Abstract
Keywords
Introduction
Heavy metals are non-biodegradable substances that accumulate in the tissues of living organisms, entering food chains and ultimately being absorbed by humans. This distinguishes them from other toxic contaminants. Even small quantities of heavy metals can inflict severe neurological or physiological harm on the human body (Wong et al., 2003). Iron is one of the most abundant metals in the Earth’s crust and among the most common inorganic contaminants. Iron is the most common element found naturally in water resources in soluble forms, such as ferrous iron (bivalent iron, Fe(II) or Fe(OH3) or complexed forms like ferric iron (trivalent iron, Fe(III)) found in precipitates such as Fe(OH)3. Additionally, iron can exist in water in bacterial forms. The presence of iron in water makes it unable to be used directly as drinking water (Brown & Calas, 2011). High concentrations of iron will alter the water taste and may cause liver damage, gastrointestinal problems like nausea and diarrhea (Jomova et al., 2022). It may also cause hemochromatosis conditions, which result from chronic iron accumulation in body tissues (Shander et al., 2012). Therefore, the World Health Organization (WHO) stated that iron concentration should be reduced when it is present in a concentration above 0.3 mg/L (Edition, 2011; Talaat et al., 2010). The water should be subjected to treatment when the concentration of iron exceeds the previously mentioned concentration level before drinking. Excessive iron concentrations in surface and groundwater not only threaten public health but may also cause significant environmental degradation (Ghosh et al., 2020). High levels of iron can change the physical and chemical properties of aquatic environments by encouraging the creation of iron oxides and hydroxides (Chen et al., 2025). These compounds build up in sediments and make the water less clear. This mechanism limits sunlight penetration, disrupts photosynthesis, and in the end, hinders the growth of aquatic plants and algae (Lu et al., 2020). Consequently, reducing iron concentrations in natural water bodies is crucial for both human safety and the preservation of aquatic ecosystems, as well as maintaining ecological equilibrium.
Numerous researchers have explored strategies for reducing iron concentration in drinking water. Recent field-applicable technologies incorporate adsorption, oxidation/filtration, enhanced coagulation/filtration, ion exchange, reverse osmosis, membrane distillation, enhanced lime softening, activated alumina, and hybrid methods (Chowdhury et al., 2016; Pal, 2015). Hameed et al. (2019) have evaluated different techniques for the removal of iron from groundwater at pH 7.92, water temperature 27.2 degrees Celsius, and contact time 30 min (Hameed et al., 2019). Their results showed that the iron removal efficiency could reach 99.7%, 92.4%, 83.1%, 76.4%, and 26.2% in the case of applying biological treatment, Birm filtration, greensand filtration, Zeolite filtration, and chlorination, respectively (Hameed et al., 2019). Desta et al. (2022) have evaluated the removal of iron from groundwater using laboratory-scale aeration and sand filter, where the iron concentration has lowered from 5.79 mg/L to 5.79 and 4.11 mg/L (Desta et al., 2022).
