Abstract
Keywords
Introduction
Hepatitis B virus (HBV) infection is a major cause of cirrhosis and the most common etiology of hepatocellular carcinoma worldwide with over 290 million people chronically infected with HBV and over 800,000 deaths annually [1,2]. While an HBV vaccine is effective in preventing infection [3,4], millions of people remain unvaccinated with 1.5 million new infections per year. Although approved treatment options for chronic hepatitis B (CHB) infection, including pegylated interferon (pegIFN) and nucleos(t)ide (NUC) reverse transcriptase inhibitors (NRTIs) are available, only a limited number of patients with CHB infection achieve functional cure, or a sustained loss of hepatitis B surface antigen (HBsAg) with undetectable HBV DNA 6 months after completing therapy [5,6] with currently available treatments. Elimination of HBsAg is achieved with pegIFN treatment in only 11% of patients after 3 years [3] and in ≤10% with NRTIs after 5 years [7,8]. And although certain patients can stop NUC-based therapy (e.g., non-cirrhotic, HBeAg-negative patients who have had at least 3 years of antiviral treatment, undetectable viral load and no signs of advanced fibrosis), most patients require lifelong maintenance therapy for HBV infection, which is associated with a substantial economic burden [9,10], a risk of breakthrough drug resistance, and potential drug toxicity. Further, although the risk of hepatocellular carcinoma (HCC) is significantly decreased in CHB treated with NUC-based therapy, HCC may occur despite an optimal antiviral response (Sarin et al., 2015; Terrault et al., 2018; EASL, 2017) [3,4,11]. Thus, there is a need for treatment options for patients with CHB infection that provide a sustained clinical response and functional cure.
Novel drugs in early stage development may offer a potential for a functional cure, including viral entry inhibitors, epigenetic control of cccDNA, immune modulators, RNA interference techniques, ribonuclease H inhibitors, and core inhibitors (also categorized as capsid assembly modulators or core protein allosteric modulators) [5,12]. Results from early Phase 1 studies with 28-day treatment with core inhibitors in patients with HBV infection report approximately 2- to 3-fold log10 IU/mL decreases in mean HBV DNA concentration in viremic CHB patients [13–15].
EDP-514 is a novel HBV core inhibitor, which blocks HBV replication by stimulating premature core assembly and preventing proper encapsidation of viral pregenomic (pg) RNA [16]. As a class II core inhibitor, EDP-514 stimulates the assembly of core dimers into empty capsid-like structures but does not result in the aggregation and degradation of core protein. In stable cell lines expressing HBV, EDP-514 potently inhibited encapsidation of viral RNA and production of viral DNA. In cell lines or primary human hepatocytes where the expression of HBeAg or HBsAg depends on cccDNA, EDP-514 reduced the level of HBeAg and HBsAg indicating that it had prevented cccDNA formation. In cell culture systems, EDP-514 was active against all eight HBV genotypes (A through H) tested, showed no significant cytotoxicity, and inhibited replication of HBV reverse transcriptase resistant mutants. A promising early nonclinical safety and pharmacological profile support EDP-514 as a viable candidate for clinical evaluation in patients with HBV.
In this first-in-human study, the safety, tolerability, and pharmacokinetics (PK) of EDP-514 were assessed after single and multiple ascending doses in healthy subjects during fasted and fed states, and the antiviral activity of EDP-514 was evaluated in NUC-suppressed patients with CHB infection.
Methods
The study was conducted in compliance with the International Conference on Harmonisation Good Clinical Practices guidelines, the Declaration of Helsinki, and national regulations for clinical trials. The study protocol and informed consent were reviewed and approved by Institutional Review Boards/ethics committees for respective participating centers. Written informed consent was obtained from all participants prior to any study procedures.
