Abstract
Introduction
The Clinical Trials Update is a regular feature devoted to highlighting ongoing and recently completed clinical trials in Huntington's disease (HD). Clinical trials previously reviewed in this section are listed in Table 1. For current and completed studies discussed here, endorsement status by the European Huntington's Disease Network (EHDN) is now also indicated.
Clinical trials previously reviewed by the Huntington's disease clinical trials update.

IONIS-HTTRx, RG6042 and tominersen refer to the same molecule.
VX15/2503 and pepinemab refer to the same molecule.
AAVrh10.CAG.hCYP46A1, BV-101 and AB-1001 refer to the same molecule.
In this edition, we expand on the launch of the phase II/III clinical trial of SKY-0515 (NCT06873334) 13 (FALCON-HD) from Skyhawk Therapeutics and the phase I/II clinical trial of SPK-10001 (NCT06826612) 14 from Spark Therapeutics. We also report positive topline data from uniQure's phase I/II clinical trial of AMT-130 (NCT04120493; NCT05243017)15,16 after 36 months of follow-up. Further updates include recent developments in Roche's tominersen programme within GENERATION HD2 (NCT05686551), 17 progress with votoplam (PTC518) in PIVOT-HD (NCT05358717; NCT06254482)18,19 by PTC Therapeutics and developments in the collaborative PTC Therapeutics/Novartis programme. We additionally discuss regulatory developments regarding pridopidine following the negative PROOF-HD (NCT04556656) 20 study. Finally, we provide an updated listing of all registered and ongoing clinical trials in Huntington's disease.
We tabulate all currently registered and ongoing clinical trials in Tables 2–4. For further details on the methodology used, please refer to the first edition of this series. 1
Pharmacological clinical trials registered at the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) for people with Huntington's disease (HD) since the first edition of the “Huntington's Disease Clinical Trials Corner”.

AAV: adeno-associated virus; AMP: adenosine monophosphate; AMPK: AMP-activated protein kinase; ASO: antisense oligonucleotide; HD: Huntington's disease; HD-ISS: Huntington's Disease Integrated Staging System; HTT: huntingtin; MAPK: mitogen-activated protein kinase; mHTT: mutant huntingtin; miRNA: microRNA; MRI: magnetic resonance imaging; mRNA: messenger ribonucleic acid; NAC: N-acetylcysteine; NAD+: nicotinamide adenine dinucleotide (oxidised form); NMDA: N-methyl-D-aspartate; N/S: not specified; PD: Parkinson's disease; PMS1: post-meiotic segregation 1; PPARα: peroxisome proliferator-activated receptor alpha; RNA: ribonucleic acid; SCA1: spinocerebellar ataxia 1; SCA3: spinocerebellar ataxia 3; TD: tardive dyskinesia; TEAEs: treatment-emergent adverse events; TFC: Total Functional Capacity; UHDRS: Unified Huntington's Disease Rating Scale; VMAT2: vesicular monoamine transporter 2. Note: IONIS-HTTRx, ISIS 443139, RG6042 and tominersen refer to the same molecule. New trials added since the last Clinical Trials Update are indicated by *. Pharmacological trials terminated are indicated by †.
Invasive non-pharmacological clinical trials registered at the WHO ICTRP for people with HD since the first edition of the “Huntington's Disease Clinical Trials Corner”.

AD: Alzheimer's disease; CBD: corticobasal degeneration; DBS: deep brain stimulation; ET: essential tremor; GP: globus pallidus; GPi: globus pallidus internus; HD: Huntington's disease; HT: Holmes tremor; ICTRP: International Clinical Trials Registry Platform; MNC: mononuclear cells; MR: magnetic resonance; MS: multiple sclerosis; N/S: not specified; RNA: ribonucleic acid; PD: Parkinson's disease; TD: tardive dyskinesia; WD: Wilson's disease; WHO: World Health Organization.
Non-invasive non-pharmacological clinical trials registered at the WHO ICTRP for people with HD since the first edition of the “Huntington's Disease Clinical Trials Corner”.