Adsorption is another technology that can be applied to remove iron from water. Dey et al. (2022) used plants leaf biosorbents for the removal of iron from synthetic water (Dey et al., 2022). Their results showed that the mango leaf biosorbent has the best performance for iron removal, reaching removal efficiency of 100% at pH 5.5, contact time 60 min, temperature 25 degrees Celsius, rotation speed 150 rpm, and dosage 4.9 g (Dey et al., 2022). Activated carbon is used as an adsorbent due to its highly porous characteristic, where, the positively charged Fe2+ ion will be adsorbed and attached to the negatively charged surface of activated carbon (bin Jusoh et al., 2005). Thinojah and Ketheesan (2022) have evaluated the removal of iron from synthetic groundwater (SGW) using laboratory-scale oxidation and granular activated carbon (GAC) filter (Thinojah & Ketheesan, 2022). The maximum iron removal efficiency of 88% was achieved in 240 min and the maximum reported adsorption capacity of GAC was 0.17 mg Fe/g GAC (Thinojah & Ketheesan, 2022). Birm is another material that can be used for iron removal from water. Birm is sand coated with manganese and could oxidize iron, and it has its way in practical uses in the treatment of municipal water as well as the treatment of water used for drinking. Birm material have an effective particle diameter of 0.48 mm and percentages of MnO2 25% to 45%, this medium known as a catalyzing agent to oxidize iron (Hameed et al., 2019). It is an effective and economical way to reduce dissolved iron compounds from the raw water supply, where it acts as an insoluble catalyst to promote the interaction between dissolved oxygen (DO) and iron compounds. Birm’s physical properties provide excellent filtration modes that can be easily cleaned by backwashing to remove the deposit. Birm is not consumed in the iron removal process, thus providing a tremendous economic advantage over many other iron removal methods (Barlokov, 2010; Lim, 2021). The iron removal efficiency could reach 92% using birm filter material (Hameed et al., 2019).
Electrocoagulation is another method used to remove iron from drinking water. Vasudevan et al. (2009) have applied the electrocoagulation process, where stainless steel was used as the cathode and aluminum alloy was used as the anode (Vasudevan et al., 2009). The maximum iron removal efficiency of 98.8% was achieved at pH 6.5 and a current density of 0.06 A dm−2 (Vasudevan et al., 2009). Ion Exchange Technology is another iron removal technology used in water treatment. Ion exchange process removes nitrate ions, heavy metals, as well as undesirable calcium and magnesium ions that are responsible for hardness of water. Fink (2018) stated that the conventional ion exchange resin made from 17 phenol, acrylates or polyamines could be further modified and designed accordingly to allow higher selectivity of specific active site towards certain metal ions. Polymeric resin will interact with the dissolved ion in the water by attracting the ion of opposite charge. The attractive force and bond strength between the attached ion and the functional group are relatively weak (Musa et al., 2023). Therefore, ion exchange process occurs continuously through trapping of particular ion and releasing of another ion in water. Other than chemical precipitation, ion exchange, and adsorption process, oxidation is another conventional technology that has been widely adopted in water treatment plants for iron removal (Khatri et al., 2017). Oxidation is a process that involves electron transfer (Shammas et al., 2005). Chemical oxidation is a treatment process that converts the dissolved ferrous iron (Fe2+) in water into ferric iron (Fe3+) with the aid of oxidizing agents. Oxidation of iron in a water treatment plant can be carried out using liquid oxidants such as sodium hypochlorite, potassium permanganate, or hydrogen peroxide, or using dissolved gases such as oxygen, chlorine, and ozone (El Azher et al., 2008). The chemical oxidation method is considered superior to other technologies due to its capability to effectively remove iron from water (Elsheikh et al., 2020). Besides that, most of the chemical oxidants used for iron removal have powerful antibacterial effects and be able to act as strong disinfectants in water treatment plants (El Araby et al., 2009; Villamena, 2013).
Each treatment method mentioned above presents distinct advantages and limitations depending on some factors, such as operational requirements and cost constraints. Adsorption systems are simpler to design and operate, requiring low energy to operate (Belcaid et al., 2022, 2023). Oxidation and filtration techniques enhance the limited adsorption capacity by transforming soluble Fe²+ into insoluble Fe(OH)3, facilitating easy filtration, attaining elevated removal rates, and reducing regeneration requirements (Khatri et al., 2017). Compared with adsorption and ion exchange, oxidation/filtration offers accelerated reaction kinetics, reduced operational expenses, and greater scalability. It does not necessitate frequent regeneration or costly resins, and it effectively eliminates both dissolved and colloidal forms of iron (Gkika et al., 2025). Pre-oxidation and filtration overcome resin fouling by eliminating iron before ion exchange, hence decreasing maintenance requirements (Fabris et al., 2007). Oxidation/filtration is more economical and environmentally sustainable, as it circumvents the brine-disposal issues associated with ion exchange (Amini et al., 2015). Electrocoagulation eliminates the reliance on chemicals of oxidation and ion exchange by generating coagulants through electrochemical processes, thereby minimizing the need for chemical handling and storage (Ghernaout et al., 2011). However, oxidation-filtration systems surpass electrocoagulation in energy efficiency and scalability, particularly for rural or decentralized applications (Mao et al., 2023). Biological treatment surpasses the chemical reliance of oxidation and coagulation techniques by employing natural microbial catalysis, hence substantially lowering operational expenses (xiangyu et al., 2025). However, oxidation-filtration systems provide expedited and more manageable performance, rendering them more appropriate for rapid or high-demand treatment situations (Khader et al., 2024).