Study design
This was a 2-part randomized, double-blind, placebo-controlled study that assessed the safety, tolerability, PK and antiviral activity of EDP-514 (Figure 1). Part 1 was a single ascending dose (SAD) and multiple ascending dose (MAD) study conducted in healthy subjects. Part 2 was a MAD study in HBV DNA-suppressed CHB patients on NUC therapy (i.e., NUC-suppressed) who were either hepatitis B e antigen (HBeAg)-positive or HBeAg-negative. Registered at clinicalTrials.gov: NCT04008004. Pharmacokinetic profiles of SAD and MAD steady state in healthy subjects, and NUC-suppressed CHB patients. (A) Mean EDP-514 plasma concentrations over time for the SAD phase for all dose groups on a semi-logarithmic scale, (B) Mean EDP-514 plasma concentrations over time on Day 14 for the MAD phase for all dose groups on a semi-logarithmic scale, (C) Mean EDP-514 plasma concentrations over time in NUC-suppressed CHB patients on a linear scale. SAD, single ascending dose; MAD, multiple ascending dose; CHB, Chronic hepatitis B.
In the Part 1 SAD phase, 8 subjects per cohort were randomized 3:1 to a single oral 50, 100, 200, 400, 600 or 800 mg dose of EDP-514 or placebo in a fasted state. The effect of food on the PK profile was evaluated in the 400 mg cohort with 10 subjects randomized 4:1 to receive EDP-514 or placebo. After subjects completed the fasted part of the 400 mg cohort and a minimum 7-day washout period, they received a second 400 mg dose of EDP-514 or placebo in a fed state. For the MAD phase, dosing in the EDP-514 200 mg group was initiated following a review of safety and PK data from the 400 mg SAD fasted and fed cohorts. Subsequently, subjects were randomized to 200, 400 (fed and fasted cohorts) or 800 mg doses of EDP-514 or placebo once daily for 14 days. In Part 2, 8 NUC-suppressed CHB patients per cohort were randomized 3:1 to 200, 400 or 800 mg doses of EDP-514 or placebo once daily for 28 days. Throughout the study, all decisions on progressing to the next dose cohort were based on recommendations from the Safety Review Committee.
In Part 1, subjects fasted for at least 8 h prior to each dose of study drug and until 4 h postdose. Subjects in the fed cohorts received a standard high-fat meal in the SAD, and standard meal in the MAD, and study drug was administered within 10 min of completing the meal. In Part 2, CHB patients received EDP-514 with no dietary or fluid restrictions.
Subject population
Healthy subjects ages 18–65 years were eligible based on physical examination, medical history, and clinical laboratory testing, and if they were willing to use acceptable forms of contraception. Subjects were excluded for any clinically relevant medical or psychosocial condition that could interfere with study participation. Subjects were excluded for the use of any prescription medication or receipt of any vaccine or investigations drug within 28 days or 5 half-lives prior to the first dose of study drug, or use of nonprescription drugs, dietary or herbal supplements, hormone replacement therapy or cytochrome P450 3A4 or p-glycoprotein inducers or inhibitors within 14 days of study drug.
NUC-suppressed chronic hepatitis B patients were ages 18–70 years with a BMI of 18–35 kg/m2. Patients were required to have HBsAg detectable in serum/plasma at screening and in the most recent HBsAg serum/plasma testing at least 6 months previously; HBV DNA levels in serum/plasma less than the lower limit of quantitation (<LLOQ) at screening and no HBV DNA serum/plasma test values ≥LLOQ over the previous 12 months; and must have been prescribed HBV NUC treatment with no change in regimen for 12 months prior to screening.
Chronic hepatitis B patients were excluded for a prior diagnosis of cirrhosis or documented extensive bridging fibrosis or cirrhosis (i.e., Metavir ≥3 or Ishak fibrosis score ≥4 by a prior liver biopsy, or FibroSure at Screening with a score of ≥0.48 and AST to platelet ratio index ≥0.45, or FibroScan with a result ≥9 kPa at Screening or within 6 months of screening), or prior history of hepatocellular carcinoma or screening alpha-fetoprotein ≥50 ng/mL without imaging or evidence of lack of hepatocellular carcinoma by imaging in the past 3 months, or alpha-fetoprotein <50 ng/mL at screening without imaging. At screening, patients were excluded if alanine aminotransferase (ALT) values were >2 X upper limit of normal (ULN), direct bilirubin >1.2 X ULN, or international normalized ratio [INR] >ULN. Patients also were excluded for coinfection with human immunodeficiency virus (HIV), hepatitis C virus (HCV), hepatitis D virus (HDV).
