Abstract
Keywords
Introduction
Differential equations play a prominent role in various fields such as physics, chemistry, biology, mathematics, engineering, and other disciplines.1–6 There are few phenomena in different fields of science occurring linearly. Most problems are essentially nonlinear and are described by nonlinear differential equations. Finding exact or approximate solutions of these nonlinear equations is interesting and important. However, its exact solution might be too complex to use for a practical application, or sometimes, it is impossible to obtain its exact solution. To overcome this difficulty, there are numerous methods undertaken to find out numerical as well as analytical series solutions for nonlinear problems: Variational Iteration Method (VIM), 7 Adomian Decomposition Method (ADM), 8 Homotopy Analysis Method (HAM), 9 Harmonic Balance Method (HBM), 10 Homotopy-Perturbation Method (HPM),11,12 Haar wavelet quasilinearization method, 13 and Haar wavelet operational matrix method 14 are some proven instances. Although the nonlinear analytical techniques are fast developing, they still do not completely satisfy the mathematicians and engineers.
Nonlinear chemistry problems are considered nonlinear reactions with oscillatory influx of reactant to the response medium. The chemical kinetics problem is a system of three-dimensional non-linear ordinary differential equations (ODEs), and it is regarded as Rober problem
15
in the field of chemistry. Oscillations and the frequency are happened in dynamic systems in every area of science such as beating of the human heart, Lotka–Volterra system in ecology. Chemical systems, which are held very-far-from-equilibrium, exhibit a broad range of phenomena: chemical entropy, which may develop into stable states or give rise to chemical oscillations or chaotic dynamics.
16
The dynamical model of the chemical kinetics system that appears in chemical applications is as follows
There are a few literatures that deal with the mentioned chemical kinetics system. The
Recently, a great attention has been given to the application of semi-analytical technique such as the iterative method (IM or DJM), which was introduced by Daftardar-Gejji and Jafari 26 and further modified by Bhalekar and Daftardar-Gejji.27–29 Very recently, the IM was employed for solving various kinds of nonlinear mathematical problems that can be found in the following literatures.30–33 In this research, we have applied the IM to unravel the nonlinear chemical kinetics equations. The main advantages of IM are its easy solutions procedure, and sometimes, within a few iterations, we can obtain the decent accuracy.
This paper is organized as follows: in “The solution approach based on the iterative method” section, we provided the outline of the solution approach based on the IM. In “Formulation and mathematical framework of chemical kinetics systems” section, we illustrated the chemical kinetics reactions and their applications. Also, we formulated the mathematical equations based on the rate of reactions. In “Analytical solutions,” we applied the solution approach to the nonlinear chemical kinetics models. In “Results analysis” section, we have discussed the results in detail. Finally, the concluding remarks are given in final section.
The solution approach based on the iterative method
The iterative method (IM) was suggested by Daftardar-Gejji and Jafari. 34 The solution procedure of the method is discussed below:
Let us consider the general equation
Here, 
The nonlinear operator 
Then series solution becomes
We define the consecutive relation



Then
Formulation and mathematical framework of chemical kinetics systems
Chemical kinetics reactions
Chemical kinetics, also known as reaction kinetics, is a physical chemistry branch concerned with understanding the rate of chemical reactions. Various frequency calculations can be combined to derive kinetic parameters for the study of chemical kinetics reaction.35,36 The frequency of the molecular collisions has the proper orientation between particles and the appropriate temperature so that the reaction can occur.37,38 Also, the action of external vibrations with the frequency coinciding with that of intrinsic intermediate concentration and oscillations can increase the reaction rate. 39 Experimental studies of the chemical reaction dynamics of vibrationally excited molecules reveal different vibrations’ ability to control the course of a chemical reaction. 40
Let us assume three compounds of a model of chemical reaction which are denoted by 


The concentrations of chemical compounds
Mathematical models explaining chemical reaction kinetics provide chemists and chemical engineers with tools to better understand and explain chemical processes such as food decomposition, microorganism formation, stratospheric ozone decomposition, and the chemistry of biological systems.6,41 These models may also be used in the design or alteration of chemical reactors to maximize product yield, isolate materials more effectively, and remove environmentally damaging by-products. 42
Mathematical model of chemical kinetics systems
In order to demonstrate the chemical reactions discussed above, the following example arises in chemistry problems20,21





Analytical solutions
Case I
Integrating the system of equations (14) to (16) from 0 to 


We let the following non-linear terms


In view of equations (6) to (9), we obtain the successive components of the IM solution for the selected model as






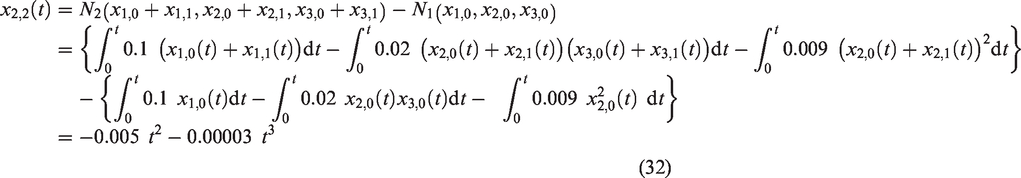




Case II
Integrating the system of equations (14) to (16) from 0 to 


We let the following non-linear terms


In view of equations (6) to (9), we obtain the successive approximations of the IM solution for the selected model as


