Abstract
Introduction
The onset of menstruation is the hallmark of female reproductive maturity. 1 It marks the interface between girlhood and womanhood; hence, it is an important aspect of female reproductive health.2,3 Unfortunately, the onset of menstruation is usually coupled with challenges that affect between 50% and 95% of women.2,4 These challenges include delayed menses, irregular menstrual periods, painful menstrual periods, and heavy menstrual bleeding, among others.1,4,5 Of these, painful menstrual periods, clinically referred to as dysmenorrhea, are the most prominent. 6 Dysmenorrhea is characterized by mild to severe lower abdominal pain just before and/or during the menstrual period. 6 It is usually associated with headache, dizziness, bloated feeling, nausea, vomiting, backache, and leg pain. 7
Dysmenorrhea is classified into either primary or secondary types. Primary dysmenorrhea is when the condition occurs without an identifiable organic pathology.3,6 In contrast, secondary dysmenorrhea results from anatomic and/or evident pelvic pathology such as endometriosis, chronic pelvic inflammatory disease, and adenomyosis. 3 The latter condition is less common compared to the former. 1 Dysmenorrhea is one of the most leading reasons for gynecological consultations, 5 with a global prevalence of up to 90%. 2 Unfortunately, there is paucity of reports on the prevalence of dysmenorrhea in Zimbabwe. This may be due to ethno-societal beliefs, which perceive menstrual pain as a natural phenomenon that must be endured leading to underdiagnosis. 8 It may be because of various other factors that are yet to be explored.
Dysmenorrhea negatively affects the relationships, professional, recreational activities, and emotional well-being of sufferers. 4 Among college students and school-going girls, dysmenorrhea can be incapacitating leading to absenteeism from school or work and, in some severe cases, hospitalization, thus affecting productivity and quality of life. 9 Also, sociodemographic, lifestyle, and dietary factors such as ethnicity, place of residence, marital status, smoking cigarettes, alcohol, and coffee consumption, respectively, have all been linked to the prevalence and/or severity of dysmenorrhea.1,4,6,8 Therefore, an increased understanding of the status of dysmenorrhea among women in Zimbabwe is required. This will inspire strategies to prevent or alleviate the social and financial burden dysmenorrhea place on students, families, and communities. In this regard, this study aimed to determine the prevalence of dysmenorrhea and to identify associated risk factors among university students in Zimbabwe.
Material and methods
Study design and setting
A self-designed, semi-structured questionnaire was used in a cross-sectional study to assess the prevalence of dysmenorrhea and associated risk factors among female students enrolled at the Midlands State University (MSU) using the method described by Ameade et al. 1 with modifications. MSU is one of the oldest and most well-established universities in postindependence Zimbabwe. It is situated in the city of Gweru, the capital of the Midlands province of Zimbabwe. MSU has 10 faculties and 4 Institutes offering undergraduate and postgraduate degrees to students enrolled from all 10 provinces of Zimbabwe.
Inclusion and exclusion criteria
Female students who were registered for studies at MSU aged 18–40 years (undergraduate or postgraduate) were eligible to participate in the study. They were required to be accommodated at MSU Gweru's main campus residence. The study excluded female students who were not willing to participate in the study. Female students who had a known history of pelvic pathology or underlying gynecological diseases such as adenomyosis, pelvic inflammatory diseases, endometriosis, and hysteromyoma were also excluded from the study.
Sample size determination
Sample size was estimated using single proportion sample size formula for prevalence studies as described by Dobson et al., 10 and it was calculated as follows
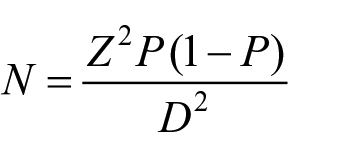
The following assumptions were considered:
Sampling technique and procedure
The study participants were chosen using a simple random sampling technique.1,11 This was done by first obtaining the occupancy lists of all the female students in each of the 17 female Halls of residence at MSU. A code was then assigned against each student’s name, creating a new sampling frame. By using these numerical codes, the required number of study participants was randomly selected at each Hall of residence using OpenEpi computer software. The selected assigned codes were then used to ask for consent to participate in the study.
