Abstract
Introduction
Cancer is the second leading cause of death worldwide, with nearly 20 million new cancer cases and more than 9.7 million cancer-related deaths occurring annually. 1 Moreover, cancer incidence and mortality are still increasing annually, and the number of new cancer cases is expected to reach 35 million by 2050, 2 posing a serious threat to human life and health. For different types of cancer, clinical management strategies involving various treatments, such as surgery, radiotherapy, interventional therapy, targeted therapy, immunotherapy, and traditional Chinese medicine (TCM), have been developed and achieved positive clinical therapeutic effects.
Compared to other treatments, TCM has been shown to have therapeutic effects such as improving the physical condition, immunity, and pain of patients with cancer, as well as reducing the toxic side effects of drugs,
3
significantly improving patients’ quality of life and playing a unique role in cancer treatment. Among TCMs with antitumor effects,
Many studies have examined the antitumor components of
This review aims to systematically summarize current research on GLPs, covering their structural characterization, anti-tumor molecular mechanisms, and immunomodulatory activities, and to critically evaluate their clinical application prospects. By integrating existing evidence, this review aims to clarify the therapeutic potential of GLPs as natural anticancer agents and to facilitate their further development toward standardized, evidence-based, and globally applicable cancer therapeutics.
Structural Characteristics of GLPs
GLPs are an active ingredient extracted from
Overall, the chemical composition of GLPs is complex, with varying molecular weights. Generally, their biological activities increase with molecular weights. To a certain extent, the numbers of branched chains, the number of monosaccharides, and biological activity increase with the main chain length of GLPs. 13 Systematic studies have examined the structures of more than 200 GLPs, of which more than 20 have been identified (Table 1).
Biological Activities of Structurally Diverse GLPs Obtained via Different Extraction Methods

Antitumor Effects of GLPs
Due to their inherently low cytotoxicity, it was initially proposed to use GLPs as an immunomodulator to facilitate clinical anti-tumor therapy. However, as research progressed, they were found to exert certain inhibitory effects on various cancers, including osteosarcoma, melanoma, breast, gastric, and hepatocellular carcinoma. 13
For instance, GLPs have been reported to effectively inhibit the proliferation, migration, and invasion of human osteosarcoma MG63 and U2-OS cells while simultaneously inducing apoptosis, indicating their potent cytotoxic and anti-metastatic effects in osteosarcoma models. In another study, co-incubation of MDA-MB-231 (triple-negative breast cancer) and B16-F10 (melanoma) cells with varying concentrations of GLPs significantly suppressed tumor cell proliferation. At concentrations above 250 µg/mL, GLPs significantly reducing the cell viability of both cancer types in a time and concentration-dependent manner. These findings suggest that GLPs can be used in the treatment of melanoma and triple-negative breast cancer. 31 A Chinese study further explored the inhibitory effect of GLPs on gastric cancer, using human gastric cancer cell lines MKN28, AGS, NCI-N87, and GES-1. It confirmed that GLPs could significantly reduce the viability of gastric cancer cells and induce apoptosis. The half-maximal inhibitory concentration (IC50) is 5.36 ± 2.58 mg/ml for a 48 h incubation. 32
Additionally, another study evaluated the effect of GLPs in a murine hepatocellular carcinoma (HCC) model in vivo. In this study, the mouse hepatoma cell line Hepa1-6 was used to establish a transplanted tumor model, and different doses of GLP (100 and 200 mg/kg) were administered by gavage daily for 14 days. The results showed that GLP could significantly inhibit the growth of Hepa1-6 xenografts in a dose-dependent manner, with the highest inhibition rate of 65%.
33
This
Collectively, these findings indicate that GLPs possess significant anti-tumor activity across various solid malignancies. This provides a strong scientific rationale for their further development and clinical application in cancer therapy.
Anti-Tumor Mechanisms of GLPs
GLPs exert potent anti-tumor effects through multiple mechanisms, including inhibition of tumor cell proliferation, induction of apoptosis, and suppression of invasion and metastasis. Moreover, GLPs modulate key intracellular signaling cascades via multi-target molecular interactions, ultimately disrupting tumor growth and survival. Overall, the anti-cancer actions of GLPs are mediated through several major signaling pathways, as summarized below.