Full eligibility criteria are provided in the supplementary information.
Study assessments
Safety was assessed by physical examination, vital signs (heart rate, blood pressure, respiratory rate, body temperature), clinical laboratory testing (chemistry, hematology, urinalysis), 12-lead electrocardiogram (ECG), and reports of adverse events (AEs). Partial thromboplastin time (PTT), prothrombin time (PT), and INR were measured at screening, Day 1, and end of study in healthy subjects, and at each study visit in CHB patients.
In Part 2, patients were assessed at screening for the presence of HAV, HDV, HCV, HEV, and HIV and for HBV DNA, hepatitis B core-related antigen (HBcrAg), hepatitis B e-antigen (HBeAg), hepatitis B surface antigen (HBsAg), and HBV RNA at each study visit.
In Part 1, plasma samples were collected for PK analysis of EDP-514 and metabolites (EP-027513, EP-029109, EP-029564) at 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8, 10, 12, 15, 24, 30, 36, 48, 60, 72, 96, and 120 h for the SAD phase and for the MAD phase at Day 1: 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8, 10, 12, 15 h; Days 2 (24 h), and predose Days 3 through 13; Day 14: 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8, 10, 12, 15, 24 (Day 15), 48 (Day 16), 72 (Day 17), and 96 (Day 18) hours postdose. In the SAD phase only, urine samples were collected 0 (predose) and at 0–6; 6–12; 12–24; 24–48; 48–72; and 72–120 h intervals.
For Part 2, plasma samples were collected for PK analysis of EDP-514 and metabolites on Day 1: 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8 h and on Day 28: 0 (predose), 0.5, 1, 2, 3, 4, 5, 6, 8 h. At all other visits, PK samples were collected predose and at 1–3 h postdose and at least 1 h later but prior to administration of the next dose.
Statistical analysis
Approximately 82 healthy subjects were to be evaluated in Part 1 and at least 24 NUC-suppressed chronic hepatitis B patients were to be evaluated in Part 2, which was considered sufficient to characterize the safety, tolerability, and PK for each EDP-514 dose level in healthy subjects and to provide preliminary information on antiviral activity, safety, and PK for each EDP-514 dose level in CHB patients.
For safety data, no formal statistical analyses were performed. PK parameters for each dose level were calculated from the concentrations of EDP-514 and its metabolites (EP-027513, EP-029109, EP-029564) measured in predose and postdose plasma samples. For each EDP-514 dose level, descriptive statistics (sample size, arithmetic means, geometric means, standard deviation, % coefficient of variation [CV], % geometric CV, minimum, median, and maximum) were reported. Dose proportionality was assessed using linear regression analysis. To assess the effect of food on the PK of EDP-514, log-transformed PK parameters (area under the concentration curve [AUC] for 0-infinity, 0 to time [AUC0-inf, AUC0-t] and peak plasma concentration [Cmax]) were determined, and point estimates and their associated 90% confidence intervals (CIs) for the ratios of PK parameters under fed relative to fasted conditions were calculated. For the MAD cohort, Cmax, trough plasma concentration (Ctrough), and AUC0-last were calculated for plasma EDP-514 and its metabolites as applicable.
For each EDP-514 dose cohort in Part 2, HBV-specific biomarkers were evaluated at baseline, on treatment through Day 28, and at 4 weeks posttreatment. The secondary antiviral endpoint was HBV DNA levels over time in NUC-suppressed CHB patients. HBV DNA levels were summarized by treatment using descriptive statics. The percentage of patients with virologic failure defined as a serum/plasma HBV DNA level ≥LLOQ, which was confirmed to be ≥LLOQ on repeat testing was determined. Other antiviral parameters assessed over time included HBsAg, HBeAg, HBcrAg, and HBV RNA levels.
The safety population included all subjects who received at least one dose of study drug. The PK population was all subjects who received active study drug and had any measurable plasma concentration at any timepoint. The antiviral population was all patients who received at least one dose of study drug and had any on treatment HBV DNA data.