The study questionnaire was prepared in English and was formulated by referencing similar studies,1,6 modifying it to fit research objectives. Prevalence of dysmenorrhea, menstrual characteristics, and factors associated with dysmenorrhea over the previous 6 months were assessed. This was done to limit recall bias among respondents. 4 Demographics and lifestyle factors such as current age, age of menarche, marital status, maternal status, physical exercise, smoking, alcohol, and coffee consumption were considered. Questionnaires were piloted among 20 randomly selected female students across all fields of study. This was done to ensure the correction of elusive questions before it was administered for actual data collection. Prior to the distribution of questionnaires, the principal investigator and trained research assistants provided a brief explanation of the study contents including the meaning of any medical/gynecological terms that might be difficult to understand. Respondents were also encouraged to ask the research assistants to clarify any doubts pertaining to the questions from the questionnaire. The study was conducted between June and October 2020.
Operational definitions
Physical exercise refers to full body activity for at least 30 min, three times a week.6,12 Consumption of coffee (caffeinated) was frequent if coffee was consumed at least once every 2 days. 6 Cigarette smoking and alcohol consumption was evaluated on a dichotomous level (Yes or No) with questions including: do you smoke cigarettes; do you drink alcohol, respectively. 13 A respondent was identified as a smoker; they smoked at least one cigarette per day, while alcohol drinking referred to consuming at least one glass of wine (250 mL) per month. 6 Dysmenorrhea was considered based on the questions: have you experienced abdominal pain/cramps just before/during menstruation in the past 6 months? Do you suffer from the following conditions adenomyosis, pelvic inflammatory diseases, endometriosis, and hysteromyoma (the conditions were briefly explained before the distribution of questionnaires)? 6 Menstrual pain refers to lower abdominal pain/cramping, which may be associated with dizziness, headache, nausea, vomiting, fatigue, diarrhea, insomnia, and irritability. Menstrual pain was considered to be mild when it was experienced without inhibiting school/daily activities and did not necessitate the use of analgesic, moderate if school/daily activities were affected, requiring an analgesic which gave relief. Severe menstrual pain referred to pain that clearly inhibited school/daily activities with poor response to analgesics.4,14,15 Menstrual cycles were considered to be regular if they ranged between 21 and 35 days. 14 Self-reported lifestyle and menstrual characteristics such as physical exercise, smoking, alcohol consumption, menstrual pain, and regularity were considered for the previous 6 months. 14 Family history of dysmenorrhea was considered as positive if the respondent’s first-degree relative (mother or sister) suffered from dysmenorrhea. 14 The impact of dysmenorrhea on school/daily activity was evaluated by a single question with two responses (Yes or No) with relation to daily life, attention in class, homework, social activities, sports, work, family relations, relationships, and sexual activities.1,15
Statistical analyses
Data were entered into Epi Info version 7.1.1 and then imported into STATA version 16 for statistical computations. Continuous variables were tested for normality using the Shapiro–Wilk test. For normally distributed variables, mean and standard deviation (SD) were reported. Median and interquartile range (IQR) were used for nonnormal variables. Categorical variables were presented using frequencies and percentages (%). To test for the association between the presence of dysmenorrhea and independent variables, the Pearson chi-square test was used or Fisher’s exact test, where appropriate. All variables with a p-value < 0.20 in the bivariate model were considered candidates for the Stepwise Multivariable logistic regression model (MLR). The strength of associations was summarized using prevalence odds ratios (PORs) and corresponding 95% CIs estimated from the final model. The Hosmer–Lemeshow test was used to evaluate the goodness-of-fit for the model (p < 0.05), indicating a significant lack of fit. For statistical significance, a p-value < 0.05 was considered.
Results
A total of 420 questionnaires were distributed, and 400 participants responded, giving a response rate of 95% which was considered satisfactory for this study. Of the 400 questionnaires returned, 18 were excluded from the analysis due to incompleteness resulting in 382 evaluable samples.
Sociodemographic characteristics
The sociodemographic characteristics of the respondents are summarized in Table 1. The minimum and maximum age of the respondents was 18 and 35 years, respectively, with 52.6% aged between 21 and 25 years. Up to 91.1% of the respondents were single, 8.6% were married, and one (0.3%) was a widow.
Sociodemographic characteristics (n = 382).

SD: standard deviation.