Apoptotic Pathway
The key molecular mechanisms underlying the anti-tumor effects of GLPs are summarized in Figure 1. GLPs have been shown to regulate multiple intracellular signaling cascades that are crucial for tumor cell survival and apoptosis. Primarily, GLPs activate both the

Molecular mechanisms of GLP-induced apoptosis in tumor cells.
Caspase-Dependent Signaling
Caspases are cysteine-dependent aspartate-specific proteases that act as central executors of apoptosis.
34
Multiple studies have confirmed the involvement of caspase signaling in GLP-induced tumor cell death. Zhong et al
35
found that GLPs,
Mitochondrial Pathway
Mitochondria are the “switch” of apoptosis regulation in tumor cells, and their abnormal functions are closely related to tumorigenesis and treatment resistance. The mitochondrial pathway's involvement in anti-tumor activity is related to the release of cytochrome C (CYCS), activation of caspase cascade, regulation of BCL-2 family proteins, and open of mitochondrial membrane permeability transition pore (MPTP). GLPs have been shown to impair mitochondrial membrane potential, promoting the release of CYCS, activation of CASP9 and CASP3, and subsequent apoptotic vesicle formation. For example, Shang et al 38 found that GLPs significantly inhibited breast cancer growth in a dose-dependent manner. GLPs disrupted the mitochondrial membrane potential and, increased CYCS levels. Gao et al 39 found that selenium-containing GLPs (Se-GLPs) could promote apoptosis and inhibit proliferation in breast cancer cells by disrupting mitochondrial membrane potential, increasing the CYCS level and activating CASP3, CASP9, and poly ADP-ribose polymerases (PARP). These studies support that regulation of mitochondrial integrity and permeability transition is a major route by which GLPs trigger apoptosis.
cAMP/PKA Signaling Pathway
The cyclic adenosine monophosphate (cAMP) / protein kinase A (PKA) signaling pathway is an important intracellular second messenger system widely involved in proliferation, metabolic regulation, and stress response. Several studies have confirmed that GLPs promote tumor apoptosis through the cAMP/PKA signaling pathway. Xu et al 40 found that GLPs significantly increased cAMP, PKA and adenylate cyclase activities in tumor upregulating the protein expression of PKA and downregulating the protein expression of protein kinase C. Zhang et al 27 demonstrated that PSG-1 significantly increased cAMP and PKA activities. It significantly increased PKA protein levels and decreased PKC and intracellular Ca2+ levels, the activation of cAMP/PKA signaling pathway, and inactivation of the Ca2+/PKC signaling pathway, by GLPs lead to inhibition of apoptosis and tumor growth. These results suggest that GLPs promote tumor cell death partially by shifting the balance toward cAMP/PKA pro-apoptotic signaling.
Fas Cell Surface Death Receptor
The Fas signaling pathway represents a major extrinsic apoptotic mechanism, initiated by death receptors located on the cell surface and closely associated with immune regulation and tumor immune evasion. Upon Fas ligand (FasL) binding, Fas receptors recruit Fas-associated death domain protein (FADD), leading to the assembly of the death-inducing signaling complex (DISC). This complex subsequently activates caspase-8, triggering the downstream caspase cascade and ultimately resulting in apoptosis.
Several studies have confirmed that GLPs promote apoptosis by triggering the Fas-mediated extrinsic apoptotic pathway. Chen et al
41
demonstrated that treatment of HepG2 cells with
GLPs trigger programed cell death through multiple apoptotic pathways: (A) Fas-mediated extrinsic pathway: GLPs enhance Fas receptor activation on the tumor cell surface, leading to recruitment and activation of caspase-8, thus initiating the apoptotic cascade. (B) Mitochondrial (intrinsic) pathway: GLPs induce intracellular stress and mitochondrial outer membrane permeabilization (MOMP), resulting in cytochrome c release and apoptosome formation, which activates caspase-9. (C) Caspase execution pathway: Both extrinsic and intrinsic apoptotic signals converge on the caspase-3-dependent execution cascade, leading to the cleavage of critical cellular substrates and apoptotic cell death. (D) cAMP/PKA signaling modulation: GLPs regulate cAMP/PKA signaling, which interacts with both apoptotic pathways to fine-tune the magnitude of apoptosis and enhance tumor-specific cytotoxicity.