A pilot-scale plant was developed by some researchers for iron removal from water. Galangashi et al. (2021) have developed a pilot fluidized bed column containing greensand, where a 41.74% iron removal efficiency was achieved in a bed height of 19 cm (Galangashi et al., 2021). Bolobajev et al. (2022) have developed a pilot scale system containing hydrous manganese oxide slurry for iron removal from drinking water, where a 97% iron removal efficiency was achieved (Bolobajev et al., 2022). Pruss et al. (2021) have developed a pilot-scale system that consists of aeration and filtration units, where the iron concentration was reduced on average by 72% (Pruss et al., 2021). Another pilot-scale system was constructed to treat water quality extracted from the riverbank filtration (RBF) unit, which consists of oxidation and filtration units, where a 90% iron removal efficiency was achieved (ElHadary et al., 2024).
Despite extensive research on various iron removal techniques, including adsorption, ion exchange, biological treatment, and electrocoagulation, most studies were performed at the laboratory level, concentrating on discrete processes with limited practical application. Moreover, few studies have explored the integration of oxidation, aeration, and multi-stage filtration at a pilot scale to study iron reduction in drinking water. The economic feasibility of integrated systems and their operational optimization using modern data-driven methodologies remains little investigated. Consequently, this research was conducted with three principal aims: (1) To design and construct a pilot-scale integrated water treatment system that incorporates oxidation, aeration, and filtration (Birm, activated carbon, and sand) for efficient iron reduction, (2) To assess the impact of critical operational parameters, such as chlorine dosage, oxidation duration, aeration time, and filtration sequence on overall treatment efficacy, and (3) To create an artificial neural network (ANN) model that can predict and optimize iron removal efficiency under diverse conditions, accompanied by an economic evaluation to ascertain system viability. The objectives together seek to develop a technically and economically feasible solution for reducing elevated iron levels in drinking water, especially in areas where iron contamination remains a persistent issue.
Materials and Methods
Synthetic Water Sample Preparation
The first step in the lab work was to prepare water samples with a specific concentration of iron using iron solution with a concentration of 5,000 mg/L from SDFCL brand. Water samples with iron concentrations ranging from 1.0 to 1.5 mg/L were prepared through a two-stage dilution process to ensure accuracy. First, an intermediate standard of 50 ± 0.05 mg/L was prepared from the commercial 5,000 mg/L stock solution. This intermediate was then diluted to the target concentrations. The final diluted samples were verified via atomic absorption spectroscopy before use, confirming concentrations within a narrow margin of error (e.g., 1.0 ± 0.002 mg/L). Water samples with iron concentrations ranging from 1.0 to 1.5 mg/L were prepared to check the efficiency of the system in removing iron from water. Water samples were collected every treatment stage to determine the efficiency of each unit in the system. The collected water samples were analyzed for iron in the drinking water laboratory at the Housing and Building National Research Center (HBRC) using a Thermo Scientific iCE 3000 Series Atomic Absorption Spectrometer. The analysis was conducted at a wavelength of 248.3 nm with a spectral bandwidth of 0.2 nm, using an air-acetylene flame. A multi-point calibration curve was established using standard solutions ranging from 0.1 to 2.0 mg/L, prepared from a certified stock solution, and the correlation coefficient (R²) was maintained above 0.999. All measurements were performed in triplicate to ensure precision. Prior to analysis, an acid with a ratio of 1:1 HNO3:H2O was added to the samples (Alvarez-Bastida et al., 2018).
