Abstract
Introduction
Sepsis is defined as life-threatening organ dysfunction resulting from a dysregulated host response to infection. Due to its high prevalence, high mortality rate, high cost, complex pathogenesis, and clinical heterogeneity, it has been listed by the World Health Organization (WHO) as one of the major global public health problems. 1 It is estimated that about 5 million people die from it worldwide each year. A dysregulated host response to infection causes it and is often accompanied by coagulation dysfunction and endothelial dysfunction. 2 In 2017, members of the Scientific and Standardization Committee (SSC) of the International Society on Thrombosis and Hemostasis (ISTH) developed the criteria for sepsis-induced coagulopathy (SIC). 3 SIC is a clinical syndrome characterized by life-threatening organ dysfunction and coagulation disorders, resulting from the host's dysregulated response to infection. Studies indicate that SIC patients diagnosed under new criteria have a mortality rate approaching one-third. 4 Without timely intervention, some may rapidly develop disseminated intravascular coagulation (DIC). Given SIC's high morbidity and mortality, early identification of high-risk groups and targeted interventions are crucial for improving sepsis patient outcomes. Early detection of SIC is therefore essential for effective sepsis management. The pathophysiological mechanisms of SIC are highly complex. Current research primarily focuses on identifying biomarkers with enhanced sensitivity and specificity to improve diagnostic efficiency. However, few biomarkers have been established as the gold standard for SIC diagnosis. Most existing biomarkers suffer from limitations such as low sensitivity, high testing costs, and prolonged turnaround times, making them impractical for clinical application. The established risk factors for SIC include elevated LDL-C levels, decreased absolute lymphocyte count, increased blood lactate levels, and elevated procalcitonin levels, all of which are independent risk factors for SIC. 4 This study investigates the risk factors for septic thrombosis through clinical baseline data, thromboelastography, and microbial characteristics, aiming to provide insights for clinical prevention of SIC and improve its prognosis.
Materials and Methods
Study Subjects
A retrospective analysis was conducted on the clinical data of 170 patients with sepsis admitted to the Intensive Care Unit of Xinjiang Medical University Affiliated Cancer Hospital from January 2023 to December 2024. Inclusion criteria: ① Met the diagnostic criteria for sepsis in Sepsis 3.0, ② Met the diagnostic criteria for sepsis-induced coagulopathy (SIC), ③ Age > 18 years. Exclusion criteria: ① Had a history of hematological tumor diseases ② Patients with severe liver or kidney diseases ③ Patients with splenomegaly ④ Patients with congenital deficiencies in coagulation factors or vitamin K, hemophilia ⑤ Patients with acute hemorrhage or thromboembolic events ⑥ Patients who received cardiopulmonary resuscitation within 12 h before diagnosis ⑦ Patients with a history of coagulation dysfunction ⑧ Pregnant or lactating women ⑨ Patients who were lost to follow-up or had incomplete clinical data. The diagnostic criteria for SIC: Iba 3 were proposed in 2017 as the diagnostic criteria for sepsis-induced coagulopathy (SIC), which is the first scoring system specifically designed for coagulation disorders in sepsis. This standard includes INR, PLT, and sequential organ failure assessment (SOFA) score. A total score of ≥ 4 points can diagnose SIC; among them, the SOFA total score is the sum of the SOFA scores of the respiratory, circulatory, liver, and kidney sections, and since the inclusion of the SOFA score is to confirm the presence of sepsis, even if the SOFA score is greater than 2 points, the SOFA score is limited to 2 points.
Ethics Statement
The Ethics Committee of Xinjiang Medical University Affiliated Tumor Hospital approved this study.Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article. (Urumqi, China, approval number: K-2024282)
Data Collection
This study systematically collected a variety of general data of the patients, including gender, age, body mass index, and baseline vital sign parameters. At the same time, information such as smoking history, drinking history, previous diseases and tumor type was also recorded. The analysis items included blood routine, coagulation function, thromboelastography, and bacterial culture results. In addition, clinical data such as SOFA score at ICU admission, APACHE II score, infection site and total hospital stay were also summarized. For IL-6 sample collection et al, the initial fasting blood sample taken within 24 h of the patient's ICU admission serves as the reference value., the initial fasting blood sample taken within 24 h of the patient's ICU admission serves as the reference value.
Detection of Hematological Parameters
For blood cell analysis and PCT measurement, the BECKMAN COULTER DxH800 analyzer was used; for blood gas analysis, the RADIOMETER ABL90FLEX analyzer was employed; and for thromboelastography, the HAEMOSCOPE 5000 SERIES thromboelastography analyzer was utilized.
Statistical Analyses
Data analysis was conducted using SPSS 27.0 statistical software. For measurement data that followed a normal distribution, the mean ± standard deviation ((x ± s)) was used to represent them, and t-tests were employed for comparisons between groups; for data that did not follow a normal distribution, the median (lower quartile, upper quartile) [M(QL, QU)] was used to represent them, and non-parametric tests were employed for group comparisons; for count data, frequencies were used to represent them, and χ2 tests were employed for comparisons between groups; indicators with statistical significance in the univariate analysis were included in the multivariate binary logistic regression analysis, and differences were considered statistically significant if
Results
Comparison of General Conditions Between the two Groups of Patients
This study screened the included and excluded criteria and finally included a total of 170 patients with sepsis. Among them, 131 were in the SIC group and 39 were in the non-SIC group. The incidence of SIC was 77.06%. There were no statistically significant differences in gender, age, body mass index, baseline vital signs, smoking and drinking history, treatment-related factors, infection site, SOFA score, and APACHE-II score between the two groups (
Comparison of the General Conditions of the two Groups of Patients ([(