Antioxidative Protection Against DNA Damage
Excessive accumulation of reactive oxygen species (ROS) and other free radicals derived from cellular metabolism, UV exposure, environmental toxins, and chronic inflammation can induce oxidative DNA damage. Such damage, including DNA strand breaks, oxidation, and replication errors, lead to genomic instability and irreversible mutations. Over time, these alterations may activate oncogenes or disable tumor suppressor genes, thereby promoting malignant transformation, uncontrolled cell proliferation, and cancer development. Therefore, enhanced antioxidative defense represents a critical mechanism for preventing tumor initiation and progression.
Multiple studies have demonstrated that GLPs exert substantial antioxidant and DNA-protective effects. Seweryn et al 45 demonstrated that GLPs promoted proliferation, differentiation, activation, while regulating nitric oxide (NO) production, In addition, GLPs inhibited the aging of endothelial cells, and decrease plasma levels of superoxide dismutase (SOD) and malondialdehyde (MDA), and indirectly scavenge oxygen free radicals and increased glutathione peroxidase (GPX) levels. Similarly, Yu et al 46 showed that GLPs significantly increased the activities of major antioxidant enzymes including the antioxidant enzyme activities of SOD, GPX and catalase (CAT) and lactate dehydrogenase (LDH). As a results, the GLPs enhance the body's capacity to neutralize excessive ROS and ameliorate oxidative stress. Further studies revealed that GLPs maintain DNA integrity by scavenging hydroxyl radicals, preventing oxidative base modification such as 8-OHdG formation, and supporting DNA repair pathways, ultimately reducing mutation accumulation in tumor-prone tissues. These findings indicate that the reduction of oxidative damage may serve as a vital upstream mechanism through which GLPs prevent carcinogenesis and suppress tumor progression47, 48
Collectively, GLPs enhance endogenous antioxidant defenses, preserve genomic stability, and mitigate ROS-induced DNA lesions, thereby contributing significantly to their anti-tumor pharmacological effects.
Immunomodulatory Mechanisms
Cancer immunotherapy enhances anti-tumor responses by promoting tumor antigenrecognition, restoring immune surveillance, and remodeling the immunosuppressive tumor microenvironment. As a natural bioactive macromolecule, GLPs have been widely recognized as potent immunomodulators capable of regulating both innate and adaptive immune responses.
GLPs exert broad immunoregulatory effects by modulating the proliferation, activation, and effector functions of diverse immune cells. In addition, GLPs stimulate the secretion of multiple cytokines, chemokines, and growth factors, thereby enhancing immune cell differentiation and cytotoxic activity while promoting antigen presentation and tumor clearance. Through these coordinated immune responses, GLPs contribute to both innate immune activation and adaptive immune priming, ultimately strengthening systemic anti-tumor immunity and suppressing tumor progression 23 (Figure 2).

Overview of the immunomodulatory effects of GLPs in anti-tumor immunity. (A) T cell immune responses: GLPs enhance antigen uptake, processing, and presentation by antigen-presenting cells, thereby promoting T-cell priming. They also provide co-stimulatory signaling and stimulate cytokine production (eg, IL-12), supporting the activation and clonal expansion of naive T cells. Activated cytotoxic T lymphocytes (CTLs) subsequently migrate into tumor tissues, recognize tumor-associated antigens, and induce cancer cell death. (B) B cells immune responses: GLPs activate B cells following antigen recognition through the B-cell receptor with assistance from T cells. Activated B cells proliferate and differentiate into antibody-secreting plasma cells. Tumor-specific antibodies contribute to anti-tumor immunity through multiple effector mechanisms. (C) TLR4-p38 MAPK/NF-κB signaling pathway in macrophages: GLPs bind to TLR4 on macrophages, leading to simultaneous activation of p38 MAPK and NF-κB signaling cascades. These pathways synergistically induce transcription and translation of TNF-α, resulting in increased synthesis and secretion of TNF-α and subsequent enhancement of macrophage-mediated tumoricidal activity. The accompanying Table 2 summarizes their key mechanisms of action-including apoptotic pathway, antioxidative protection against DNA damage and exert the immune function, along with their associated molecular targets and signaling pathway.
