two weeks, dose titration was planned, which was at the sole discretion of the treating physician. Patients were also contacted telephonically every two weeks for assessing compliance and adverse effects. A final follow-up was scheduled at the end of the eighth week.
End Points
The primary end points of the study included the following: difference in the mTCNS from baseline to eight weeks and proportion of patients showing ≥50% reduction in the mTCNS from baseline to eight weeks. The secondary end points of the study included the following: difference in the VRS for pain and the Geriatric Depression Score (GDS) from baseline to eight weeks and recording of adverse events, if any, according to the prescribed proforma issued by the Pharmacovigilance Program of India (PvPI). Casualty assessment was done using the Naranjo Probability Scale, and severity assessment was done using Hartwig’s scale.
Statistical Analysis
The clinical trial being a pilot study, a sample size of 80 was chosen with 20 patients in each arm. Continuous data were presented using mean and standard deviation. Categorical data were presented in percentage/proportion. For the analysis, we considered the intention-to-treat method, which included all randomised participants who had taken at least a single dose of the drug.
9
Normality of the data was checked with the Shapiro–Wilk test.
Funding
All India Institute of Medical Sciences Raipur has funded the study, provided the interventional drugs for eight weeks free of cost and waived off the charges for the blood tests concerned with screening on academic grounds.
Results
Participants were recruited between March 15, 2021, and February 8, 2022. Follow-up continued till April 2022. During the recruitment period, 128 patients were screened for eligibility. The recruitment process suffered a serious setback due to the OPD shutdown from April to July 2021 because of the ongoing second wave of the COVID-19 pandemic. Out of 128 patients, 48 patients were excluded. The reason for excluding patients were as follows: prior treatment with trial drugs (
CONSORT Flow Diagram.

In the amitriptyline arm, 15 patients completed the study per protocol. Three patients had an inadequate response to the treatment and opted out of the study. One patient had excessive sedation and requested the physician for a change of treatment and was hence considered as a dropout and one patient was lost to follow-up. In the duloxetine arm, 12 patients completed the study per protocol. There were eight dropouts in this arm. One patient died due to a COVID-19 infection, one patient left the study due to severe headache, another patient left the study due to excessive sedation, one patient refused to participate in the trial further after two weeks of recruitment, two patients had inadequate response and hence opted out of the study, and two patients were lost to follow-up. In the gabapentin arm, a total of 14 patients completed the study per protocol. There were six dropouts in this arm. One patient exited the study due to inadequate response, and five patients were lost to follow-up. In the pregabalin arm, a total of 18 patients completed the study per protocol. Two were lost to follow-up in this arm.
In our study, the mean age of the patients was ̴ 65 years, with a range of 60–77 years, and a majority of them were males (56.3%). A majority of patients (61.3%) had normal body mass index (BMI). On average, the participants had at least two comorbidities. and most of them (62.5%) were diabetics. Among the diabetic population, the mean duration of diabetes was ̴ 10 years. The triad of diabetes mellitus, hypertension and dyslipidemia was found in 15 patients out of the 80 recruited patients (Table 1). Patients were assessed with three scales: the mTCNS, the VRS and the GDS, both at the baseline and at the end of the study. The baseline scores and change in scores at eight weeks of all the 80 recruited patients are depicted in Table 2 and Table 3 respectively.
Demographic and Clinical Characteristics of Participants.