Abbreviations: BMI, Body Mass Index; BSA, Body Surface Area; MAP, Mean Arterial Pressure; CRRT, Continuous Renal Replacement Therapy; CHD, Coronary Heart Disease.
Comparison of Thromboelastogram Between the Two Groups of Patients
In terms of thromboelastography, the R value (min) in the SIC group was higher than that in the non-SIC group, and the difference was statistically significant (
Comparison of Thromboelastography Between SIC Group and non-SIC Group([(

Abbreviations: R, Reaction Time; K, Kinetics Time; a, Alpha Angle; MA, Maximum Amplitude; LY30, Lysis Index; CI, Coagulation Index; GK, Kinetics Time (K) and G Value (G).
Comparison of Laboratory Indicators Between the Two Groups of Patients
The absolute count of lymphocytes and the concentration of magnesium ions in the SIC group were lower than those in the non-SIC group, while the levels of interleukin-10 and interleukin-6 in the SIC group were higher than those in the non-SIC group, and the differences were statistically significant (
Comparison of Laboratory Indicators Between the two Groups of Patients([(

Abbreviations: WBC, White blood cell count; NEUT, Neutrophil count; LYMPH, Lymphocyte count; Mono, Monocyte count; RBC, Red blood cell count; HGB, Hemoglobin; HCT, Hematocrit; PLT, platelet count; PCT, platelet; CREA, Creatinine; ALT, Alanine Aminotransferase; AST, Aspartate Aminotransferase; APTT, Activated Partial Thromboplastin Time; APACHE, Acute Physiology and Chronic Health Evaluation; CRP, C-reactive Protein; TNF-a, Tumor Necrosis Factor-alpha; PT, prothrombin time; TT, Thrombin Time; FBG, Fibrinogen; SOFA, Sequential Organ Failure Assessment; TT, thrombin time; PT-INR, Prothrombin Time - International Normalized Ratio; PH, Potential of Hydrogen; IL-12p70, Interleukin-12, p70 heterodimer; IL-2, Interleukin-2; IL-6, 6-Interleukin; IL-1β, 1β-Interleukin; IL-10, 10-Interleukin; IL-γ, γ-Interleukin; IL-17, 17-Interleukin; IL-4, 4-Interleukin; IL-8, 8-Interleukin.
Comparison of Bacterial Culture Results Between the SIC Group and the non-SIC Group(n,%).

Multi-Factor Analysis of the Impact on SIC
The tumor types, surgical history, and laboratory indicators (absolute value of lymphocytes, interleukin-10, interleukin-6, and magnesium ions) that had statistical significance in the single-factor analysis were included in the multivariate binary logistic regression analysis. There was no multicollinearity among the above indicators. The results showed that interleukin-6 was an independent risk factor for SIC (
Multivariate Logistic Regression Analysis of Sepsis-Induced Coagulopathy.

