Abstract
Introduction
The use of neoadjuvant chemotherapy (NACT) in non-metastatic, invasive breast cancer aims to reduce the risk of distant recurrence by eradicating micrometastatic disease at an early stage. 1 This approach is also employed to downstage the extent of disease in the breast and/or regional lymph nodes, thereby providing valuable information to guide subsequent adjuvant therapies. 2 Downstaging can facilitate less extensive surgical interventions, including breast-conserving procedures, thereby enhancing cosmetic outcomes and reducing postoperative complications. 3 Furthermore, the extent of residual invasive disease, referred to as pathological complete response (pCR), after NACT serves as a strong prognostic factor for recurrence risk and overall survival. 4
Breast cancers have traditionally been categorized as either HER2-positive or HER2-negative based on immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) testing. Specifically, tumors with IHC 0 or 1 + are considered HER2-negative, and IHC 2 + cases with negative FISH results are also classified as HER2-negative, whereas IHC 3 + or FISH-positive tumors are defined as HER2-positive. However, a distinct entity known as HER2-low breast cancer, characterized by low HER2 expression (IHC 1 + or 2 + with negative FISH), has recently emerge. 5 Approximately 15% of breast cancer cases are classified as HER2-positive, while 55% to 60% of all breast cancers are characterized as HER2-low. 6 Among HER2-low tumors, about 80% exhibit a luminal-like molecular profile and 15–20% are triple-negative. 7 This subgroup has challenged the conventional binary classification of HER2 status. Notably, the DESTINY-Breast04 trial demonstrated the efficacy of a HER2-targeted therapy in previously treated metastatic HER2-low breast cancer, contradicting earlier assumptions that these tumors are unresponsive to HER2-directed agents. 8 These findings underscore the need for further research to clarify the biological and clinical behavior of HER2-low breast cancer and to establish optimal management strategies for this newly defined entity.
In the present study, we aimed to evaluate the impact of HER2-low positivity, recently recognized as clinically relevant in metastatic settings but not yet a routine consideration in neoadjuvant or adjuvant therapy, on hormone-receptor-positive (HR-positive) breast cancer patients receiving NACT. By focusing on this under-investigated population, our study seeks to generate additional clinical evidence for a subgroup that constitutes the majority of breast cancer cases but frequently fails to achieve desirable pCR rates with chemotherapy. The results are expected to provide a foundation for future research and contribute to a better understanding of the biological mechanisms underlying treatment response in HER2-low breast cancer.
Methods
Study participants
This retrospective study included breast cancer patients aged 18 years and older who received neoadjuvant chemotherapy (NACT) between 2015 and 2025 at the Medical Oncology Clinics of Kütahya Evliya Çelebi Training and Research Hospital and Kütahya City Hospital. A total of 61 patients were identified through institutional oncology databases. Demographic and clinical data, including age, weight, comorbidities, and smoking history, were collected from medical records. The Eastern Cooperative Oncology Group (ECOG) performance status was recorded at baseline. Only patients who underwent surgery after completing NACT were included in the analysis.
Inclusion criteria were: (1) histologically confirmed locally advanced breast cancer (≥T2 or node-positive disease), (2) completion of the full planned neoadjuvant regimen at our center, and (3) availability of pathology results from the same institution.
Exclusion criteria included: incomplete chemotherapy for reasons unrelated to toxicity, prior malignancy, and surgery performed at another center.
Pathological assessment
Pathological analyses were performed in the institutional pathology department. Tumor grade, estrogen receptor (ER), progesterone receptor (PR), HER2, and Ki-67 index were determined using pre-treatment core biopsy specimens obtained before NACT. This approach was used to avoid potential variability in HER2 expression following systemic therapy. Tumor grade was categorized as Grade 1, 2, or 3. ER and PR expression were reported as percentages of positive tumor nuclei and grouped as <1%, 1–9%, and ≥10%.
HER2 status was assessed according to ASCO/CAP guidelines.