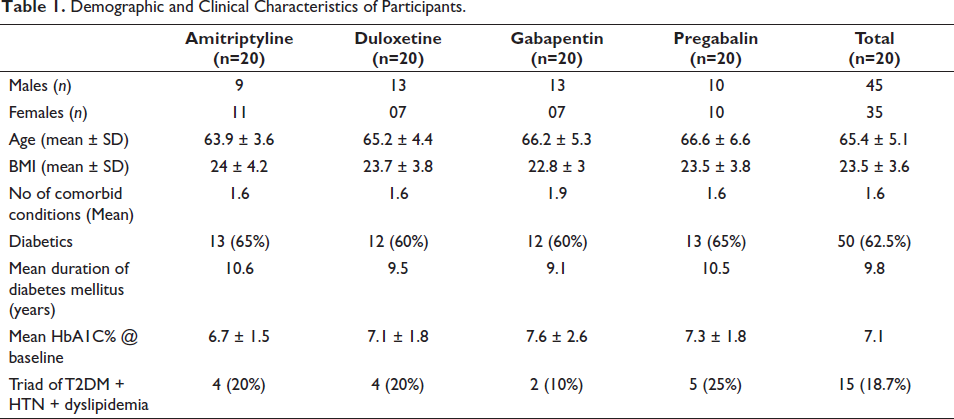
Baseline Scores of the Patients: Modified Toronto Clinical Neuropathy Score (mTCNS), Verbal Rating Scale (VRS), and the Geriatric Depression Scale (GDS).
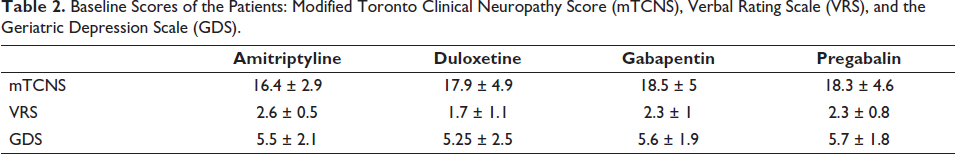
Change From Baseline in Different Scores at End Of the Study.
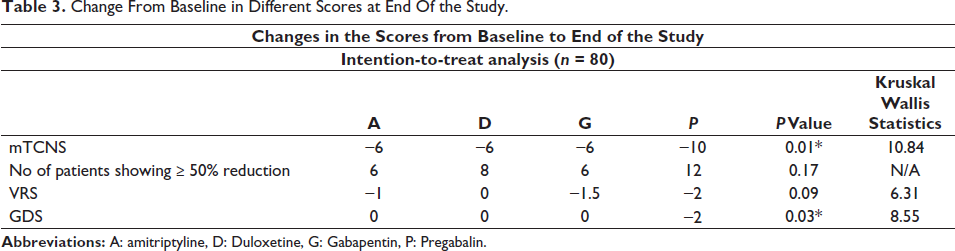
We observed improvements in the mTCNS at week 8 for all the four treatment pathways, with statistically significant differences between the amitriptyline and pregabalin arms (
Dunn’s Multiple Comparisons Test for mTCNS.

We also observed improvements in the VRS at week 8 for all the four treatment pathways, with no significant differences among the four treatment arms. We observed improvements in the GDS at week 8 for all the four treatment pathways; statistically significant difference was found between the amitriptyline and pregabalin arms (
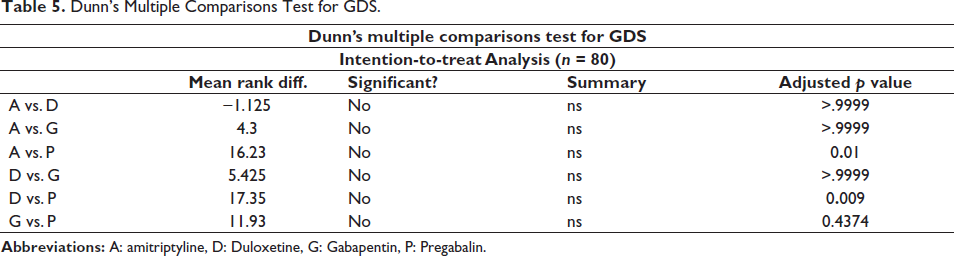
Dunn’s Multiple Comparisons Test for GDS.