Results
Disposition and baseline characteristics
In Part 1, 50 subjects were randomized in the SAD phase and 32 subjects in the MAD phase (Supplementary Figure 1). One (16.7%) subject in the SAD 100 mg group withdrew consent. One subject (16.7%) in the MAD 800 mg group withdrew consent. Thus, 49 subjects in the SAD phase, and 31 subjects in the MAD phase completed the study.
In the SAD phase, subjects were mostly male (38 [76%]), of Black/African American (22 [44.0%]) or white (23 [46%]) race, with a mean age of 42.1 years and a mean BMI of 26.1 kg/m2 across all cohorts (Supplementary Table 1). In the MAD phase, subjects were mostly male (21 [65.6%]), white (17 [53.3%]) or Black/African American (13 [40.6%]), with a mean age of 41.2 years and a BMI of 25.7 kg/m2 (Supplementary Table 1).
Incidence of treatment-emergent adverse events occurring in healthy subjects.

TEAE, treatment-emergent adverse events.
Safety and tolerability
In Part 1 SAD and MAD phases, EDP-514 was well tolerated with a favorable safety profile in healthy subjects. Seven (14.0%) subjects in the SAD phase and 7 (21.9%) in the MAD phase reported at least one treatment-emergent adverse event (TEAE) and no dose-related trends were observed (Table 1). In the SAD phase, headache was the most frequently TEAE in four (8.0%) subjects and was considered possibly related to study drug in one (12.5%) subject in the 400 mg fed group and one (16.7%) in the 600 mg fasted group. Dizziness and blurred vision in one (16.7%) subject in the 200 mg fasted group were considered possibly related to study drug. All TEAE were of mild severity, and all events resolved by the end of the study. No discontinuations for an AE, deaths or serious AEs were reported. In the MAD phase, the most frequently reported TEAEs were headache in three (9.4%) subjects and nausea in two (6.3%) subjects. Two events of headache and one of nausea were considered related or possibly related to study drug. In both SAD and MAD phases, no clinically relevant changes from baseline were observed for clinical laboratory tests, vital signs or ECG findings.
Incidence of treatment-emergent adverse events in NUC-suppressed chronic hepatitis B patients.

TEAE, treatment-emergent adverse events.
Pharmacokinetics and antiviral activity
Pharmacokinetic parameters for single ascending dose, multiple ascending dose healthy subject cohorts, and multiple-ascending dose NUC-suppressed chronic hepatitis B cohorts.

Values are presented as geometric mean (% coefficient of variation), except for Tmax, which is the median (range).
Abbreviations: AI, accumulation index; AUC0-inf, area under the plasma concentration-time curve from time 0 extrapolated to infinity; AUC0-last, area under the plasma concentration-time curve from time 0 to the last measurable non-zero concentration; AUC0-tau, area under the curve to the end of the dosing period; Cmax, maximum observed plasma concentration; C24, plasma concentration at 24 h postdose; Ctrough, plasma concentration at the end of the dosing interval; CL/F, apparent total clearance of the drug from plasma after oral administration; CLss/F, apparent total clearance at steady state; Lz, apparent terminal elimination rate constant representing the fraction eliminated per unit time; t1/2, apparent terminal elimination half-life of medication, calculated as 0.693/λz; Tmax, time to reach Cmax; Vd/F, apparent volume of distribution after oral administration; Vdss/F, apparent volume of distribution at steady state.
a
For EDP-514, geometric mean cumulative amount excreted unchanged (Ae0-120) of EDP-514 in urine increased in a dose-related manner and ranged from 484,000 to 5,780,000 ng. Geometric mean cumulative fraction of dose excreted (fe0-120) unchanged in urine ranged from 0.56% to 1.21% over 120 h across all doses. Geometric mean renal clearance (CLR) ranged from 48.1 to 118 mL/h and was independent of dose across the majority of the dose range and was not impacted by food.
