Abstract
Keywords
Introduction
Axonal transection appears to be the pathologic correlate of irreversible neurologic impairment in multiple sclerosis (MS), and axonal damage is evident at the earliest stages of disease. 1 –4 For most patients (85%) with clinically definite MS (CDMS), the initial presentation of disease is a clinically isolated syndrome (CIS), that is, an episode of neurological dysfunction followed by complete or partial recovery. 2 The progressive nature of MS and the extent of subclinical damage already detectable at the first demyelinating event suggest that early immunomodulatory treatment may be most effective, by inhibiting the cascade of events leading to irreversible axonal damage. 3,5
PreCISe (Early Glatiramer Acetate Treatment in Delaying Conversion to CDMS in Subjects Presenting with a Clinically Isolated Syndrome) was a randomized, controlled study to evaluate the effects of glatiramer acetate (GA; Copaxone®) on conversion to CDMS in patients with CIS and MRI lesions suggestive of MS. The prospective PreCISe study design included a two-year open-label treatment phase in which all patients received active treatment with GA after the 36-month double-blind, placebo- controlled phase. Based on results of a pre-planned interim analysis during the double-blind phase showing that GA significantly reduced the risk of developing CDMS by 45% and time to conversion was prolonged by 115% versus placebo, 6 the Data Monitoring Committee (DMC) recommended terminating the double-blind phase early and allowing all patients to enter the open-label extension phase. We evaluated the relative effects of early GA treatment (at randomization) compared with effects of delayed GA treatment in patients originally randomized to placebo.
Methods
Study design and patients
The PreCISe trial, initiated in 2004 and completed in June 2010, was conducted at 80 sites in 16 countries. Study design, patient eligibility criteria and conduct of the double-blind phase of PreCISe are reported in detail elsewhere. 6 Briefly, patients aged 18–45 years presenting with a CIS with unifocal manifestation and ≥2 T2-weighted lesions measuring ≥6 mm were eligible if not more than 90 days had elapsed since the clinical attack. Patients were equally randomized to receive subcutaneous (SC) GA 20 mg or placebo daily. Scheduled site visits occurred at screening, baseline, months 1 and 3, and every three months thereafter. Procedures during regular site visits included complete neurological exams (Kurtzke Expanded Disability Status Scale (EDSS), 7 functional systems (FSs), and ambulation index (AI)), MRI scans, vital sign assessments and compliance review.
The criterion to enter the prospectively planned open-label study phase was either a second relapse or the end of the double-blind phase, whichever came first. The onset of CDMS, defined by Poser criteria, 8 involved the occurrence of a second relapse, that is, the appearance of one or more new neurological abnormalities or the reappearance of one or more previously observed neurological abnormalities lasting ≥48 hours and preceded by a stable or improving neurological state for ≥30 days. This criterion differed from the 24-hour duration of exacerbation indicated in the Poser criteria. 8 The second relapse must have occurred in the absence of fever or known infections. Relapse must have been accompanied by objective neurological changes consistent with an increase of ≥0.5 EDSS points or one grade in the score of two or more FSs; or two grades in the score of one FS, compared with the previous disability assessment. Participants could continue to receive open-label GA treatment at the discretion of the investigator/patient for up to a total of 60 months (five years).
Brain MRI scans were performed at screening, baseline, every three months during the double-blind phase prior to the interim analysis, every six months in the open-label phase, and at study termination or early discontinuation post-interim analysis. The same MRI parameters and image assessment methods were employed during the double-blind and the open-label phases, and are reported elsewhere. 6 The Neuroimaging Research Unit in Milan, Italy served as the MRI analysis centre.
Patients provided written informed consent before undergoing any study-related procedures. The protocol and consent documents were approved by institutional review boards and ethics committees of participating centres. The trial is registered with clinicaltrials.gov (NCT00666224).
Statistical analysis
All open-label endpoints were exploratory and were selected to assess potential GA neuroprotective effects by comparing clinical and MRI outcomes in patients assigned to GA treatment at randomization (early-treatment cohort) with those in patients originally randomized to placebo who later received GA (delayed-treatment cohort). Exploratory endpoints were tested at the nominal level α = 0.05. Patients and investigators were kept unaware of the initial treatment allocation throughout the open-label phase of the study.
