Abstract
Introduction
Seasonal influenza A viruses (IAVs) are transmissible pathogens responsible for acute respiratory infections that can lead to up to 650,000 deaths per year worldwide. 1 Young children, the elderly and people with chronic illnesses are at a higher risk of developing severe complications. 2 During severe IAV infection, the imbalance between pro-inflammatory and anti-inflammatory cytokines is associated with a deleterious clinical outcome. As a complement to annual immunization programs, antivirals targeting key viral enzymes, such as the neuraminidase (NA) and the polymerase acidic (PA) endonuclease, can play an important role in controlling seasonal influenza infections. 3 However, antivirals alone have not been shown to provide a significant reduction in mortality rates for severe influenza. 4 As the exacerbation of the inflammatory response could be influenced by host-related factors in addition to factors related to viral replication, the use of compounds with the potential to modulate the host immune response could provide important clinical benefits.
Previous reports showed that protectins D1 (PD1) and DX (PDX), pro-resolving mediators that are formed during the resolution phase of acute inflammation, contributed to enhance the return of inflamed/injured tissue to homoeostasis. 5 These protectins were involved in the defence of the host against various viral infections, including those caused by herpes viruses, 6 human respiratory syncytial virus 7 and virulent human and avian influenza A (H1N1) and A (H5N1) strains.8,9
In a previous study, we synthesized a series of structurally simplified PDX analogues
10
and assessed their activity against influenza A (H1N1) viruses,
Materials and methods
Cell culture and virus stock preparation
Madin-Darby canine kidney cells overexpressing the ⍺2-6 sialic acid receptor (ST6-GalI-MDCK cells), kindly provided by Y. Kawaoka from the University of Wisconsin-Madison, WI), 12 were maintained in minimum essential medium (MEM) supplemented with 10% FBS and puromycin (7.5 µg/mL).
Influenza A/Puerto Rico/8/1934 (H1N1) virus stock was prepared by infecting ST6GalI-MDCK cells in MEM medium supplemented with 1 μg/mL tosyl phenylalanyl chloromethyl ketone (TPCK)-treated trypsin (Sigma, Oakville, ON, Canada). After 4 days of incubation, resulting into complete cytopathic effects, viral production was harvested, aliquoted and frozen at – 80°C. An aliquot was thawed and titrated by plaque assay using 12-well plates containing confluent ST6GalI-MDCK cells.
Compounds
Oseltamivir-phosphate (Tamiflu) was purchased from a local pharmacy (at the CHU de Québec, Québec, Qc, Canada), and the protectin DX analogue AN-137B was synthesized as previously described 10 and stored at −80°C under nitrogen and protected from light. No detectable degradation was observed after 1 year of storage under these conditions, as confirmed by HPLC and 1 H proton nuclear magnetic resonance (1H NMR) analyses. AN-137 B was resuspended in propylene glycol: dimethylsulfoxide (DMSO) (92: 8) immediately before injection to ensure compound stability and accurate dosing.
Mouse experimental infections
Animal studies were performed using groups of nine 6- to 8-week old female C57BL6 mice (Charles River, St-Constant, Canada) that included a group of untreated animals and three treatment groups consisting of oral oseltamivir and/or intraperitoneal AN-137B (1 mg/kg, once daily for 5 days). The dose of AN-137B and the route of administration were selected from preliminary experiments assessing the compound toxicity. In addition, given the fact that native protectin (PDX) and other analogues have demonstrated efficacy in several inflammatory and infectious models, we anticipated a measurable biological response of AN-137B at the selected dose. We also preliminary tested different oseltamivir doses and virus concentrations before selecting the parameters proposed in this study. Consequently, by using a PR8 inoculum of 200 PFU, we expected to obtain a 100% mortality rate in untreated animals and we also targeted a minor protection with 1 mg/kg of oseltamivir to better see eventual benefits of the combination. Animals were initially weighed and randomly assigned to different treatment groups. Treatments started 4 h before viral inoculation. Finally, a group of uninfected animals was used as control. Experimental viral infection was performed under isoflurane anaesthesia by intranasal inoculation of 35 µL of PBS containing 200 plaque forming units (PFUs) of influenza A/Puerto Rico/8/1934 (H1N1) virus. Mice were weighed daily for 14 days. Animals whose weight loss reached ≥20% from their initial weight were humanely euthanized. Four mice per group were sacrificed on day 5 post-infection (p.i.), and their lungs were aseptically removed and homogenized for determination of lung viral titres (LVTs) and cytokine levels (
For the determination of LVTs, harvested lung tissues were homogenized in 1 mL of PBS containing 2 x antibiotic solution (penicillin, streptomycin and amphotericin B) using Omni Tip homogenizer (OMNI International, GA, USA). Cells were pelleted by centrifugation (600 g, 5 min) ,and supernatants were titrated by plaque assay on ST6GalI-MDCK cells. Supernatants of lung homogenates were also used for viral RNA (vRNA) extraction with the MagNA Pure LC system (Total nucleic acid isolation kit, Roche Molecular System, Laval, QC, Canada), followed by reverse transcription quantitative PCR (qRT-PCR) assay as previously described. 11 The assay was performed with the QuantiTect Virus + ROX Vial Kit (Qiagen, Toronto, ON, Canada) on a LightCycler 480 system (Roche Molecular System). The results are expressed as number of vRNA copies per mL.