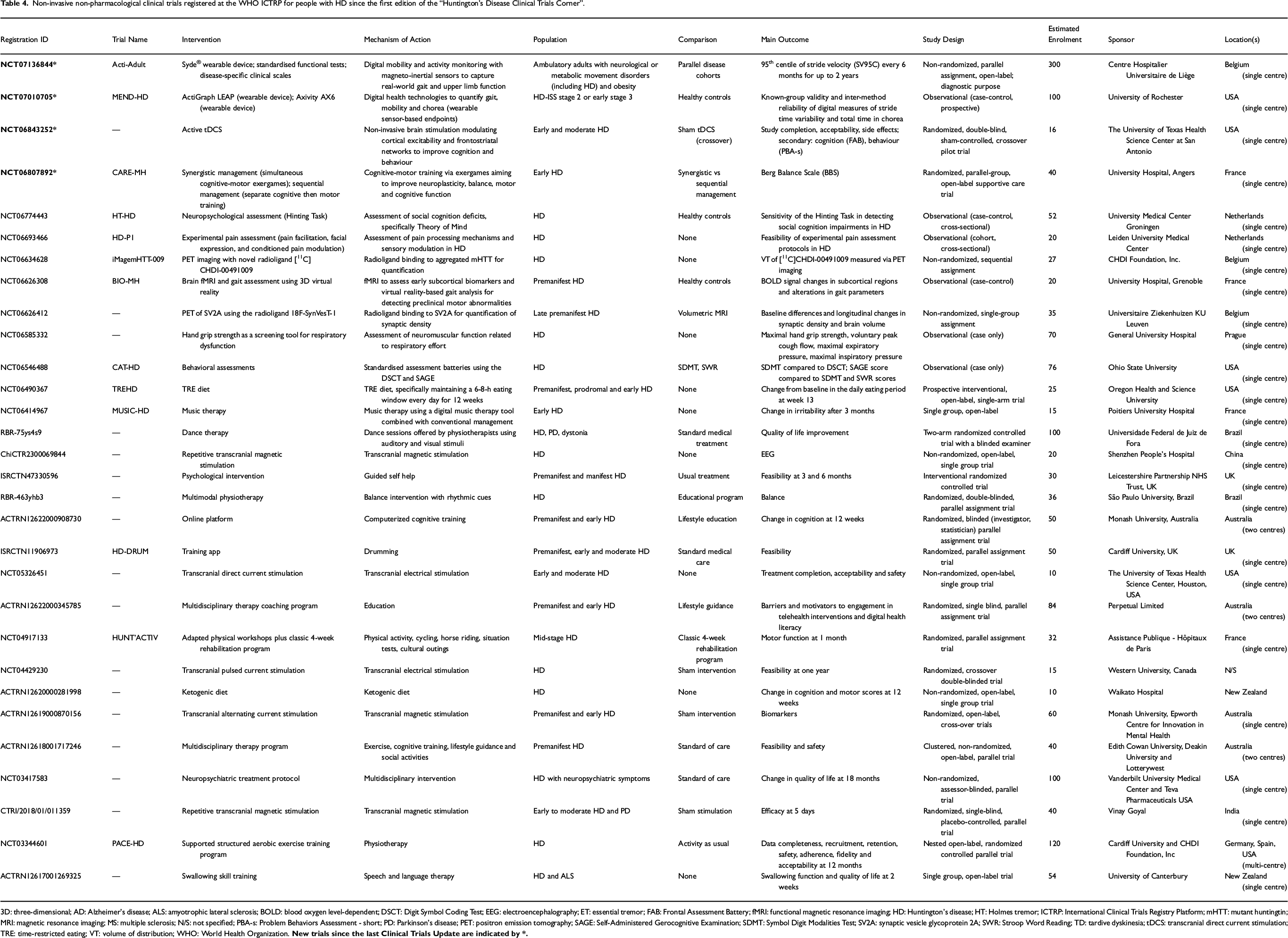
3D: three-dimensional; AD: Alzheimer's disease; ALS: amyotrophic lateral sclerosis; BOLD: blood oxygen level-dependent; DSCT: Digit Symbol Coding Test; EEG: electroencephalography; ET: essential tremor; FAB: Frontal Assessment Battery; fMRI: functional magnetic resonance imaging; HD: Huntington's disease; HT: Holmes tremor; ICTRP: International Clinical Trials Registry Platform; mHTT: mutant huntingtin; MRI: magnetic resonance imaging; MS: multiple sclerosis; N/S: not specified; PBA-s: Problem Behaviors Assessment - short; PD: Parkinson's disease; PET: positron emission tomography; SAGE: Self-Administered Gerocognitive Examination; SDMT: Symbol Digit Modalities Test; SV2A: synaptic vesicle glycoprotein 2A; SWR: Stroop Word Reading; TD: tardive dyskinesia; tDCS: transcranial direct current stimulation; TRE: time-restricted eating; VT: volume of distribution; WHO: World Health Organization.
If you would like to draw attention to specific trials, please feel free to email us at: mena.farag@ucl.ac.uk and e.wild@ucl.ac.uk.
Ongoing clinical trials
A list of all registered clinical trials is given in Tables 2–4.
SKY-0515 (NCT06873334) 13
Primary outcome measures are change from baseline to 12 months in blood mHTT protein levels, MRI-derived brain volumes and UHDRS scores. Other outcome measures include changes in other HD-related biomarkers and long-term safety of SKY-0515.
On 20th June 2025, Skyhawk Therapeutics announced that the first patient was dosed in the phase II/III FALCON-HD study (NCT06873334) 13 in Adelaide, Australia, 24 marking the initiation of the registrational trial programme across multiple sites in Australia and New Zealand.