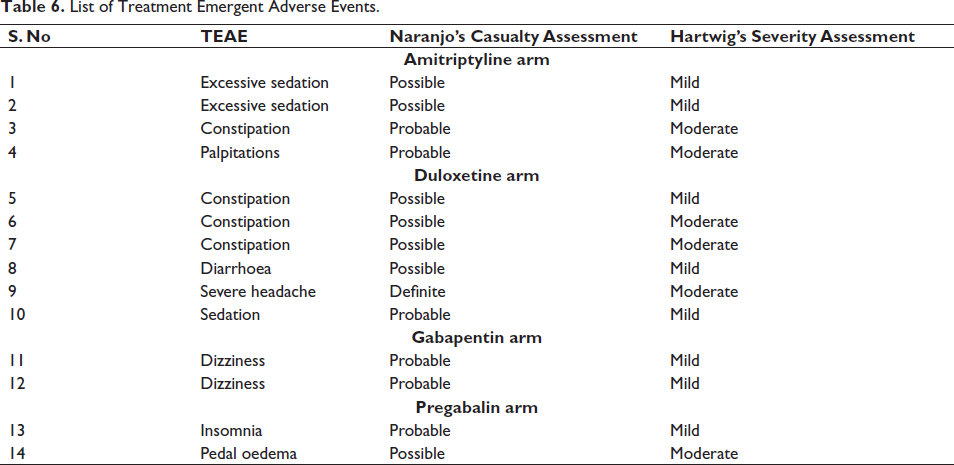
A total of 14 treatment emergent adverse events were noted, with the distribution of 4, 6, 2 and 2 in amitriptyline, duloxetine, gabapentin and pregabalin arms, respectively. According to Naranjo’s causality assessment, 1 ADR was definite, 6 ADRs were probable, and 7 ADRs were possible. According to Hartwig’s severity assessment, 8 ADRs were mild and 6 ADRs were moderate in severity. The most common system involved was the central nervous system followed by the gastrointestinal system and cardiovascular system. All the ADRs were reported PvPI. The list of ADRs, their causality and severity assessment are depicted in Table 6. All the adverse events were well known, and no serious adverse events were noted.
List of Treatment Emergent Adverse Events.
Discussion
The present study was conducted in a tertiary care teaching hospital of Central India to compare the first-line drugs for peripheral neuropathy in the elderly population. The demographic data in our study followed a similar trend published in the previous literatures.10–17 Our study showed that all four drugs yielded improvements in the mTCNS at the end of eight weeks with statistically significant differences between the amitriptyline and pregabalin and the duloxetine and pregabalin treatment pathways. The 50% responder rate in the mTCNS was similar among the four treatment arms (
In a randomised, double-blind clinical trial comparing the efficacy and safety of amitriptyline and pregabalin in diabetic peripheral neuropathy, it was found that pregabalin was comparable to amitriptyline in pain reduction. However, the incidence of treatment-emergent adverse events (TEAE) was more in the amitriptyline arm compared to the pregabalin arm. In the amitriptyline arm, the most common TEAE noted was increased sleep duration. In the pregabalin arm, peripheral oedema and daytime somnolence were the most common TEAE. The authors also observed similar adverse events and effectiveness in study. 18 In another randomised, double-blind, parallel-group clinical trial, the analgesic efficacy of pregabalin, amitriptyline and duloxetine was assessed in diabetic peripheral neuropathic patients. The results showed that analgesic efficacy was comparable among groups. We also reported a comparable response in VRS. However, pregabalin was superior to amitriptyline in reducing the severity of the neuropathy. The maximum TEAE was reported with pregabalin in this study. However, in our study, the duloxetine arm had a maximum number of TEAE. 19 Our results slightly differ from those of that study.