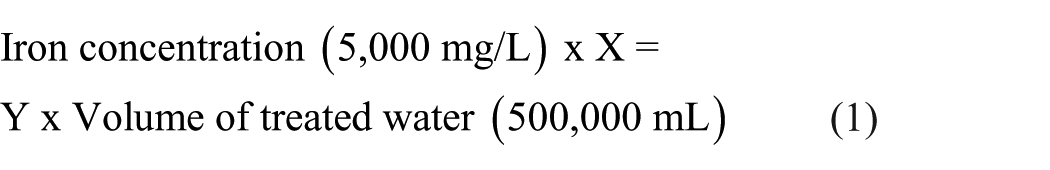
where X is the required quantity of solutions in mL and Y is the required concentration of iron in mg/L.
Design of the System
The system includes three main stages: oxidation, aeration, and filtration (birm, activated carbon, and sand), as shown in Figure 1. The filtration train consists of an activated carbon filter followed by a sand filter and finally a Birm filter. This sequence was specifically chosen because the primary role of the activated carbon unit is to remove residual chlorine, thereby protecting the manganese dioxide coating of the downstream Birm media from oxidative degradation. The sand filter in this configuration serves as a polishing step to capture any fine particles or carbon fines, ensuring final effluent clarity. The mobile plant was contained in a trailer (length: 4.80 m; height: 1.70 m; width: 1.00 m). The system includes the following elements: three tanks of food-grade polyethylene, two pumps CALPEDA brand (Qpump equal 2 m3/h and Hpump equal 30 m, four air pumping devices, a chlorine injection pump with a chlorine tank. The filtration part consists of a multi-layer sand filtration filter, a multi-layer carbon filtration filter, and a multi-layer Birm filtration filter. All the filters have a diameter of 10 inches, a height of 54 inches, and a maximum pressure of 10 bar with a manual control system. The oxidation part includes two options. The first option is oxidation by oxygen in the air, achieved by pumping air into the raw water. The second option is oxidation using chlorine. The chlorine used in this study had a concentration of 6%. All units are made of polyvinyl chloride (PVC). The equalization tank has a diameter of 75 cm and a height of 120 cm. These tanks also contain a mechanical pump to help achieve the flow of water from the main tank (raw water tank) to treatment tanks, and an additional pump to deliver water to the filter, which is the final stage in the system. The filtration part of the system consists of three stages. The first stage is the multi-layer sand filter, followed by the carbon filter and the birm filter. Birm material has an effective particle diameter of 0.48 mm and percentages of MnO2 25% to 45%. Each filter has a specific mission; the multi-layer sand is operative in removing large particles, suspended solids, and particles such as clay and silt that cause turbidity in water (Kalay et al., 2021). Carbon filters were used to remove chlorine, dissolved organic substances in water, color, taste, and smell (Liu et al., 2022). The last filter has the birm material, which is sand coated with manganese and can oxidize iron. The system was designed, constructed, and tested in the period from February 2024 to June 2025.

Pilot-scale system that consists of oxidation and filtration units.