Interleukin-6 (IL-6) and interleukin-1 beta (IL-1β) levels in lung homogenates from infected mice were determined using specific mouse Enzyme Linked Immunosorbent Assay (ELISA) kits in duplicate following the manufacturer’s instructions (Sigma-Aldrich).
Statistical analyses
All analyses were made using the PRISM software (GraphPad Inc, San Diego USA). Viral titres and cytokine levels in lung homogenates of mice were compared using the unpaired Student’s t test. Mouse body weight changes and survival rates were compared with one-way ANOVA and the Chi-square test, respectively.
Results
As expected, intranasal inoculation of mice with 200 PFUs of influenza A/Puerto Rico/8/1934 (H1N1) virus resulted in clinical signs of infection such as body weight loss, lethargy and mortality that were observed in the four infected groups of mice with no signs being observed in uninfected animals. As shown in Figure 1, 4/5 animals (80%) in untreated and infected animals and all animals (5/5; 100%) in groups that received single oseltamivir or AN-137B treatment were humanely euthanized or died between day 7 and day 9 p.i. Only, 2/5 animals (40%) of mice that received the combination of oseltamivir and AN-137B were humanely euthanized. Accordingly, important body weight losses, in particular during days 5–9 p.i., were observed in infected animals while no signs of infection were observed in the uninfected group (Figure 2). Of interest, body weight loss was remarkably lower in the group that received the combined oseltamivir and AN-137B treatment (Figure 