In another randomised, double-blind, cross-over, active-control trial, the efficacy and safety of duloxetine and amitriptyline were evaluated in diabetic peripheral neuropathic patients. Amitriptyline and duloxetine showed a similar reduction in pain. In our study, both drugs were comparable in pain reduction and reducing the severity of neuropathy. The incidence of TEAE among amitriptyline and duloxetine was comparable. We also reported a similar trend in the incidence of TEAE in our study. 20 Another randomised, double-blind study assessed the efficacy and safety of duloxetine and gabapentin in diabetic peripheral neuropathy. The effects of duloxetine and gabapentin were comparable in pain reduction as judged by the visual analog scale (VAS). The effects of gabapentin were similar to that of duloxetine. We also reported a similar finding; that is, gabapentin and duloxetine were comparable in reducing pain and the severity of neuropathy. In the study, the gabapentin arm had significantly more TEAEs than the duloxetine arm. However, we reported comparable TEAEs in both groups. Dizziness/imbalance was an important adverse event reported in the gabapentin arm in both studies. 21
Another clinical trial assessed the efficacy and safety of duloxetine and gabapentin in diabetic peripheral neuropathy. Both gabapentin and duloxetine showed a comparable reduction in pain as well as symptoms and severity of peripheral neuropathy. Treatment-emergent adverse reactions were also comparable between both arms. Our study is in excellent agreement with the present study regarding effectiveness and safety. 22 In a cross-over clinical trial, the efficacy and safety of pregabalin and gabapentin were evaluated with peripheral neuropathic patients due to chronic kidney disease requiring haemodialysis. In the study, the short-form McGill pain questionnaire (SF-MPQ) was used to assess pain. Although both drugs led to a significant reduction in pain, they were comparable to each other. The incidence of TEAEs was also comparable between both arms. Similarly, in our study, both drugs were comparable in terms of effectiveness and ADR profile. 23
In our study, pregabalin was statistically significant in improving features suggestive of depression according to the GDS compared to amitriptyline and duloxetine. Upon exploring the literature, pregabalin was found to have promising antidepressant effects.
Sleep disturbance is a major concern with chronic pain disorders like peripheral neuropathy. This may be attributable to the nocturnal awakenings due to outbursts of pain episodes. Pregabalin decreases midnight awakenings and sleep-onset latency, thereby increasing sleep quality. 24 It has been documented that neuropathy is a risk factor for depressive symptoms because of pain and unsteadiness. Further, it has also been recommended that screening for depression should be done during the baseline assessment for neuropathy. 25 Amitriptyline at low doses does not exert its antidepressant action, while duloxetine at low doses may have some antidepressant action. However, both the drugs require a minimum period of 8–12 weeks to show antidepressant action. Amitriptyline and duloxetine are effective in managing chronic pain irrespective of the features of depression, indicating their different mechanisms of analgesia. 26 Pregabalin causes better improvement in symptoms of peripheral neuropathy, and hence, the symptoms suggestive of depression might have been improved.
Our study was exclusively dedicated to the geriatric patients suffering from peripheral neuropathy and was conducted amidst several hardships caused by the COVID-19 pandemic. However, our study also had several limitations. The sample size is less in our study. Nevertheless, as per sample size calculation criteria for a pilot study, an adequate number of participants were enrolled in each interventional arm. 27 In the duloxetine treatment arm, there were more dropouts (8/20). The diagnosis of peripheral neuropathy was established clinically; hence, our study did not include the NCS. However, NCS was difficult to perform (considering COVID-19 pandemic) and due to financial constraints; hence, it was not carried out as a routine investigation in our study. The causes for non-diabetic peripheral neuropathy were not explored in our study. However, the patients were ensured adequate care and appropriate treatment by the treating physician. The number of patients lost to follow-up was relatively higher in our study, mainly due to the COVID-19 pandemic and the OPD shutdown for three months.
Conclusion
Pregabalin was found to be more effective when compared to amitriptyline and duloxetine in the treatment of peripheral neuropathy in elderly population as indicated by the reduction in the mTCNS scores. In terms of quality of life as judged by the Geriatric Depression Scale, a better response was seen in pregabalin arm when compared to amitriptyline and duloxetine arm. In elderly population, duloxetine caused more adverse events although none were serious adverse event. Future studies with adequate sample size will enhance the robustness and generalizability of the results.