EDP-514 exhibited an increase in metabolite (EP-027513, EP-029109, EP-029564) exposures with dose (data not shown). For EP-027513, EP-029109, and EP-029564, the metabolic ratio for AUClast ranged from 0.06 to 0.09, 0.04 to 0.06, and 0.03 to 0.05, respectively, across the dose range of 50–800 mg, and AUC was not impacted by food at the 400 mg dose.
For the metabolites, the median Tmax was generally comparable across fasted doses, and mean t1/2 appeared to be independent of dose and food.
In healthy subjects, following 14 days of dosing over a dosage range of 200–800 mg once daily, steady-state geometric mean Cmax, AUClast, and AUCtau of EDP-514 increased 1.8-, 1.7-, and 1.7-fold, respectively (Table 3 and Figure 1B). Geometric mean plasma concentration over 24 h (C24) for EDP-514 on Day 14 ranged from 414 to 663 ng/mL (Table 3), and steady state appeared to be reached by Day 3 (data not shown). At steady state, median EDP-514 Tmax ranged from 2.5 to 4.0 h postdose and geometric mean t1/2 ranged from 15.7 to 20.4 h and were independent of dose and food.
Following administration of 400 mg EDP-514 with a standard content meal, EDP-514 PK exposures showed a mild increase (1.5- to 1.7-fold) compared to the fasted state, which was similar for Day 1 and Day 14. Steady-state median Tmax was not impacted by food. Under fed conditions at steady state, Cmax was 46% and AUCtau was 72% higher, respectively, compared to fasted conditions. Less than 2-fold accumulation of EDP-514 was observed, with the geometric mean accumulation index ranging from 1.39 to 1.58 following multiple dosing.
Multiple ascending doses of EDP-514 in healthy subjects generally showed an increase in metabolite (EP-027513, EP-029109, EP-029564) exposures with dose (data not shown). For EP-027513, EP-029109, and EP-029564, the metabolic ratio for AUCtau on Day 14 ranged from 0.09 to 0.15, 0.07 to 0.09, and 0.0 to 0.06, respectively, for doses of 200–800 mg, and the administration with food had less of an effect compared to the parent drug EDP-514. Median Tmax was generally comparable across doses and geometric mean t1/2 appeared to be independent of dose and food.