Treatment Scenarios
The four treatment scenarios were designed systematically to isolate the effect of key operational parameters. Scenario 1 established the baseline influence of chlorine dose. Scenario 2 built upon this by investigating the effect of extended oxidation time using the optimal dose. Scenario 3 introduced aeration to evaluate its role in enhancing floc formation and finalizing oxidation after the optimal chemical conditions were set. Finally, Scenario 4 tested the system’s maximum performance by applying double filtration under the most favorable preceding conditions. This sequential approach allowed for a clear attribution of the iron removal efficiency to each specific treatment intensification. Figure 2 shows the different treatment scenarios. The first scenario was to apply chlorine with a dose ranging from 0.21 to 0.63 mg/L, oxidation time 60 s, and a single filtration of 60 s. The second scenario is to apply chlorine with a dose of 0.63 mg/L, followed by a single filtration of 60 s. The third scenario is to apply chlorine with a dose of 0.63 mg/L and oxidation time 240 s, aeration time ranging from 5 to 15 min, and a single filtration of 60 s. The fourth scenario is like the third scenario, but a double filtration is applied simultaneously for 60 min. All scenarios were conducted with an initial concentration of iron ranging from 1 to 1.5 mg/L. All experiment runs were conducted in triplicate. The main objective of all previous scenarios was to allow the Ferric Hydroxide to form according to the reaction act on Equation 2 (Mahasti et al., 2019). To evaluate the efficiency of each treatment stage and isolate the effect of oxidation from adsorption in the carbon filter, water samples were collected at multiple points: after oxidation, after aeration, and after each filtration unit. Crucially, samples taken immediately before the activated carbon filter were vacuum-filtered through Whatman No. 42 filter paper to separate the formed ferric hydroxide precipitates. The analysis of this filtrate confirmed that the iron removal occurred primarily during the oxidation/precipitation stage, with the filters serving to physically remove the pre-formed solids.
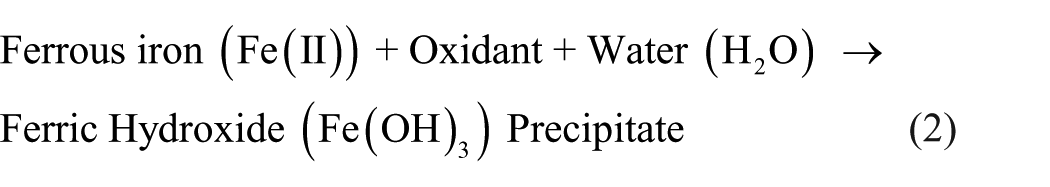

Different treatment scenarios.
Artificial Neural Network
ANN Application
This study created an ANN model to forecast the overall iron removal efficiency of the integrated oxidation-aeration-filtration system. The ANN model was developed using the MATLAB R2023a Neural Network Toolbox. The treatment mechanism encompasses the oxidation, followed by aeration and filtering through Birm and activated carbon media; thus, the ANN reflects the overall system behavior rather than solely the oxidation process. The ANN was chosen for its capacity to capture nonlinear interactions among operational variables that together influence removal efficiency. Therefore, the ANN was applied as a data-driven modeling tool to capture the combined nonlinear interactions among all operational parameters, such as chlorine dose, oxidation time, aeration time, and filtration duration, that collectively influence iron removal efficiency.
ANN Structure
A neural network composed of input, hidden, and output layers was structured to predict the iron removal efficiency. The input layer receives data from six experimental parameters: initial concentration of iron, chlorine dose, oxidation time, aeration time, filtration cycle 1, and filtration cycle 2. As shown in Figure 3, the structure used for the prediction of iron removal efficiency was 6 – 10 – 1, where 10 neurons were included in the hidden layer. The estimation of the actual number of neurons and hidden layers was conducted using trial and error procedure. Three groups were formed from the target and input vectors: training the network utilized 60% of the data, model validation utilized 20% of the data, while the remaining 20% was used for testing the developed network.

ANN of 6 – 10 – 1 structure used for the prediction of iron removal efficiency.
ANN Properties
Feedforward backpropagation algorithm was used in this study for data classification. The target data was compared with the output data using the mean squared error (MSE) (Equation 3), root mean squared error (RMSE) (Equation 4), Chi-square (χ²) (Equation 5), and coefficient of determination (R²) (Equation 6) (Belcaid et al., 2024). By propagating the MSE back from the output layer to the input layer, the values of the weights and biases are adjusted until the maximum number of iterations is reached.