2). Survival curve of mice. Kaplan–Meier survival curves of non-infected (NI) and infected mice with 200 PFUs of influenza A/Puerto Rico/8/1934 (H1N1) virus. Four groups of infected- mice included a group that received saline (untreated) and those that received single or combined therapy with oseltamivir (gavage) and AN-137B (intraperitoneal) (1 mg/kg, once daily for 5 days). Mortality was recorded in subgroups of 5 animals during 14 days post-inoculation and analysed using the log-rank (Mantel–Cox) test. Body weight losses of mice. Mean percent weight changes ± standard deviation (as compared to initial weights) of mice inoculated intranasally with 200 PFUs of influenza A/Puerto Rico/8/1934 (H1N1) virus were recorded daily until day 14 post-inoculation. Infected mice included a group that received saline (untreated) and those that received single or combined therapy with oseltamivir (gavage) and AN-137B (intraperitoneal) (1 mg/kg, once daily for 5 days). *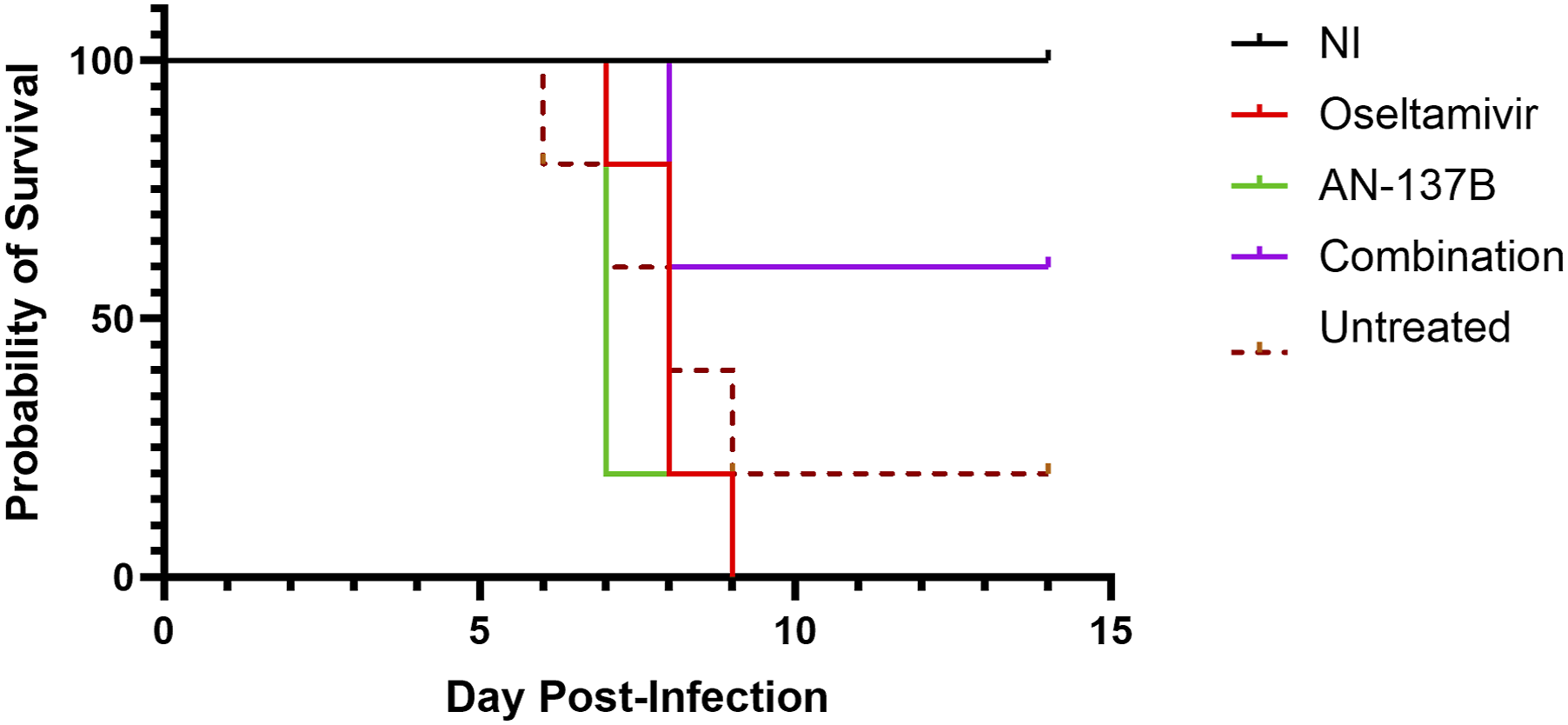

Mean LVTs determined on day 5 p.i. were comparable in the group of untreated mice and those of animals that received single oseltamivir or AN-137B treatment (4.76 ± 0.9 × 105, 3.03 ± 0.83 × 105 and 3.86 ± 0.68 × 105 PFU/mL, respectively). By contrast, the group of mice that received the combined treatment had a mean LVT of 2.53 ± 0.63 × 105, which was significantly lower than the untreated group ( Mouse lung viral titres. Mean lung viral titres ±standard deviation for 4 mice infected intranasally with 200 PFUs of influenza A/Puerto