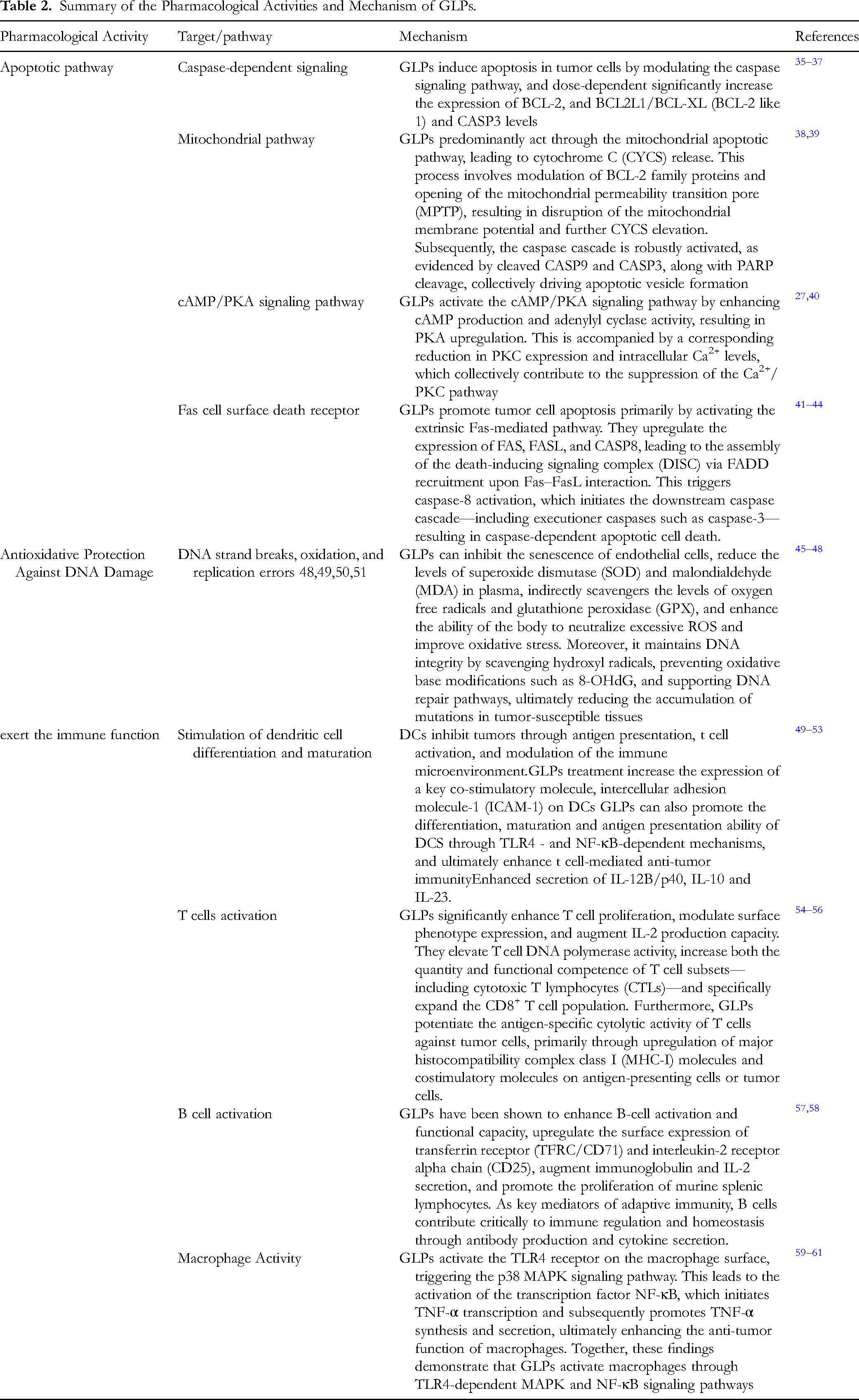
Summary of the Pharmacological Activities and Mechanism of GLPs.