SKY-0515 is designed to reduce both HTT and PMS1 proteins, the latter implicated in somatic CAG repeat expansion, a mechanistic process increasingly recognised as a key driver of neuronal loss in HD.25,26 Genome-wide association and mismatch repair studies indicate that targeting PMS1 and other DNA repair genes could help prevent progressive neuronal pathogenesis in striatal and cortical regions.27,28 This important phase II/III clinical trial of SKY-0515 will assess whether HTT and PMS1 lowering can offer greater therapeutic benefit than HTT reduction alone in early-stage HD.
SPK-10001 (NCT06826612) 14
Primary outcome measures are safety and tolerability, measured by the incidence and severity of treatment-emergent adverse events (TEAEs) over a five-year follow-up period, and change from baseline in UHDRS TFC score at 24 months. Secondary outcome measures include changes from baseline in motor progression assessed by digital motor testing at 12, 18 and 24 months; changes in composite UHDRS (cUHDRS) scores at 12, 18 and 24 months; and changes in UHDRS TFC scores at 12 and 18 months.
These findings and the update from Spark Therapeutics and Roche represent an important step in the development and progress of gene therapy for HD, particularly in light of the news below from the first gene therapy to be tested in HD patients. Early preclinical data support target engagement and tolerability of SPK-10001; however, safety, efficacy and clinical benefit in humans remain to be established. Long-term monitoring will be essential to determine the durability of any therapeutic effects, ongoing safety and potential functional impact as results from the ongoing phase I/II clinical trial become available.
Breaking news
In this section we provide brief updates about ongoing or recently terminated clinical trials.
The
The
The
On 25th July 2025, Prilenia and Ferrer announced that the European Medicines Agency's Committee for Medicinal Products for Human Use had recommended refusal of marketing authorisation for pridopidine in HD.
38
This decision follows the outcome of the phase III
Footnotes
Acknowledgements
The authors have no acknowledgments to report.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: SJT receives research grant funding from the CHDI Foundation, Vertex Pharmaceuticals, the UK Medical Research Council, the Wellcome Trust and the UK Dementia Research Institute that receives its funding from DRI Ltd., funded by the UK MRC, Alzheimer's Society and Alzheimer's Research UK. EJW is supported by CHDI Foundation, Inc. EJW reports grants from CHDI Foundation and F. Hoffmann-La Roche Ltd.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MF was a sub-investigator in the ALN-HTT02 (NCT06585449), GENERATION HD2 (NCT05686551), PTC518 (NCT05358717; NCT06254482) and uniQure AMT-130 (NCT05243017) clinical trials.
SJT has undertaken consultancy services for Annexon, Alphasights, Alnylam Pharmaceuticals Inc., Atalanta Pharmaceuticals (SAB), F. Hoffmann-La Roche Ltd/Genentech, Guidepoint, Horama, Locanobio, LoQus23 Therapeutics Ltd (SAB), Novartis Pharma, PTC Therapeutics, Sanofi, Spark Therapeutics, Takeda Pharmaceuticals Ltd, Triplet Therapeutics (SAB), University College Irvine and Vertex Pharmaceuticals Incorporated. All honoraria for these consultancies were paid through the offices of UCL Consultants Ltd, a wholly owned subsidiary of University College London. SJT has a patent Application number 2105484.6 on the FAN1-MLH1 interaction and structural analogues licensed to Adrestia Therapeutics. SJT was an investigator in the ALN-HTT02 (NCT06585449), IONIS HTTRx (NCT02519036), IONIS HTTRx OLE (NCT03342053), GENERATION HD1 (NCT03761849), GENERATION HD2 (NCT05686551), Roche Natural History Study (NCT03664804), Roche GEN-EXTEND (NCT03842969), PTC518 (NCT05358717; NCT06254482), SHIELD-HD (NCT04406636) and uniQure AMT-130 (NCT05243017) clinical trials.
EJW has undertaken consultancy/advisory board work with Hoffman La Roche Ltd, Triplet Therapeutics, Takeda, Vico Therapeutics, Voyager, Huntington Study Group, Teitur Trophics, EcoR1 Capital, PTC Therapeutics, Alnylam, Annexon Biosciences, Remix Therapeutics and Skyhawk Therapeutics. All honoraria for these consultancies were paid through the offices of UCL Consultants Ltd, a wholly owned subsidiary of University College London. EJW was an investigator in the ALN-HTT02 (NCT06585449), Amaryllis (NCT02197130), LEGATO-HD (NCT02215616), IONIS HTTRx (NCT02519036), IONIS HTTRx OLE (NCT03342053), GENERATION HD1 (NCT03761849), GENERATION HD2 (NCT05686551), Roche Natural History Study (NCT03664804), Roche GEN-EXTEND (NCT03842969), Roche GEN-PEAK (NCT04000594), PTC518 (NCT05358717; NCT06254482), uniQure AMT-130 (NCT05243017) and VIBRANT-HD (NCT05111249) clinical trials.
SJT and EJW are Editorial Board Members of this journal but were not involved in the peer review process of this article nor had access to any information regarding its peer review.
Data availability
Data sharing not applicable as no datasets generated and/or analysed for this study.