9
Immunohistochemistry (IHC) scoring was defined as:
0 = no staining or incomplete faint membrane staining in ≤10% of tumor cells; 1+ = incomplete faint membrane staining in >10%; 2+ = weak to moderate complete membrane staining in >10%; 3+ = strong complete membrane staining in >10%.
Cases with IHC 0 were defined as HER2-zero, and cases with IHC 1 + or 2 + and negative FISH as HER2-low. FISH testing was accepted from accredited external laboratories when required. HER2 negativity was defined as a dual-probe HER2/CEP17 ratio <2.0 with an average HER2 copy number <4.0 signals per cell. Pathologists assessing pCR were blinded to the patients’ HER2 status.
Radiological and clinical assessment
Baseline clinical stage was determined by physical examination and breast ultrasonography, supplemented by thoracic and abdominal CT or PET-CT when indicated. Treatment response was evaluated by comparative ultrasound or PET-CT at the end of NACT. Radiological response was classified as complete, partial, stable, or progressive disease.
Chemotherapy regimens
Patients received one of two standard NACT regimens based on clinical suitability. Regimen 1 (AC + Taxane): doxorubicin (60 mg/m2) plus cycloph osphamide (600 mg/m2) every 14–21 days for four cycles, followed by paclitaxel (80 mg/m2 weekly for 12 weeks or 175 mg/m2 every 3 weeks) or docetaxel (75 mg/m2 every 3 weeks for four cycles). Regimen 2 (TC): docetaxel (75 mg/m2) plus cyclophosphamide (600 mg/m2) every 3 weeks for four to six cycles. The distribution of regimens between groups was comparable, with no significant difference in pCR outcomes.
Definition of pathological complete response
pCR was defined according to standard criteria as the absence of residual invasive carcinoma in both the breast and axillary lymph nodes (ypT0/is ypN0) following NACT.
Data analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows, Version 25.0. Descriptive data are presented as means ± standard deviation, medians, or frequencies (%). Categorical variables were compared using the chi-square or Fisher's exact test, and continuous variables with the Mann–Whitney U test. Statistical significance was defined as a two-sided p-value < 0.05. Missing data were minimal (<5%) and handled by complete-case analysis. A post-hoc power analysis, based on the observed pCR rates (27.3% vs 7.1%; n = 61; α = 0.05, two-sided), indicated a statistical power of approximately 0.59 to detect the observed difference between groups.
Ethics statement
This study was approved by the Bezmialem Vakıf University Non-Interventional Clinical Research Ethics Committee (Approval No: E-54022451-050.04-206872), explicitly specifying the names of the participating centers. Additionally, administrative and educational planning board approvals were obtained from each center. As this was a retrospective study utilizing anonymized data, informed consent was not required in accordance with national regulations and institutional policy.