Stimulation of Dendritic Cell Differentiation and Maturation
Dendritic cells (DCs) are the most potent professional antigen-presenting cells and serve as a critical bridge between innate and adaptive immunity. DCs combat tumors through antigen presentation, T-cell activation, and regulation of the immune microenvironment. Chen et al 49 showed that GLPs, a biologically active β-glucan, could stimulate monocyte-derived DC maturation and induce the differentiation of monocytic leukemia cells into functional DCs with enhanced immunostimulatory activity. Lin et al 50 further showed that GLPs treatment altered the transcriptional profile of human DCs and promoted a Th1-biased immune response in BALB/c mice, suggesting improvements in anti-tumor immunity. Moreover, Lin et al 51 found that treatment with PS-G, a branched (1→6)-β-D-glucan fraction of GLPs, increased the cell surface expression of key costimulatory molecules on DCs including CD8, CD86, CD83, CD40, intercellular adhesion molecule 1(ICAM1) /CD54, and human leukocyte antigen (HLA-DR), as well as increased the levels of the IL-12/p70, its subunits IL12A/p35 and IL12B/p40, as well as IL-10 production. In addition, PS-G also enhanced T-cell activation and promoted elevated T-cell release of IFN-γ and IL-10 while suppressing endocytic ability, a hallmark of DC maturation. Mechanistic investigations indicated that Toll-like receptor 4 (TLR4), an essential pattern recognition receptor, mediates these immunoregulatory effects, as TLR4 blockade attenuated PS-G-induced IL-12B/p40 and IL-10 expression, confirming the involvement of TLR4-dependent signaling. 52 Lin et al 53 further reported that GLP-derived recombinant LZ-8 enhanced DC maturation by increasing cell surface expression of CD80, CD86, CD83, and HLA-DR, accompanied by increased secretion of IL-12B/p40, IL-10, and IL-23. rLZ-8 treatment also activated inhibitor of κB kinase (IKK) and nuclear factor-κB (NF-κB), and promoted mitogen-activated protein kinase (MAPK) phosphorylation, indicating the activation of key innate immune signaling pathways. Collectively, these findings demonstrate that GLPs can promote DC differentiation, maturation, and antigen-presenting capacity through TLR4 and NF-κB-dependent mechanisms, ultimately enhancing T-cell-mediated anti-tumor immunity.
T Cells Activation
T cells play a central role in anti-tumor immunity by recognizing and eliminating malignant cells. however, tumors evade immune attack through various mechanisms, and immunotherapy enhance their activity by reducing T cell inhibition or enhancing T cell function. Li et al 54 experimentally confirmed that moderate amounts of GLPs could significantly increase T cells’ proliferation, surface phenotypic expression and ability to induce IL-2, and enhance the activity of T cell, DNA polymerase and increase the number and function of T cell subclasses and functions. Zhou et al 55 demonstrated that GLPs could enhance the killing effect of T cells on tumor cells by inducing major histocompatibility complex class I molecules and co-stimulatory molecules. Shao et al 56 demonstrated that GLPs could enhance cytotoxic T cell (CTL) activity, increase the number of CD8 +T cells, activate and improve tumor-cell-specific killing, enhancing the immunocompetence of T cells in tumor-bearing mice. These findings collectively demonstrate that GLPs enhance anti-tumor immunity by promoting T-cell activation, proliferation, and effector function, making them promising immunomodulatory agents for cancer therapy.