where


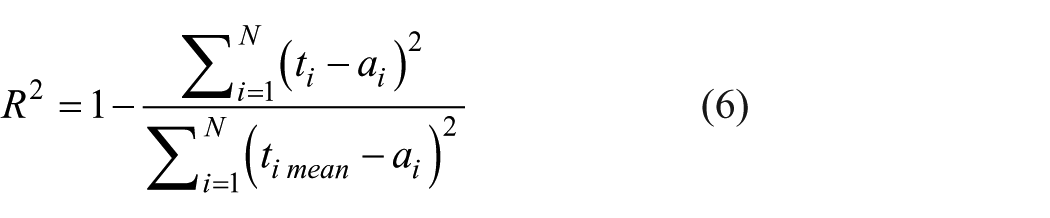
where
The network was trained using Levenberg–Marquartd method (trainlm) method. The “purlin” (Equation 7) transfer function was selected for output layer, which is mainly used for function fitting. The “tansig” (Equation 8) transfer function was selected for hidden layer, which is mainly used for pattern recognition. Applying the “tansig” function means that the neuron’s net input can have any value between positive and negative infinity and the output is limited between −1 and +1.


Results and Discussion
Studied Scenarios
Figure 4(a) shows the iron removal efficiency for scenario 1 at a constant oxidation time of 60 s and filtration time of 60 s. The results showed that the iron removal efficiency has increased with the increase in the chlorine dose. For example, at an initial concentration of 1.5 mg/L, the iron removal efficiency has increased from 50.2% to 65.5% when the chlorine dose has increased from 0.21 mg/L to 0.63 mg/L. This is mainly attributed to the ability of chlorine to convert soluble ferrous iron (Fe²+) into insoluble ferric iron (Fe3+), which allows for its removal through filtration (El Azher et al., 2008). The results also showed that the iron removal efficiency has decreased with an increase in the initial concentration of iron. For example, at a chlorine dose of 0.63 mg/L, the iron removal efficiency decreased from 77.0% to 65.5% when the initial concentration increased from 1.0 mg/L to 1.5 mg/L. This occurs because chlorine interacts with iron to generate iron compounds, and as iron concentrations increase, the reaction may become less efficient, potentially leading to partial oxidation and removal (Qin et al., 2021).

Iron removal efficiency for: (a) scenario 1 at constant oxidation time 60 s and filtration time 60 s, (b) scenario 2 at chlorine dose 0.63 mg/L and filtration time 60 s, and (c) scenarios 3 and 4 at chlorine dose 0.63 mg/L, oxidation time 240 s, and filtration time 60 s.
For the second scenario, the results showed that the iron removal efficiency has increased with the increase in oxidation time, refer to Figure 4(b). For example, at an initial concentration of 1.5 mg/L, the iron removal efficiency has increased from 65.5% to 78.9% when the oxidation time has increased from 60 s to 240 s. Longer oxidation time allows for more complete oxidation and precipitation, leading to higher removal rates (Hoko et al., 2021; W. Li et al., 2018; P. Li et al., 2023; Qin et al., 2021; Shrestha et al., 2024).
For the third scenario, the results showed that the iron removal efficiency has increased with an increase in aeration time, refer to Figure 4(c). For example, at an initial concentration of 1.3 mg/L, the iron removal efficiency has increased from 87.7% to 91.2% when the aeration time has increased from 5 min to 15 min. For full oxidation, adequate aeration time is essential. Some iron may stay dissolved and be challenging to extract if there is insufficient aeration (Coles & Rohail, 2020; Drewnowski et al., 2019; Müller et al., 2024; Roy et al., 2022). The optimal aeration time also depends on pH, water temperature, and the presence of other contaminants (Rajapakse et al., 2022). Excessive aeration time can lead to over-oxidation, reduced filtration rates, and increased chemical usage to remove the fine particles (Bakhtiyari-Ramezani et al., 2025; Dong et al., 2025; Hasan et al., 2025; B. Li & Bishop, 2004).