In NUC-suppressed CHB patients, EDP-514 exposure (AUC and Cmax) increased linearly with dose on both Day 1 and Day 28, with some accumulation for Day 28 versus Day 1 (Table 3 and Figure 1C). The PK profile supported once daily dosing with median Ctrough concentrations at Day 28 that were approximately 16-fold for 200 mg, 18-fold for 400 mg, and 21-fold for 800 mg, above the protein-adjusted EC50.
For subjects with quantifiable HBV RNA at baseline (Supplementary Table 1), mean change from baseline to Day 28 for HBV RNA was −2.03, −1.67, −1.87, and −0.58 log U/mL in the 200 mg ( HBV RNA reduction in EDP-514 versus placebo groups in NUC-suppressed CHB patients. Mean (SD) change from baseline versus time for HBV RNA among those with quantifiable levels at baseline. HBV, hepatitis B virus; CHB, Chronic hepatitis B.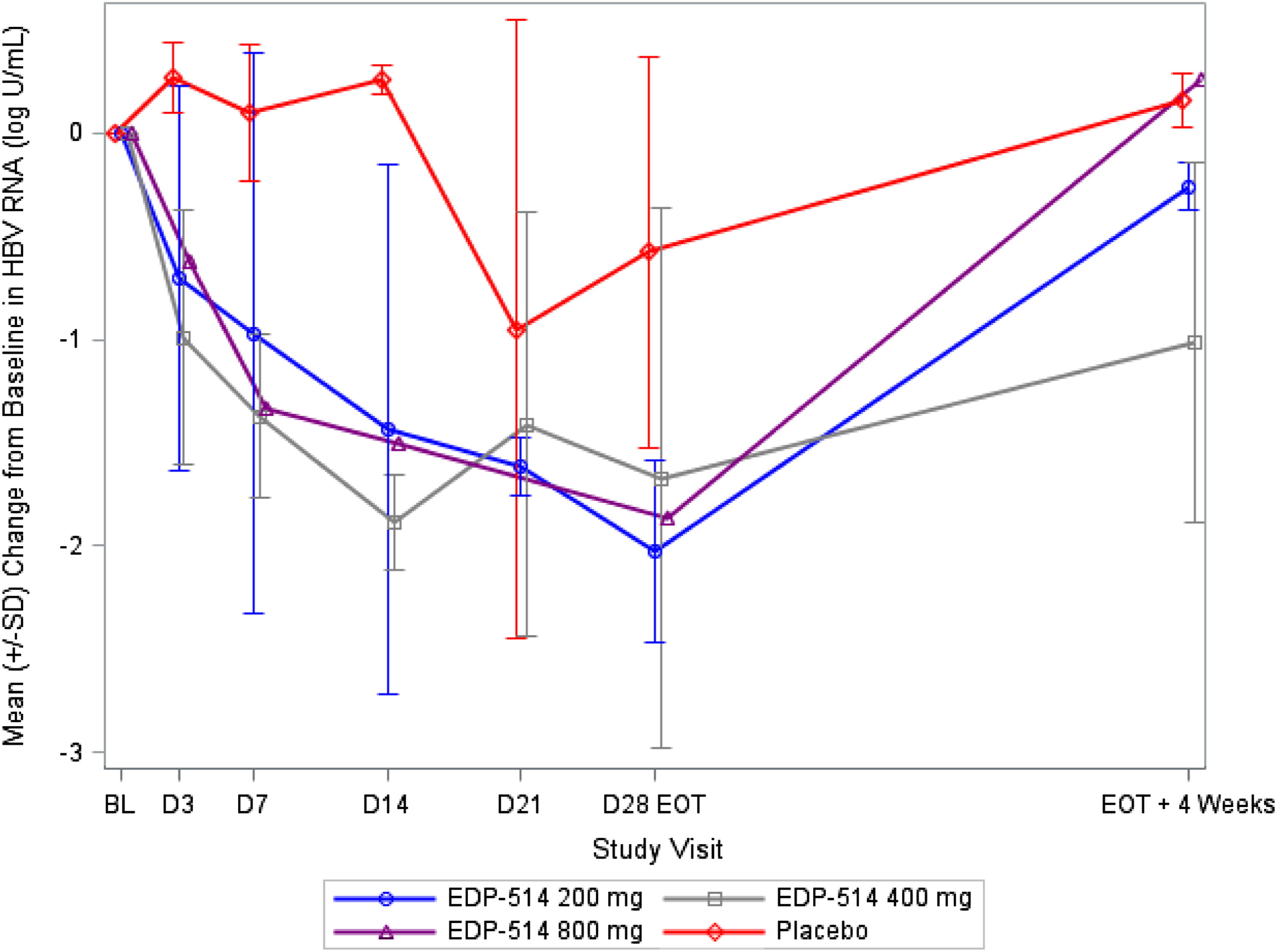
Discussion
The HBV core protein is a multifunction protein that plays a pivotal role in the HBV life cycle and is a target for novel direct-acting HBV antivirals. The HBV core protein is responsible for the assembly of capsids and is directly involved in the recruitment and packaging of viral pregenomic RNA [17]. The HBV core protein also possesses limited sequence polymorphism across HBV genotypes. Moreover, HBV core protein-mediated functions are fundamental to HBV persistence since they directly impact the intracellular replenishment of nuclear pools of HBV cccDNA [18]. Consequently, core inhibitors, molecules targeting the HBV core protein, are being developed as promising direct-acting antivirals.
In the present study, a mean HBV RNA change of −2.03, −1.67, −1.87, and −0.58 log U/mL was observed in the EDP-514 200 mg, 400 mg, 800 mg, and placebo groups, respectively, which is similar to results for other HBV core inhibitors [14,15,19]. Although some studies monitoring serum HBV RNA have suggested that this marker may be a predictor of the treatment response and cccDNA activity [20–24], more data will be needed to confirm the clinical significance and to better understand the potential biological function of extracellular HBV RNA. The decrease in HBV RNA observed with EDP-514 and other HBV core inhibitors underscores the potential of this class as a direct-acting antiviral.