Time from randomization to conversion to CDMS in the intention-to-treat (ITT) population was estimated using Kaplan–Meier methods, and outcomes in the early-treatment and delayed-treatment groups were compared using a Cox’s proportional hazards model. Proportions of patients who converted to CDMS in the early- and delayed-treatment groups were compared by baseline-adjusted logistic regression analysis. Risk of conversion to CDMS from a Cox’s proportional hazards model was also assessed for early- and delayed-treatment subgroups defined by demographics, characteristics of CIS (gender, age, presenting syndrome, steroid treatment for the initial attack) and MRI findings (disease dissemination and activity), at baseline.
Brain atrophy was defined as the per cent change from baseline to the last observed value (LOV) in brain volume measured according to the Structural Image Evaluation of Normalized Atrophy (SIENA) technique. 9 Adjusted mean per cent changes in brain volume in the early- and delayed-treatment groups were compared by analysis of covariance (ANCOVA), with treatment group, centre, baseline brain volume and study exposure as covariates. Sensitivity analyses to verify atrophy results were performed with a non-parametric approach and a covariate analysis adjusted to MRI machine type used during the study.
MRI endpoints included annualized numbers of new T2-weighted lesions, new GdE (gadolinium-enhanced) lesions, and new T1 hypointense lesions, for which rate ratios (i.e. the ratio of the adjusted means for the early- and delayed-GA-treatment groups) were calculated using a negative binomial model with study exposure as an offset in the model to account for differences in duration of follow-up between cohorts. Additional endpoints included comparison of log-transformed geometric mean volumes of T2-weighted lesions at LOV in the early- and delayed-treatment groups. Repeated measures of changes from baseline to each study visit in MRI endpoints were analysed using the general equation estimation (GEE) method for the negative binomial model, or linear mixed effects model, as appropriate.
MRI lesion detection sensitivity increases with increased frequency of MRI scans. 10 During the double-blind phase of PreCISe (MRI scans every three months), patients in both treatment groups who converted to CDMS were immediately entered into the open-label phase (MRI scans every six months); therefore, they had fewer total MRI scans than patients who did not convert to CDMS during the double-blind phase. Because there was a significant difference between the GA and placebo arms in time to CDMS conversion, 6 patients in the GA arm remained longer in the double-blind phase and had more MRI scans than patients in the placebo arm. Therefore, MRI endpoints were adjusted by the duration of exposure to the double-blind and open-label phases using study exposure as an offset for the negative binomial models or as an additional covariate in the ANCOVA model.
Annualized relapse rates (ARRs), that is, the total number of relapses per patient divided by study exposure per year, were analysed using the Quasi-Likelihood (over-dispersed) Poisson regression model with an ‘offset’ based on the log of study exposure, and country, unifocal presentation and steroid use as covariates in the model.
Disability progression was defined as ≥1.0 EDSS point increase from baseline score if baseline score was between 0 and 5.0, or an increase of ≥0.5 EDSS points if baseline score was ≥5.5, sustained for three months. Repeated measures of change from baseline to each visit in EDSS scores were analysed by baseline-adjusted ANCOVA.
Safety and tolerability assessments included adverse events (AEs), standard laboratory tests, vital signs, physical measurements and electrocardiograph (ECG) tests. Results are reported descriptively.
Results
Of 481 patients originally randomized in the double-blind phase to either GA (

Patient disposition.
Patient characteristics at baseline of the placebo-controlled phase and at entry into the open-label phase.

SD: standard deviation; GA: glatiramer acetate; EDSS: Expanded Disability Status Scale.