The fourth scenario has the same operating parameters similar to the third scenario, but a double filtration is applied at the same time 60 min. The results showed a significant improvement in the iron removal efficiency, where at an aeration time of 15 min and an initial iron concentration of 1.5 mg/L, the iron removal efficiency increased from 88.9 to 94.6% when a double filtration is applied. This is because as the filter media builds up a layer of iron oxides on its surface, it becomes more efficient at capturing iron particles (Biswas & Islam, 2022; Desta et al., 2022; Gude et al., 2018; Romanovski et al., 2021). By encouraging more iron removal through adsorption and oxidation, this layer serves as a catalyst (Noubactep, 2008; Sharma et al., 2005).
Comparison With Other Pilot-Scale Systems in the Literature
Previous studies introduced different pilot-scale systems to reduce iron concentration from water, as shown in Table 1. Based on the previous research, the highest iron removal, reaching 95%, can be achieved by applying aeration and filtration using biological roughing up flow filtration. A high iron removal efficiency reaching 92% could be achieved by applying oxidation and filtration using sand, gravel and β MnO2. These results confirm the effectiveness of both the oxidation and aeration processes in the iron removal from water. In our study, the iron removal efficiency exceeded 99% due to applying oxidation, followed by aeration and filtration.
Comparison of Effluent Quality and Performance Between the Pilot-Scale Scenarios From This Study and Other Pilot-Scale Systems.

Cost Estimation
An economic assessment was conducted to evaluate the feasibility of scaling up the proposed pilot system. The total initial capital investment for the pilot plant was approximately $6,500, with annual operational costs of approximately $1,000. Under the optimal treatment conditions, this translates to an estimated iron removal cost of $0.10 per cubic meter of treated water.
A comparison with other pilot-scale iron removal technologies, as shown in Table 2, demonstrates the cost-effectiveness of the proposed system. The cost per m3 in this study is lower than the range reported in previous studies ($0.12–$0.15 per m3), while achieving high removal efficiencies. This favorable cost-benefit ratio underscores the system’s potential for integration into riverbank filtration projects, particularly in cost-sensitive contexts.
A Comparison of the Cost-Effectiveness of This System With Previous Studies.

Artificial Neural Network
Adjusted Weights and Biases
By connecting each component of the input vector (P6 × 1) to each hidden layer neuron, a weight matrix (W10 × 6) is generated. By also adding a 10-length bias (b10 × 1) to the summation of the weighted inputs (ΣW10 × 6.P6 × 1), a net input is generated. The calculated net input (u10 × 1 = ΣW10 × 6.P6 × 1 + b10 × 1) is transferred to output layer using the “tansig” function. By connecting each hidden layer neuron (P10 × 1) to output layer single neuron, a weight matrix (W1 × 10) is generated. By adding a 1-length bias (b1 × 1) to the summation of the weighted inputs (ΣW1 × 10.P10 × 1), a net input is generated. The calculated net input (u1 × 1 = ΣW1 × 10.P10 × 1 + b1 × 1) is transferred to output layer using the “purlin” function. Figure 5 shows the weight matrix and bias of the created ANN model.

A weight matrix and bias of the created ANN model.
Training and Validation Performance
Based on the training results, the number of validation checks was 6 and the gradient magnitude was 3.258. Although the gradient magnitude exceeded the least error level (1e-5), the training phase was stopped due to reaching the maximum number of validation checks at epoch 6, as shown in Figure 6(a). The relation between the mean square error (MSE) performance and the iteration number is shown in Figure 6(b). A gradual decrease was reported for MSE of the training step until reaching the lowest value at epoch number 6. This pattern is normal and does not indicate a training step issue. On the other hand, a gradual increase was reported after epoch number 0 for the MSE of the validation step, which suggests that the model is overfitting the data. Similar behavior was noticed for validation and test curves, where both of them increased with the number of epochs. In summary, the best validation performance was 0.031018 at epoch 0.