Results
General clinical characteristics
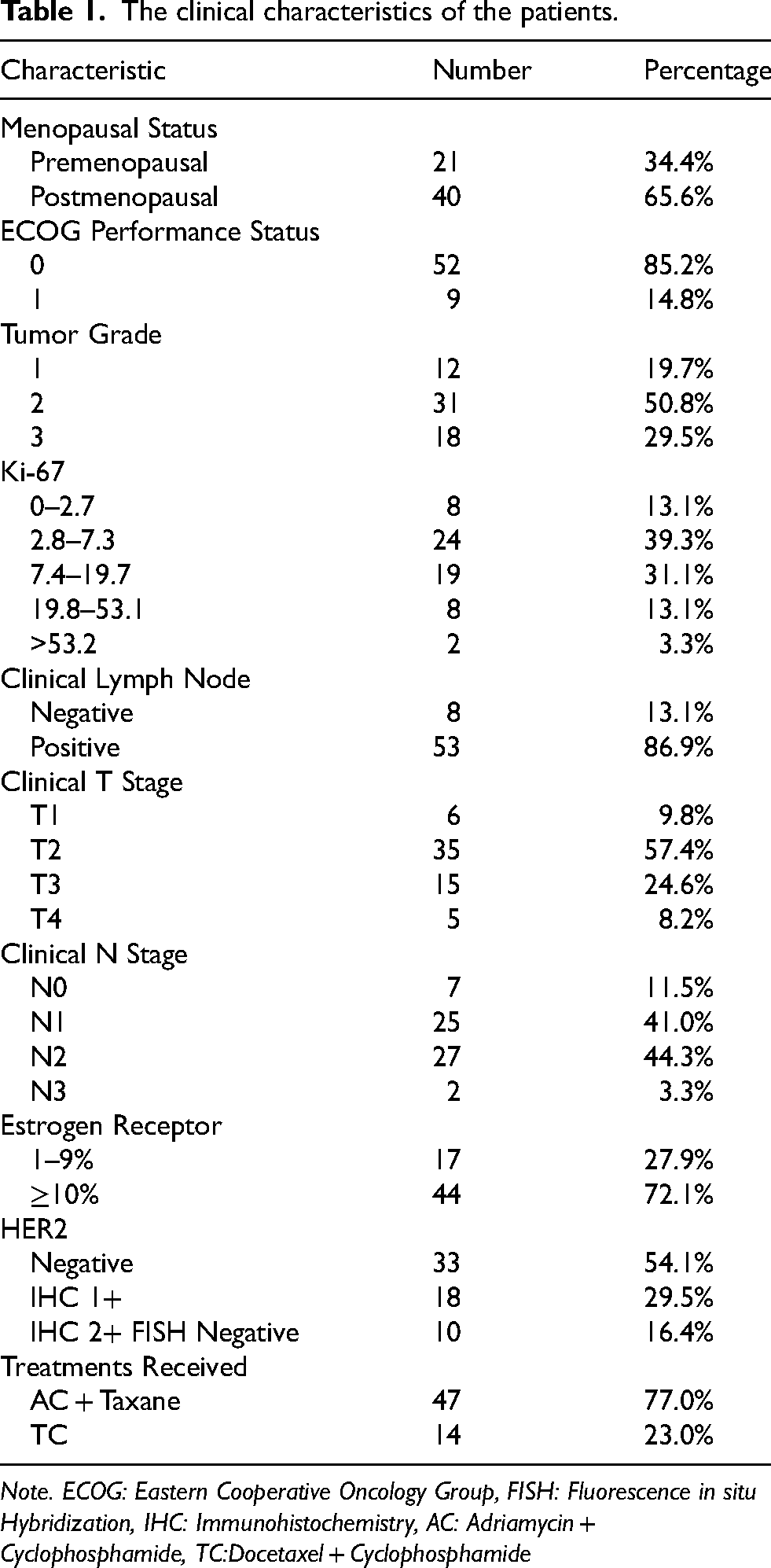
The study cohort consisted of 61 patients, including 21 premenopausal (34.4%) and 40 postmenopausal (65.6%) women. Most patients had good performance status, with ECOG 0 in 85.2% of cases. Tumor grade was predominantly Grade 2 (50.8%), followed by Grade 3 (29.5%) and Grade 1 (19.7%). The Ki-67 index was most commonly within the 2.8–7.3% range (39.3%). Clinical lymph node involvement was present in 53 patients (86.9%), and the majority of tumors were stage T2 (57.4%). ER positivity ≥10% was detected in 72.1% of patients, while PR positivity ≥10% was seen in 45.4%. Regarding chemotherapy regimens, 47 patients (77.0%) received the AC + Taxane protocol, and 14 (23.0%) received the TC regimen (Table 1). The distribution of treatment regimens between the HER2-low and HER2-zero groups was comparable (p = 0.81).
The clinical characteristics of the patients.