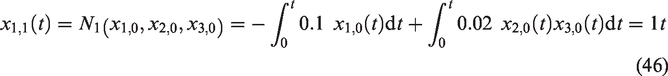








Results analysis
In general, there are no exact solutions for the non-linear chemical kinetics systems of ODEs. Thus, the validity of IM has compared with RK4 method. The IM algorithm is coded in the computer algebra package Maple together with the Maple built-in fourth-order Runge–Kutta method. The Maple environment variable digits controller is set to 16 in all calculations. The proposed technique is rapidly convergent to RK4 solutions. Also, we have obtained very good explicit series solutions by using the IM with five iterations only in all cases.
Case I
The numeric results of RK4 are obtained with the constant step size
Numeric differences between IM with RK4 solutions in chemical kinetics problem for Case I.
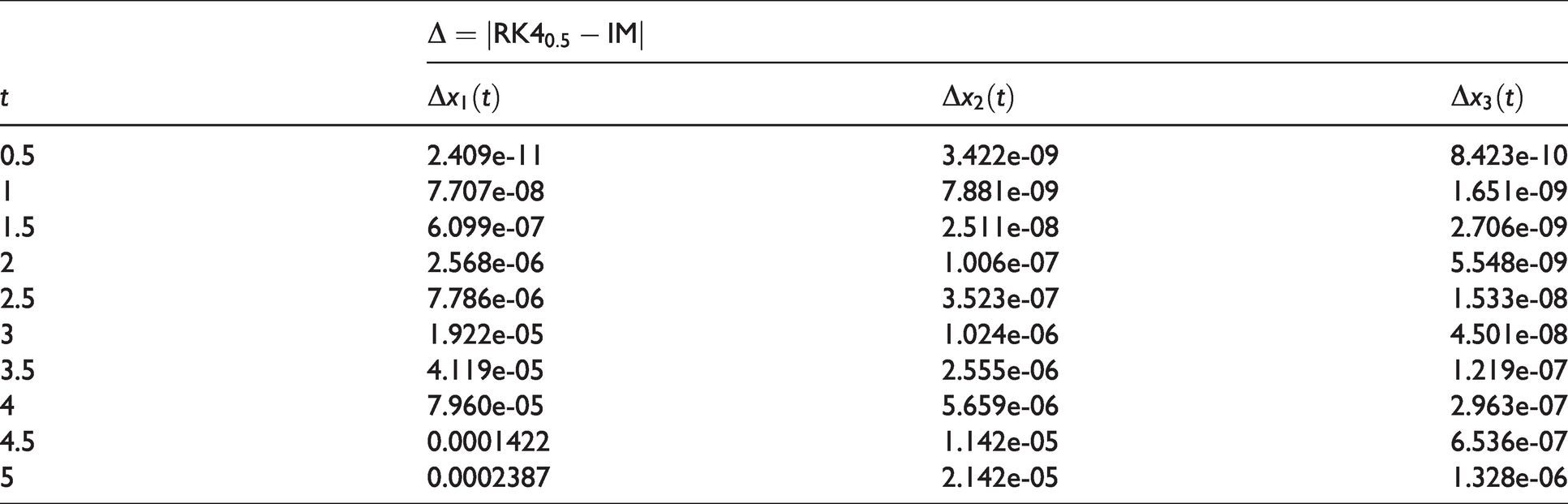

Comparison among IM (solid blue line), RK4 (space red box) for (
Case II
Similar to the previous case, we have compared the numeric solutions of IM to those of RK4. We determined the solution of fourth-order Runge–Kutta (RK4) for the constant time increments
Numeric differences between IM with RK4 solutions in chemical kinetics problem for Case II.


Comparison among IM (solid blue line), RK4 (space red box) for (
The nonlinear systems can be solved by using various numerical methods. However, numerical methods, such as RK4, cannot come up with analytical series solutions. 43 Yet, semi-analytical methods such as IM provide explicit solutions as well as numeric solutions. The first few iteration steps of IM 30 are quite similar to those of the HPM, 23 VIM, 44 and ADM 45 techniques. Although the solution procedure of IM is relatively easy.
Conclusion
In order to achieve approximate solutions for the nonlinear chemical kinetics systems, a semi-analytical method based on IM has been introduced. The approximate numeric precisions of IM have made an excellent agreement with the numeric precisions of RK4. The prime benefits of the proposed technique are its computer-friendly environment, adaptability, and avoidance of small parameters in the computation task. Also, the solution procedure is straightforward and simple as compared with the existing methods. It can be concluded that IM is a significant tool for solving nonlinear system of differential equations. The iteration algorithm proposed in this study is also effective for other nonlinear problems in the upcoming days.