EDP-514 was generally well tolerated with mostly mild AEs and no clinically relevant laboratory abnormalities or changes in vital signs or ECG. The most common AE was headache, and few subjects had events that were considered related to EDP-514. No discontinuations, serious AEs or deaths were reported in healthy subjects, and only one HBV patient discontinued for an AE that was unrelated to study drug.
After single or multiple oral doses in healthy subjects and NUC-suppressed CHB patients, respectively, EDP-514 was rapidly absorbed, and exposure increased with increasing doses. There was some degree of accumulation of EDP-514 up to Day 14 in healthy subjects and up to Day 28 in CHB patients, as exposures were higher than after the first dose. The PK profile of EDP-514 in CHB patients was consistent with results in healthy subjects and supports once daily dosing, with median Ctrough concentrations at Day 28 that were approximately 16-fold for 200 mg, 18-fold for 400 mg, and 21-fold for 800 mg, above the protein-adjusted EC50.
Loss of HBsAg is only achieved in approximately 2.5%–10% of patients after 1 year of treatment with pegIFN and NRTI. Although add-on or switching therapies may achieve HBsAg seroclearance, the probability of HBsAg loss remains unpredictable in both HBeAg-positive and -negative patients [5,15,25–27]. To achieve functional cure, multiple targets may be required. Assembly of the HBV core protein is considered as an important step to facilitate pgRNA encapsidation and viral replication [26], and represents a promising antiviral target for the treatment of CHB patients. The addition of a core inhibitor to a combination of NRTI and small interfering RNAs did not appear to improve the kinetics of HBsAg decline over time or the percentage of patients achieving HBsAg <100 IU/mL at Week 48 [28]. The combination of a core inhibitor and NRTI led to greater viral suppression than NRTI alone including treatment naïve HBeAg-positive patients and virologically suppressed HBeAg-negative patients [27] using a sensitive HBV nucleic acid assay [24]. As it is yet unknown whether the lack of effect of this combination treatment on HBsAg decline is compound-specific or class-specific, further evaluation may be warranted, in addition to investigation of combinations of direct-acting antivirals with immune modulators, which may provide potential treatment options for CHB patients [29–31].
Limitations to this study include the short treatment duration, which only allows for short-term evaluation of safety and antiviral activity. Also, the small number of study participants, particularly the lack of HBeAg-positive NUC-suppressed CHB patients, limited analyses based on demographics and baseline characteristics. In addition, the healthy subject population was predominantly Caucasian while the CHB patients were all Asian. The small sample size limited the ability to describe a dose response effect on HBV RNA kinetics.
Overall, these results demonstrate that EDP-514, a novel oral HBV core inhibitor, was generally safe and well tolerated in healthy subjects and CHB patients. The PK profile of EDP-514 supports once daily dosing with an increase in exposure with increasing doses. In NUC-suppressed CHB patients, EDP-514 resulted in a mean reduction in HBV RNA of up to 2 log after 28 days consistent with its mechanism of action as an HBV core inhibitor. These results support further investigation of EDP-514 in combination treatment for NUC-suppressed and treatment naïve patients with CHB.
Supplemental Material
Supplemental Material - EDP-514 in healthy subjects and nucleos(t)ide reverse transcriptase inhibitor-suppressed patients with chronic hepatitis B
Supplementary Material for EDP-514 in healthy subjects and nucleos(t)ide reverse transcriptase inhibitor-suppressed patients with chronic hepatitis B by Jordan J Feld, Eric Lawitz, Tuan Nguyen, Jacob Lalezari, Tarek Hassanein, Paul Martin, Steven-Huy Han, Douglas Dieterich, Jeanne-Marie Giard, Guy De La Rosa, Alaa Ahmad, Ed Luo, Annie L Conery, and Nathalie Adda in Antiviral Therapy.
Footnotes
Acknowledgments
Author contributions
Declaration of conflicting interests
Funding
Data availability
Trial registration
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