Comparison between HER2-zero and HER2-low groups
Table 2 presents the comparative characteristics of the two groups. The HER2-zero group showed a higher proportion of Grade 2 and 3 tumors, suggesting a slightly more aggressive histology, whereas the HER2-low group demonstrated a higher rate of ER and PR positivity (≥10%). Lymph node status and clinical T/N stages were similarly distributed between groups. Despite these differences, none of the baseline clinicopathological parameters showed statistical significance (all p > 0.05). Likewise, the distribution of neoadjuvant chemotherapy regimens was similar between groups, with 25 patients (75.8%) in the HER2-zero group and 22 (78.6%) in the HER2-low group receiving AC + Taxane, and 8 (24.2%) and 6 (21.4%) patients, respectively, treated with the TC regimen (p = 0.81).
Clinical characteristics of the HER2-negative and HER2-low groups.

Radiologic response
According to imaging assessments, 6 patients (18.2%) in the HER2-zero group and 4 (14.3%) in the HER2-low group achieved a complete radiologic response, whereas 23 (69.7%) and 17 (60.7%) demonstrated a partial response, respectively. Stable disease was observed in 4 (12.1%) HER2-zero and 7 (25.0%) HER2-low cases. The difference in radiologic response rates between groups was not statistically significant (p = 0.423) (Table 3).
Comparison of radiologic response rates by HER2 status.

Note. # : Pearson chi-square test, p < 0.05 considered statistically significant.
Pathological complete response
As shown in Table 4, the HER2-zero group achieved a pCR in 9 of 33 patients (27.3%, 95% CI 15.1–44.2), while only 2 of 28 patients (7.1%, 95% CI 2.0–22.6) in the HER2-low group reached pCR. This difference was statistically significant (p = 0.042), indicating a lower chemosensitivity in HER2-low tumors. A post-hoc power analysis based on these rates (α = 0.05, two-sided; n = 61) demonstrated a statistical power of approximately 0.59, confirming moderate sensitivity to detect the observed intergroup difference. These findings emphasize that even low-level HER2 expression may negatively influence pathological response to neoadjuvant chemotherapy in hormone receptor–positive breast cancer.
Comparison of pathological complete response rates by HER2 status.

Discussion
Our study demonstrated that the HER2-low positive group exhibited a proportionally lower grade and Ki-67 index compared to the HER2-zero group. Moreover, a higher proportion of tumors in the HER2-low group showed an ER positivity rate exceeding 10%. Consistent with these findings, existing literature, which indicates that HER2-low tumors are frequently hormone receptor-positive, and among HR-negative cases, tend to exhibit less aggressive features than HER2-zero tumors. 10 Another study has also demonstrated a correlation between HER2-low positivity and hormone receptor positivity, suggesting that identifying HER2-low positivity in post-neoadjuvant therapy results may offer valuable guidance for monitoring and therapeutic decision-making in certain patients. 11
Our study revealed that the HER2-low group exhibited statistically significantly lower pCR rates compared to the HER2-zero group. Because HER2 status was determined from pre-treatment biopsy specimens, our findings primarily reflect the
While there is evidence that HER2-low breast tumors display unique molecular characteristics, research has shown that the intrinsic subtypes of breast cancer are more strongly associated with HR status rather than HER2-low expression. 17 Specifically, HR-positive/HER2-low tumors predominantly exhibit luminal subtypes, similar to HR-positive/HER2-zero tumors. Conversely, HR-negative/HER2-low tumors are more often classified as the basal-like subtype. 18 These findings suggest that HER2-low breast cancer should be recognized as a heterogeneous group of tumors, with HR expression playing a primary role in determining their biological features, rather than being considered a distinct molecular entity. The European Society for Medical Oncology has also recommended that HER2-low positivity should not be treated as a separate group, but rather be classified based on HR status. 19 Furthermore, the efficacy of trastuzumab deruxtecan in HER2-low positive triple-negative breast cancer has been primarily attributed to the bystander effect of the drug, rather than HER2-low positivity indicating a distinct subtype. 20