Prevalence of dysmenorrhea and its relationship with sociodemographic characteristics
The overall prevalence of self-reported dysmenorrhea in this study was 75.9% (95% CI: 71.4% to 79.9%). The majority of respondents (89.3%) (259/290) reported that dysmenorrhea affects their school/daily activities, while 10.7% (31/290) were not affected. The prevalence of dysmenorrhea was significantly associated with disruption of school/daily activities (χ2 = 18.22, p < 0.001). The relationship between prevalence and sociodemographic characteristics is shown in Table 2. Just above half of respondents (52.8%) (153/290) who suffered from dysmenorrhea were from the age group of 21–25 years. Furthermore, 91.7% (266/290) of respondents who reported dysmenorrhea were single, while 8.3% (24/290) were married. No significant association was found between sociodemographic variables and dysmenorrhea (p > 0.05).
Prevalence of dysmenorrhea and its relationship with sociodemographic characteristics.

df: degrees of freedom; χ2: chi-square test.
Fisher’s exact test.
Lifestyle characteristics and their relationship with dysmenorrhea
Lifestyle characteristics and their relationship with dysmenorrhea are shown in Table 3. The majority of respondents did not smoke cigarettes (97.1%) (371/382) or consume alcohol (85.6%) (327/382). Also, the majority (64.2%) (245/382) drink coffee frequently or infrequently. In addition, half (50.3%) (192/382) did not physically exercise. Physical exercise, smoking, alcohol, and coffee consumption were not significantly associated with dysmenorrhea (p > 0.05).
Lifestyle characteristics and their relationship with dysmenorrhea.

df: degrees of freedom; χ2: chi-square test.
Reproductive and menstrual characteristics and their relationship with dysmenorrhea
Reproductive and menstrual characteristics and their relationship with dysmenorrhea are shown in Table 4. More than half (55.2%) (160/290) of the respondents who reported having dysmenorrhea also had a family history of the condition. A family history of dysmenorrhea was significantly related to the prevalence of dysmenorrhea (χ2 = 4.28, p = 0.04). The mean age of menarche was 13.7 years (SD = 1.39), ranging from 9 to 17 years. The age of menarche was significantly lower among those who suffered from dysmenorrhea (13.6 years) compared to those who did not (14 years) (p = 0.007). Age of menarche was significantly associated with dysmenorrhea (χ2 = 14.8, p < 0.001). Up to 73.6% (281/382) reported regular menstrual cycles, and this was significantly associated with dysmenorrhea (χ2 = 18.1, p < 0.001). The majority (93.4%) (271/290) of respondents who reported dysmenorrhea were nulliparous. Dysmenorrhea prevalence decreased with increased parity (χ2 = 8.8, p = 0.03). Menstrual pain was mild, moderate, and severe in 10.3% (30/290), 61% (177/290), and 28.6% (83/290) respondents, respectively.
Reproductive and menstrual characteristics and their relationship with dysmenorrhea.

df: degrees of freedom; SD: standard deviation; χ2: chi-square test.
Statistically significant.
Fisher’s exact test.
Multivariable logistic regression analysis of significant variables related to dysmenorrhea
Multivariable logistic regression estimates of the association between independent variables and self-reported dysmenorrhea are presented in Table 5. The odds of dysmenorrhea were 1.68 times more prevalent among respondents with family history of dysmenorrhea compared to those with no history (POR = 1.68 (95% CI: 1.03 to 2.75, p = 0.040)). Age of menarche was strongly associated with dysmenorrhea, with 81% of females who had menarche at the age 13–15 years having lower odds of dysmenorrhea compared to those who had it below the ages of 13 years (POR = 0.19 (95% CI: 0.10 to 0.45, p < 0.001)). The odds of dysmenorrhea decreased by 85% for every second pregnancy a female had, holding other variables constant (POR = 0.15 (95% CI: 0.03 to 0.82, p = 0.029)). Respondents with irregular menstrual cycles had 86% lower odds of developing dysmenorrhea in this population (POR = 0.14 (95% CI: 0.10 to 0.33, p < 0.001)).
Logistic regression analysis of significant variables related to dysmenorrhea.

CI: confidence interval; POR: prevalence odds ratio.