Proportionately more patients in the delayed-treatment group discontinued during the open-label phase (40.3%
The median number of days on GA during the open-label phase was 722 days (range: 5–1330) and 721 days (1–1395) for patients in the early- and delayed-treatment groups, respectively. Over both study phases, the median number of days on study drug (first to last dose) was 1709 (range: 92–1871) in the early GA treatment group and 1260 (46–1848) in the delayed GA treatment group, with mean study drug exposures at LOV of 390.6 patient-years and 346.0 patient-years, respectively. A total of 289 patients (60%) completed the study: 163 in the early-treatment group and 126 in the delayed-treatment group (Figure 1). Median post-randomization follow-up was 3.8 years overall (4.4 and 3.35 in the early- and delayed-treatment groups, respectively). For patients who completed the study, the median time from the first CIS symptom to LOV was 1128 days (111–1185) in the early GA treatment group, which was approximately eight months longer (median: 892 days, range: 96–1178) than the time from first CIS symptom for patients in the delayed-treatment group.
A post hoc analysis showed that for all patients (regardless of treatment arm), there was no difference in the primary outcome between those who discontinued the study early and those who remained until the end of the open-label phase. Conversion to CDMS occurred for 37.5% (72/192) of patients who discontinued and 43.6% (126/289) of patients who remained until study end (hazard ratio (HR): 0.91; 95% confidence interval (CI): 0.64–1.28).
Early GA treatment was associated with a 41% reduced risk of CDMS compared with delayed GA treatment (HR: 0.59; 95% CI: 0.44–0.80;
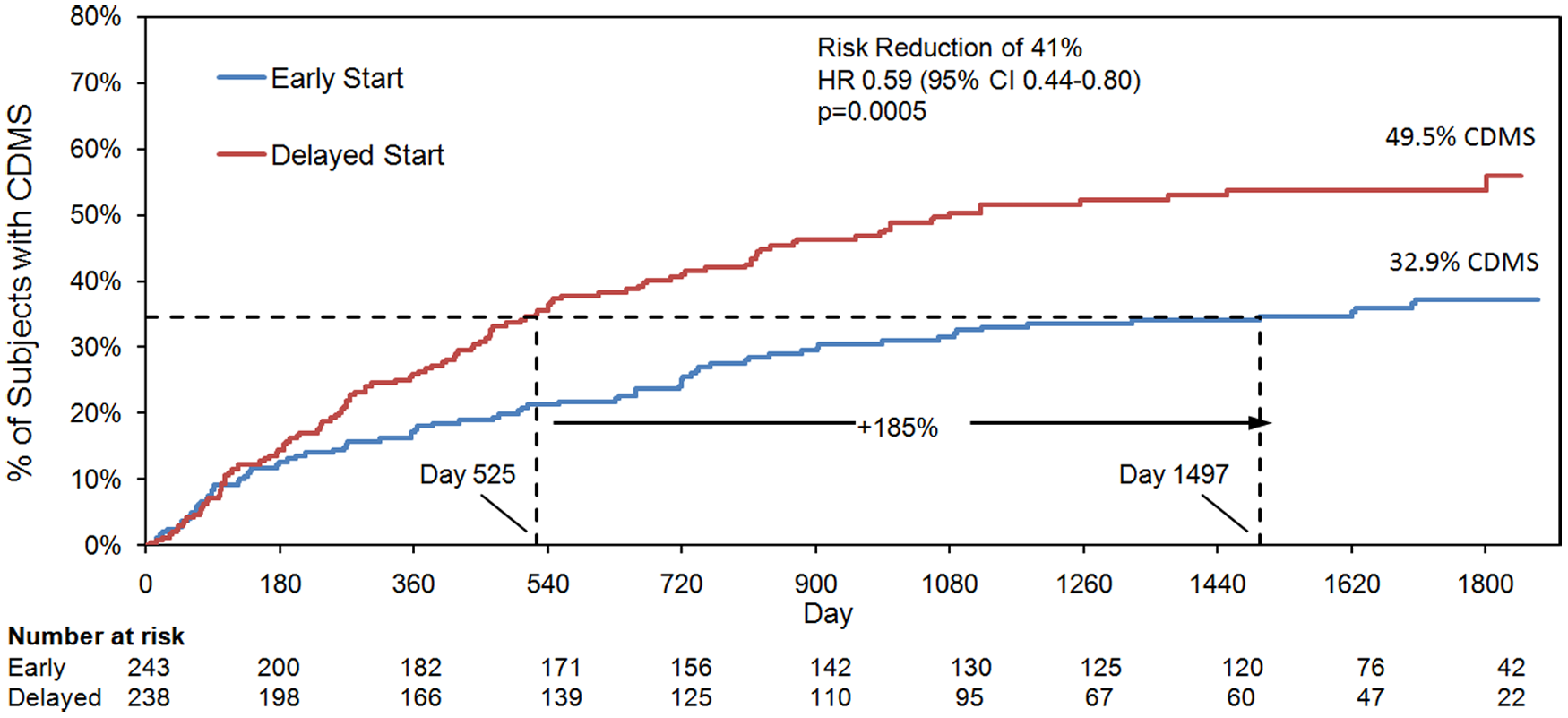
Time to conversion to clinically definite multiple sclerosis (CDMS).