A significant portion of breast cancers are HR-positive, and a substantial subset of these patients present with HER2-low tumors without metastasis, often being referred to clinics for neoadjuvant therapy.6,21 Unfortunately, as observed in our study, the treatment responses in this patient population can be suboptimal, making the decision to pursue NACT challenging.12,13 To enhance the efficacy, two potential mechanisms can be considered. Firstly, in this group where luminal features are predominant, the addition of endocrine agents or prioritizing endocrine therapy over chemotherapy could be explored. 10 Secondly, incorporating novel antibody-drug conjugates, particularly trastuzumab deruxtecan (T-DXd), which has demonstrated strong efficacy in metastatic settings, may offer new therapeutic opportunities in the neoadjuvant context. T-DXd, an antibody–drug conjugate composed of a humanized anti-HER2 antibody linked to a topoisomerase I inhibitor payload, showed remarkable clinical activity in HER2-low metastatic breast cancer in the DESTINY-Breast04 phase III trial, significantly improving both progression-free and overall survival compared with standard chemotherapy. 8 Furthermore, the TRIO-US B-12 TALENT phase II study explored the neoadjuvant use of T-DXd and reported encouraging response rates even in early-stage HR-positive, HER2-low tumors. 22 These findings suggest that ADCs targeting low HER2 expression could broaden treatment options beyond the metastatic setting and justify further evaluation in neoadjuvant trials. However, while these tumors display strong luminal characteristics, emerging evidence indicates that HER2-low status may be associated with increased endocrine therapy resistance compared to HER2-zero tumors. 23 In this context, novel antibody-drug conjugates represent a promising area for future research and may help overcome the limited efficacy observed with conventional chemotherapy or endocrine therapy in this substantial patient population.
Limitations
This single-center retrospective study was conducted with a cohort of 61 patients. The study did not include advanced tumor biology analyses, such as Oncotype DX or PAM50 genetic testing, that could have provided insights into the tumor characteristics and enabled comparisons of the study outcomes. Additionally, the long-term follow-up results, including disease-free survival and overall survival, were not evaluated to assess any potential differences in survival expectations between the HER2-low and HER2-zero groups. The analysis was limited to examining the pCR rate only. Because the number of complete-response events was limited (n = 11), multivariable adjustment was not performed to avoid overfitting; instead, effect sizes and confidence intervals were reported, and this limitation is acknowledged. Furthermore, the post-hoc power analysis (≈ 0.59) indicated moderate sensitivity to detect intergroup differences, underscoring the need for validation in larger multicenter cohorts.
Conclusion
The efficacy of trastuzumab deruxtecan (T-DXd) observed in the metastatic HER2-low population has generated considerable interest among clinicians; however, many uncertainties remain regarding its role in earlier disease stages. The potential impact of HER2-low status in the non-metastatic, hormone receptor–positive setting is of particular clinical relevance. Although available data, including the findings from our study, are limited, they suggest that HER2-low expression may negatively influence chemosensitivity in patients receiving neoadjuvant therapy. Nevertheless, the current evidence does not justify altering clinical practice or discouraging the use of NACT in this population. Given the pressing need for more effective treatment options, ongoing and future clinical trials investigating antibody–drug conjugates are essential to improve therapeutic outcomes and to address the remaining challenges in managing HER2-low breast cancer.
Footnotes
Acknowledgements
Not applicable.
Ethics committee approval
This study was approved by the Bezmialem Vakıf University Non-Interventional Clinical Research Ethics Committee (Approval No: E-54022451-050.04-206872). We confirm that the ethics approval obtained from Bezmialem Vakıf University covers data collection from all participating centers, including Kütahya Evliya Çelebi Training and Research Hospital and Kütahya City Hospital. Additionally, administrative and educational planning board approvals were obtained from each center.
Informed consent
Informed consent form is not required as the ethics committee stated that approval is not required for retrospective studies.
Authors’ contributions
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability and material
All data from the study have been stored in a database and can be obtained from Dr Mustafa Ersoy upon request.