Discussion
A high dysmenorrhea prevalence of 75.9% was noted in this study. This is comparable to prevalence rates recorded among university students in Uganda (75.8%), 2 Ghana (74%), 8 and Rwanda (77.3%), 16 though higher compared to those recorded in Ethiopia (51.3%) 4 and Cameroon (56.20%). 17 Meanwhile, dysmenorrhea was higher among university students in Egypt (92.9%) 18 and Nigeria (83.1%) 19 than our study. Variations in the prevalence of dysmenorrhea reported in these African studies also follow a global pattern. For instance, compared to our study, very low dysmenorrhea prevalence was recorded in Japan (15.8%), 20 Lebanon (26.8%) 21 and China (41.7%) 6 while higher prevalence was recorded in Ireland (91.5%), 22 Iran (89.1%), 23 and Greece (89.2%). 24 All these were cross-sectional studies conducted among university/college students. The reasons for variations in the prevalence of dysmenorrhea among all these studies may be due to differences in study settings, assessment methods, and respondents age groups, among others.3,8 However, even with such differences, dysmenorrhea prevalence that is above 65%, such as in our study, is still classified as high.4,5,25 This implies that dysmenorrhea invisibly persists and could be underdiagnosed and undertreated in Zimbabwe, drawing the need for awareness of the condition and its burden.
Various studies have shown that sociodemographic factors such as marital status, ethnic origin, and place of residence are associated with dysmenorrhea.5,9,26 However, in our study, dysmenorrhea was independent of all these factors tallying with reports from similar studies by Tavallae et al. 27 and Ameade et al., 1 who also found no association between dysmenorrhea and these factors. Noteworthy, though, dysmenorrhea disrupts the school/daily activities of the majority of the respondents who reported having the condition in our study. This is consistent with reports from other African countries like Ghana, 1 Cameroon, 17 and Ethiopia14,28 where similar dysmenorrhea studies abound. This implies that the adverse effects of dysmenorrhea among Zimbabwean university students ought not to be overlooked. There is a need for widespread public education to encourage women with dysmenorrhea to seek medical care, especially when it is severe.
When it comes to lifestyle habits, alcohol consumption, cigarette smoking, coffee consumption, and physical exercise are well-reported association factors of dysmenorrhea.1,9,29 Alcohol consumption is reported to be linked with estrogen, testosterone, allopregnanolone (a derivative of progesterone), and luteinizing hormonal imbalances, which cause painful and irregular menstrual cycles. 9 Meanwhile, the nicotine in tobacco and caffeine in coffee are both known to cause vasoconstriction leading to hypoxia-induced myometrial contraction. 29 In our study, alcohol consumption and cigarette smoking were not common among respondents despite the fact that the lifestyle is becoming more fashionable among young people globally.30,31 A couple of reasons could be attributed to this observation especially considering the sociocultural context of the study. For instance, consumption of alcohol is prohibited on campus at MSU, and campus enforcement rules may be quite restrictive. It could also be that the lifestyle of drinking and smoking is still considered to be a social taboo for women in Zimbabwe, just like in some African societies 32 leading to underreporting. Whatever the reason might be, both smoking and drinking were not associated with the prevalence of dysmenorrhea in the present study. These findings are consistent with reports by Nakame et al. 2 and Yesuf et al., 5 who also recorded no association between smoking and drinking lifestyle with dysmenorrhea. Similarly, coffee intake was not associated with the prevalence of dysmenorrhea in our study despite having a relatively higher intake among those with dysmenorrhea. This is also consistent with reports from comparable studies by Nakame et al. 2 and Okoro et al. 33 but contradicts studies by Ju et al. 34 and Unsal et al., 35 who recorded an association between coffee intake and dysmenorrhea.
The effect of physical exercise on dysmenorrhea is yet to be fully established. This is because, on the one hand, lack of exercise is reported to be associated with dysmenorrhea, 36 while on the other, physical exercise is reported to improve symptoms associated with dysmenorrhea. 11 In another study, intense exercise, especially among athletes, is reported to be associated with severe dysmenorrhea. 37 In our study, physical exercise was not associated with the prevalence of dysmenorrhea, just as in studies by Nakame et al., 2 Yesuf et al., 5 and Ameade et al. 1 Inconsistent reports on the relationship between physical exercise and dysmenorrhea may be due to differences in evaluation of physical exercise and definition of terms. For instance, physical exercise was only considered if there was full body activity at least thrice a week for at least 30 minutes in our study. Meanwhile, in a study by Muluneh et al., 11 physical exercise was self-reported as: not at all, irregular, or regular.
Family history is a well-reported association factor of dysmenorrhea.14,25 One of the most touted explanations for this association postulates that dysmenorrhea prevalence is hereditary.8,30 In addition, Osonuga et al. 8 propose that the prevalence of dysmenorrhea is higher among those with a family history due to conditioned behavior that is learned from mothers or sisters for potential societal rewards. In the present study, family history was associated with the prevalence of dysmenorrhea consistent with analogous studies by Yesuf et al., 5 Osonuga, 8 Azagew et al., 25 Nloh et al., 17 and Hailemeskel et al. 28 Those with a family history of dysmenorrhea were almost twice likely to suffer from dysmenorrhea in the present study. The risk was comparable to that reported by Hu et al. 6 and Giletew and Bekele 14 but lower compared to a report by Tadese et al., 4 where the risk was three times.