Kaplan -Meier curves were used to estimate the time to conversion to CDMS in the early- and delayed-treatment groups. A delay of 972 days (185%) in conversion to CDMS was observed for patients who received GA in the early-treatment group compared with patients who received delayed GA treatment. A Cox’s proportional hazard model was used to estimate the HR and 95% CIs.
Rates of conversion to CDMS during the open-label phase for patients who had not converted to CDMS during the double-blind phase were low and comparable in the early and delayed GA treatment groups: 12/132 (9.1%) and 7/102 (6.9%), respectively. Of patients who completed the study, 108/163 (66.2%) in the early-treatment group and 55/126 (43.6%) in the delayed-treatment group had not converted to CDMS, a 34% decrease in conversion to CDMS associated with early treatment.
Subgroup analyses of the risk for conversion to CDMS according to baseline demographic and disease characteristics showed that early treatment was associated with significantly lower risk of conversion to CDMS in all subgroups, with the exception of men and patients with lower activity on brain MRI (<9 T2 lesions, no GdE lesions), when compared with delayed GA treatment (Table 2).
Treatment effect of early versus delayed glatiramer acetate treatment on the risk of converting to clinically definite multiple sclerosis (CDMS) in subgroups of patients defined by demographic and disease characteristics at baseline and by study discontinuation status.

Percentage of patients within the early and delayed treatment subgroups who converted to CDMS.
CI: confidence interval: CIS: clinically isolated syndrome; GdE: gadolinium-enhanced.
Per cent brain volume change from baseline to LOV adjusted for study exposure was significantly lower with early GA treatment compared with delayed treatment (−0.99%
MRI and atrophy outcomes in the early and delayed glatiramer acetate (GA) treatment groups.

Means are adjusted based on study exposure.
Rate ratio = ratio of the adjusted means in the early and delayed treatment groups.
SE: standard error; CI: confidence interval; GdE: gadolinium-enhanced; LOV: last observed value

Per cent change of brain volume (ml) from baseline to LOV.
Early GA treatment was also associated with significant reductions in MRI lesion burden compared with delayed treatment, with 42% risk reduction in adjusted mean cumulative number of new T2 lesions per year (Table 4), and 22% reduction in mean T2 lesion volume at LOV (
New T2 lesions, new gadolinium-enhanced (GdE) lesions and per cent brain volume changes each year by treatment group.

Means and rate ratios are adjusted for study exposure and estimated from a negative binomial regression model.
Reported means and standard deviations for per cent brain volume changes are descriptive and are not adjusted for study exposure.
LOV: last observation; SE: standard error; SD: standard deviation.
As expected in such early patients, ARRs were low for both treatment groups in each study year; nevertheless, ARRs were significantly lower with early GA treatment each year and over the entire study duration (Figure 4). Over the five-year study, 66.7% of patients receiving early GA treatment and 50.4% receiving delayed GA treatment were relapse-free.

Annualized relapse rates (ARRs).