Abbreviations: IL-10, interleukin-10; IL-6, interleukin-6.
ROC Curve Analysis
The AUC values for predicting the occurrence of SIC by lymphocyte absolute count and interleukin-6 were 0.716 and 0.963, respectively (
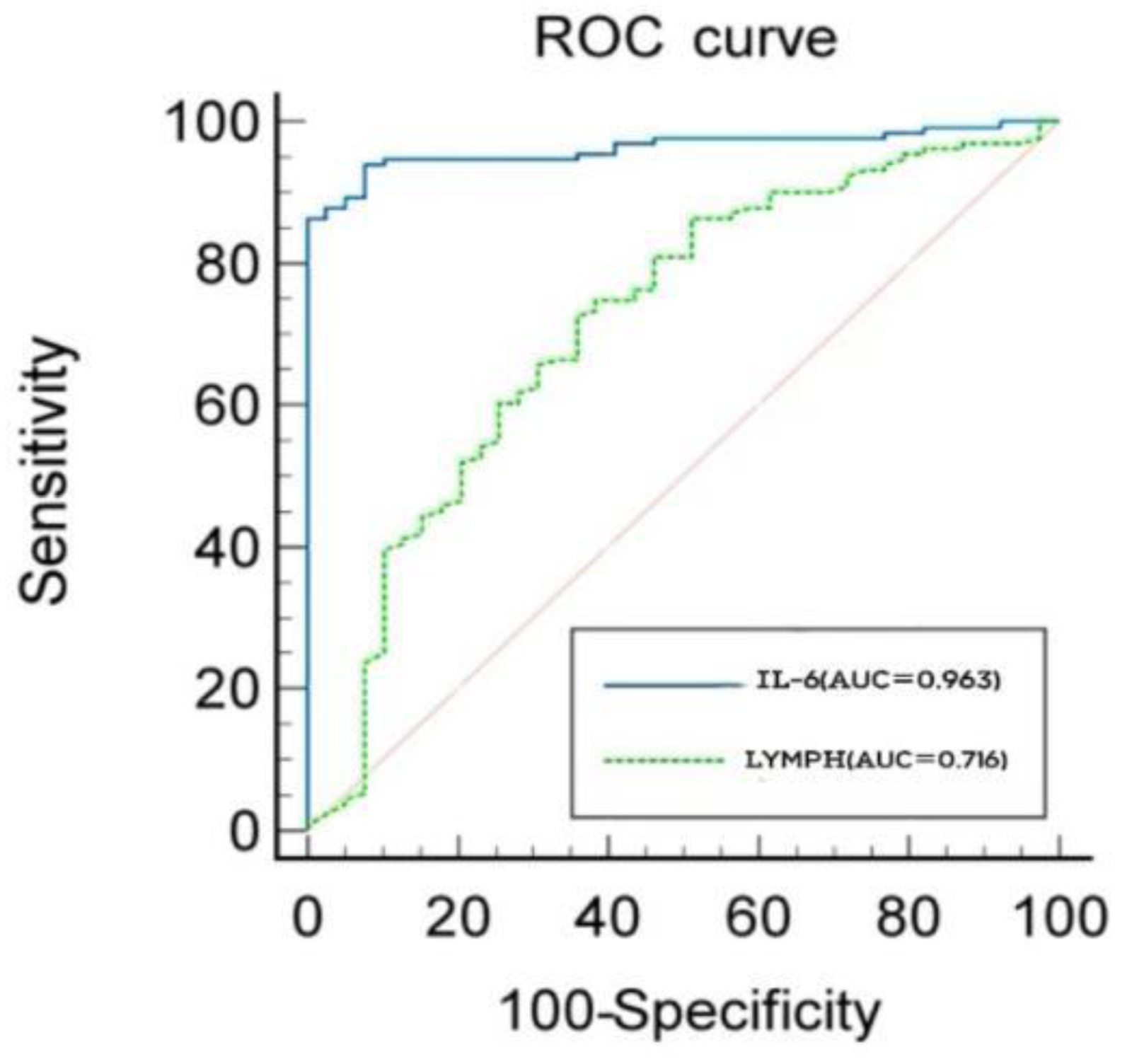
The receiver operating characteristic (ROC) curve of LYMPH and IL-6 for SIC.
ROC Curve Analysis of Lymphocyte Absolute Count and Interleukin-6 for Predicting SIC.

Abbreviations: AUC, area under curve; IL-6, interleukin-6; LYMPH, lymphocyte absolute count
Discussion
Our results demonstrated that the SIC group exhibited significantly prolonged R values (7.83 ± 3.65 vs 4.40 ± 2.70,
This study had some unavoidable limitations. Firstly, although this study verified known risk factors such as interleukin-6, the predictive value of certain indicators (such as PCT) was inconsistent with previous studies,4,24–27 in this study did not find a significant association, suggesting that it may be related to population race differences, sample size, and as a single-center retrospective study. secondly, it suffers from small sample size and potential bias.This study employed a randomized design (using the random number table method) and calculated the sample size through a pre-conducted power analysis,since no significant abnormalities in coagulation function were observed in the non-SIC group, TEG data could not be collected. Ultimately, this study recruited a small number of patients as a non-SIC.In the future, more multicenter and large-sample prospective studies are needed to conduct an in-depth exploration. In particular, future multicenter prospective studies with a double-masked design can be used to control for confounding bias.
Conclusion
The lymphocyte count and the IL-6 have certain predictive value for whether sepsis patients develop SIC. It is particularly noteworthy that IL-6 is not only an independent risk factor (OR = 1.004), but also shows extremely high predictive efficacy (AUC = 0.963). In clinical practice, when IL-6 levels rise, the possibility of SIC should be vigilant, and its continuous increase may also indicate a poor prognosis for SIC patients.