The prediction of iron removal efficiency using ANN: (a) Training and (b) best validation performance.
Regression Plot
Figure 7 shows the relationship between network outputs and network targets as represented by the coefficient of determination (

Regression plot between target and output for the prediction of iron removal efficiency using ANN.
Limitations and Recommendations for Future Work
Even though the results of this study show that the proposed pilot-scale system for reducing iron concentration in drinking water is very effective and cost-effective, some limitations should be noted to help future research and large-scale use. The first limitation is that the experiments utilized synthetic water samples prepared in controlled laboratory settings, with iron concentrations ranging from 1.0 to 1.5 mg/L. The water extracted from actual riverbank filtration contains a mixture of dissolved and suspended particles, which can interact with oxidants and filtration media and influence the treatment performance. Additionally, the pH of the solution is a key parameter governing the oxidation kinetics of Fe²+ to Fe3+ and the subsequent hydrolysis and precipitation of Fe(OH)3 Consequently, the results may not entirely reflect the system’s behavior when treating natural water with more variable characteristics. On-site testing at riverbank filtration RBF units with actual groundwater samples is also essential to confirm the system’s performance under realistic conditions. The second limitation is that this study concentrated specifically on a limited range of iron concentrations (1.0–1.5 mg/L). However, in some RBF systems, iron concentration may reach 3 mg/L. Therefore, it is essential to conduct testing throughout a broader concentration range to determine the system’s scalability and reliability. The third limitation is that the water samples were not collected after each filter to evaluate their respective contributions to the overall iron removal efficacy. Consequently, stage-by-stage sampling and analysis should be performed in future research to evaluate each filter’s specific contribution and optimize the configuration.
Summary of Study Limitations and Corresponding Recommendations for Future Work.

Conclusions
This study developed and evaluated a pilot-scale water treatment system combining oxidation, aeration, and multi-stage filtration (sand, activated carbon, and Birm) to remove iron from drinking water. The results demonstrated that the proposed system is technically effective, economically viable, and potentially scalable for integration into riverbank filtration (RBF) applications. Under the optimal operating conditions, a chlorine dose of 0.63 mg/L, oxidation time of 240 s, aeration time of 15 min, and double filtration (60 s each), the system achieved an iron removal efficiency ranging between 94.7% and 99.1%, successfully reducing iron concentrations from 1.5 mg/L to below the WHO permissible limit of 0.3 mg/L. The results also revealed that aeration substantially enhanced oxidation efficiency, while double filtration improved the final removal performance by promoting catalytic adsorption of residual iron on the filter media. Modeling results showed that the created ANN model with a structure of 6-10-1 was reliable in predicting iron removal efficiency across the six tested input parameters, with an R2-value of 0.995, indicating that the model could explain 99.5% of the variation in iron removal efficiency. This confirms that machine learning can serve as an efficient decision-support tool for forecasting system performance and optimizing treatment parameters. The economic analysis indicated a total capital investment of approximately $6,500 and annual operational costs of $1,000, corresponding to a treatment cost of $0.10 per cubic meter of water. This value is lower than those reported for comparable pilot-scale systems, emphasizing the cost-effectiveness and practical potential of the proposed technology for application in resource-constrained regions. Overall, this research provides a comprehensive framework for low-cost, high-efficiency iron removal from drinking water using combined oxidation, aeration, and filtration processes. Future work should focus on validating the system using real RBF water under field conditions, evaluating long-term operational performance, expanding the range of tested parameters, and conducting full life-cycle assessments to confirm economic and environmental sustainability at larger scales.