Rico/8/1934 (H1N1). Titres were determined on day 5 post-inoculation by using plaque assays in ST6-GalI-MDCK cells (a) and by quantification of vRNA by qRT-PCR (B). Data were analysed by using the one-way ANOVA test (GraphPad). *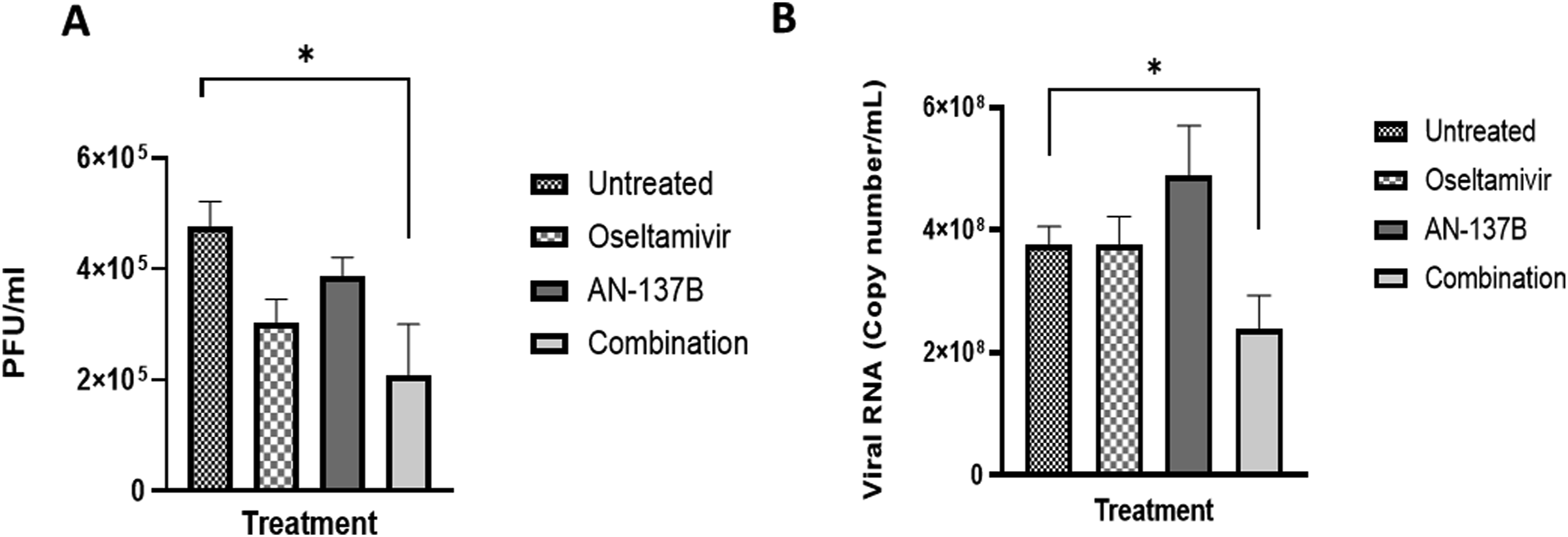
Levels of the pro-inflammatory cytokine IL-1β in lung samples were significantly lower in the groups that received oseltamivir alone or oseltamivir in combination with AN-137B, compared to the untreated group (135.40 ± 17.92 pg/mL, 128.55 ± 28.55 pg/mL and 160.42 ± 3.12 pg/mL, respectively) while IL-1β levels of the AN-137B group were not significantly reduced compared to the untreated group (141.65 ± 6.24 pg/mL (Figure 4(A)). A similar pattern was observed regarding IL-6 lung levels, that is, 565.00 ± 99.08 pg/mL, 501.20 pg/mL, 654.57 ± 13.98 pg/mL and 763.85 ± 102.35 pg/mL for the groups that received oseltamivir, oseltamivir + AN-137B, 137B and no treatment, respectively (Figure 4(A)). Cytokine levels in mouse lungs. Levels of IL-1-β (a) and IL-6 (b) were determined in lung homogenates of mice infected intranasally with 200 PFUs of influenza A/Puerto Rico/8/1934 (H1N1) virus by ELISA. Results are expressed as the mean of protein concentration in pg/mL ± standard deviation of 4 mice per group from two independent experiments. *, 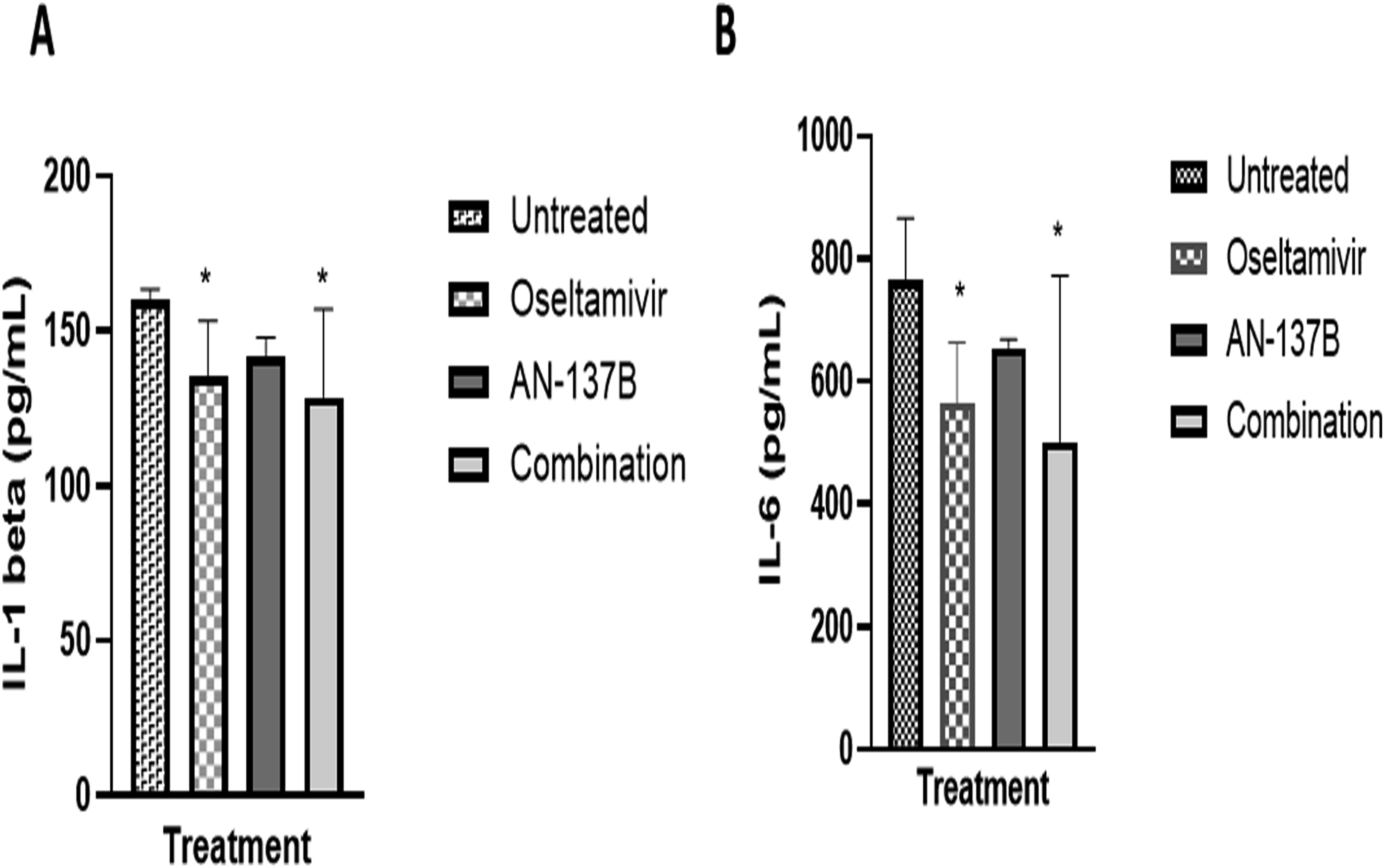
Discussion