There was little change in the mean EDSS scores from baseline to LOV in the open-label phase for either group (early-treatment, 0.12 (standard deviation, SD 1.04) and delayed-treatment 0.06 (SD 0.90)), and no significant differences between treatment groups in the proportion of patients with confirmed disability: 20.5%
GA was well tolerated, with only 71 patient withdrawals (14.8%) over five years due to AEs. AE type, frequency, and severity were consistent with the known safety profile of GA. No significant differences were detected in the incidence of any AE between the early- and delayed-treatment groups. The most common treatment-associated AEs were injection site reactions. Serious AEs were reported in 28 patients in the early-treatment group (including one death during the double-blind phase 6 ) and 32 patients in the delayed-treatment group. No differences between the early- and delayed-treatment groups were observed in laboratory assessments, vital signs or ECG measures.
Discussion
Results of the open-label phase of the PreCISe study confirm the sustained benefit of GA reported in the double-blind phase of the clinical trial, 6 which showed that GA therapy reduces the risk of developing CDMS and prolongs the time to CDMS conversion. These are the first data to show that earlier treatment with GA confers significant clinical benefits compared with delayed treatment in patients with CIS, by lowering the risk of conversion to CDMS (41%), decreasing disease burden on MRI and reducing progression of brain atrophy.
The time to a second relapse for untreated patients with CIS is one of the few clinical factors strongly associated with longer-term disability; shorter intervals are associated with worse prognosis. 12 –14 Whether this remains true for patients receiving disease-modifying therapy is unknown. In the double-blind phase of PreCISe, time between the first and second attack in patients who converted to CDMS was prolonged 115% with GA compared with placebo. 6 By study end, early treatment with GA prolonged the time to a second attack by 185% compared with delayed treatment, strongly supporting the early initiation of GA treatment.
Comparisons across clinical trials must be made cautiously because of differences in patient populations and study parameters; nevertheless, it is interesting that the long-term effect of GA in preventing conversion to CDMS is very similar to findings of other long-term studies exploring the effects of the IFNβ drugs in CIS patients. 15 –17 In contrast to those studies, however, early GA treatment conferred significant benefits in reducing T2 lesions volume and loss of brain volume compared with delayed treatment.
This is the first study to demonstrate a significant benefit of early immunomodulating therapy on reducing the extent of brain atrophy in patients with CIS compared with delayed treatment.
15,17
Because brain atrophy is considered a reliable measure of irreversible tissue loss, the clear benefit on this measure is a strong argument in favour of early treatment. The positive effects on brain atrophy are consistent with the observed reduction in annualized number of new T1 hypointense lesions in the early-treatment group versus the delayed-treatment group. Early treatment with GA was also more beneficial in patients with RRMS in the pivotal European/Canadian multicentre randomized MRI study.
11
Long-term (mean 5.8 years) follow-up of patients in the European/Canadian study showed that patients who received early treatment with GA were significantly (
Despite significant improvements with early versus delayed GA treatment on multiple MRI measures, including brain atrophy, no significant difference between the GA early- and delayed-treatment groups was observed in the rate of confirmed progression of disability, which was reported for only 21% of patients in each group over the five-year study. This may be due in part to the low rate of progression during early MS, but may also reflect the study design, in which patients in the delayed-treatment group received GA immediately upon conversion to CDMS. No significant differences between early and delayed treatment effects on disease progression have been detected in other long-term CIS studies. 15,17
GA was well tolerated, with roughly 15% of patient withdrawals due to AEs over five years. AEs reported during the open-label treatment phase were consistent with reported events during the double-blind phase, 6 and are in line with the well-established long-term safety of GA in studies with patients with RRMS. 20 There was a higher rate of termination due to AEs in the delayed-treatment group during the open-label phase of the study, probably because these patients were exposed for the first time to known AEs associated with GA (e.g. injection-site reaction), which was not the case for the early-treatment group.
Results of the current study indicate that starting GA after the first clinical symptoms suggestive of MS may have long-lasting benefit, including protection from irreversible brain tissue loss. These findings, together with the well-established safety and tolerability profile of GA, should be considered when making decisions regarding early treatment in patients with CIS.