Dysmenorrhea was severe in over a quarter (28.6%) of respondents who suffered from this condition in our study. This percentage was comparable to that reported by Ameade 1 (22.4%), higher than that reported by Osonuga 8 (14.3%) but lower compared to that reported by Giletew and Bekele 14 (58.7 %). The most plausible reason for these differences includes cultural disparities in menstrual pain perception, thresholds, and evaluation models.1,4 For instance, in our study as well as studies by Ameade et al. 1 and Giletew and Bekele, 14 menstrual pain was self-reported, while Tadese et al., 4 Osonuga et al., 8 and Hailemeskel et al. 28 used multidimensional scoring system.
Meanwhile, it is postulated that dysmenorrhea begins a few months after menarche, peaking during the late teens and early twenties. 11 Thereafter, its prevalence and severity decline with age, influenced by increasing parity in the later years of a woman’s life.8,11 This agrees with the present study observations, where an increase in parity and age of menarche was associated with a decrease in the prevalence of dysmenorrhea. However, dysmenorrhea was more common among those with regular compared to irregular menstrual cycles. This is contrary to reports by Tadese et al., 4 Hu et al., 6 and Ameade et al. 1 where dysmenorrhea was associated with irregular menstrual cycle. Nonetheless, from the present study, most dysmenorrhea sufferers were aged between 21 and 25 years, and most sufferers experienced menarche between the ages of 9 and 15 years. This implies that menstrual pain is a burden to female students in Zimbabwe during their critical academic periods in primary school, high school, and university.
Limitations of the study
While student enrollment at MSU is from all provinces of Zimbabwe, which provides representation in terms of participation, findings from one university may be limited to indexing the entire body of female students in the country. Also, since this is a maiden study on dysmenorrhea prevalence in Zimbabwe, it was a challenge to find comparison studies locally and regionally. Reporting biases, over and underreporting were inevitable given that respondents were asked to report on events like the age of menarche, which may have occurred in the distant past. Self-administered data collection was applied, and this might have added social desirability bias. Other confounders like the presence of disease/illness were not considered in this study. For instance, dysmenorrhea was self-reported and not based on clinical exclusion of underlying pathology.
Conclusion
To our knowledge, this is the first study to identify dysmenorrhea as a very common condition among university students in Zimbabwe. Family history, regular menstrual cycle, nulliparity, and lower age of menarche were associated risk factors for dysmenorrhea. Since dysmenorrhea was prominent during the critical academic years of female students’ studies, we recommend schools and universities to offer support and raise awareness among students about dysmenorrhea. Further studies in all Zimbabwean provinces and among adolescent girls are recommended for more representative findings. Importantly, the impact of dysmenorrhea and management methods used by women also need to be explored. This will facilitate the design of interventional strategies that will improve the quality of life of women suffering from dysmenorrhea in the nation and beyond.
Supplemental Material
sj-docx-1-whe-10.1177_17455057231189549 – Supplemental material for Prevalence of dysmenorrhea and associated risk factors among university students in Zimbabwe
Supplemental material, sj-docx-1-whe-10.1177_17455057231189549 for Prevalence of dysmenorrhea and associated risk factors among university students in Zimbabwe by Trust Nyirenda, Elliot Nyagumbo, Grant Murewanhema, Nyasha Mukonowenzou, Shingirai Brenda Kagodora, Cladnos Mapfumo, Michael Bhebhe and Jacob Mufunda in Women’s Health
Supplemental Material
sj-docx-2-whe-10.1177_17455057231189549 – Supplemental material for Prevalence of dysmenorrhea and associated risk factors among university students in Zimbabwe
Supplemental material, sj-docx-2-whe-10.1177_17455057231189549 for Prevalence of dysmenorrhea and associated risk factors among university students in Zimbabwe by Trust Nyirenda, Elliot Nyagumbo, Grant Murewanhema, Nyasha Mukonowenzou, Shingirai Brenda Kagodora, Cladnos Mapfumo, Michael Bhebhe and Jacob Mufunda in Women’s Health
Footnotes
Declarations
Supplemental material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