Influenza A strains, mainly from the H1N1 and H3N2 subtypes, circulate each winter season throughout the world causing serious public health and economic problems. In most people, seasonal influenza viruses replicate in the upper respiratory tract inducing a self-limited disease, whereas viral replication may progress towards the lower respiratory tract, potentially triggering secondary bacterial infections and pneumonia in the elderly, young children and immunocompromised patients.
2
Influenza vaccines have the potential to mitigate the impact of influenza epidemics and eventual pandemics; however, the protection afforded by influenza vaccines could be much lower in high-risk groups. Moreover, antigenic drifts, that occur at regular intervals in seasonal influenza A viruses, may result in a significant decrease in vaccine effectiveness.
13
Antiviral agents, including neuraminidase inhibitors (oseltamivir, zanamivir, peramivir and laninamivir) and compounds targeting the viral polymerase complex, such as baloxavir marboxil, constitute another important means for the management of seasonal influenza and are expected to play a major role in the advent of influenza pandemics. However, as for other antivirals, such as amantadine and rimantadine (in the adamantanes class), the emergence of resistant viruses constitutes a serious threat that may compromise the clinical utility of these agents. Several
The aim of this study was to investigate whether, in agreement with our recent
Due to our institutional animal care committee restrictions, we used a limited number of mice per group for survival (
As AN-137B derives from the pro-resolving lipid mediator PDX, we expected that AN-137B would exhibit
Conclusion
In conclusion, these