B Cell Activation
B cells contribute to anti-tumor immunity by producing antibodies and cytokines, regulating immune responses, and maintaining immune homeostasis. Multiple studies have shown that GLPs can enhance B-cell activation and function. Gao et al
57
investigated two groups of water-soluble GLPs (GLPD1 and GLPD2) extracted from the seeds of a
Macrophage Activity
Macrophages are essential effector cells of the innate immune system that contribute to anti-tumor immunity through direct tumor cell killing, phagocytosis, and activation of adaptive immune responses. Multiple studies have demonstrated that GLPs can enhance macrophage activation and effector function. Guo et al 59 demonstrated that GLPs could activate the immune receptor TLR4, thereby activating macrophages through the TLR4/NF-κB pathway. Zhang et al 60 demonstrated that GLPs acted on TLR4 on macrophage membranes, thereby activating the p38 MAPK signaling pathway. TLR4 activates the p38 MAPK signaling pathway, which activates the transcription factor NF-Kβ, initiating the transcription of TNF/TNF-α. Ultimately, TLR4 increases the synthesis and secretion of TNF-α, and improves the anti-tumor activity of macrophages. Lu et al 61 demonstrated that GLPs significantly enhanced multiple functions of peritoneal macrophages, including viability, phagocytic activity, the production of NO and TNF-α, as well as the cytotoxicity against B16F10-CS melanoma cells. In addition, GLPs could completely or partially inhibit TNF-α activity. Collectively, these findings indicate that GLPs enhance anti-tumor immunity by activating macrophages through TLR4-dependent MAPK and NF-κB signaling pathways, strengthening their roles in tumor cell clearance and immune regulation.
Emerging Research Fields of GLPs
With the rapid development of modern analytical and computational technologies, research on
Multi-omics technologies, including genomics, transcriptomics, proteomics, and metabolomics, provide systematic insights into GLP biosynthesis and regulatory networks. Xie et al
62
identified key genes associated with fungal growth, secondary metabolite biosynthesis, and signal transduction, laying a foundation for elucidating the molecular basis of
Structural characteristics are crucial determinants of GLP biological activity. Du et al 66 reported that molecular weight, branching degree, glycosidic linkages, and helical conformation influence solubility, bioavailability, and immune response. Liu et al 67 and Wang et al 68 showed that raw materials and extraction methods lead to variations in monosaccharide composition and structural complexity, resulting in differences in therapeutic performance. Tang et al 69 found that ultrasonic and enzymatic treatments enhance structural features contributing to antioxidant effects. Deng et al 70 identified β-(1→3,1→6)-glucan as a key motif for immune activation, while Fu et al 71 demonstrated that chain length and branching frequency significantly impact biological potency. These findings highlight the necessity of precise structural characterization and standardization for consistent clinical efficacy.
Molecular docking also contributes to mechanistic understanding at the protein, polysaccharide interaction level. Shen et al 72 showed that differential binding affinities of sulfated and non-sulfated GLPs to human serum albumin (HSA) may influence stability and transport in vivo. Additionally, Ignat et al 73 used Hi-C–assisted genome assembly to identify genes associated with GLP biosynthesis, providing a basis for metabolic engineering and improved druggability of GLP-derived products.
Anti-Tumor Clinical Applications of GLPs
GLPs have been widely applied in clinical oncology as adjuvant therapeutic agents due to their potent immunomodulatory effects and low toxicity. When used in combination with radiotherapy or chemotherapy, GLPs can alleviate treatment-related adverse effects, reduce the required dosage of cytotoxic drugs, and mitigate myelosuppression, thereby improving treatment tolerance and patient quality of life. Studies have demonstrated that GLP-based interventions can synergize with conventional tumor therapies to enhance therapeutic efficacy while minimizing systemic toxicity.
In China, ZIZHI polysaccharide tablets were approved in 2010 as an adjuvant treatment for leukopenia and hematopoietic damage caused by cancer chemotherapy and radiotherapy.
74
In addition, several fungal polysaccharides, including those derived from
Despite their growing clinical adoption, current GLP preparations also exhibit limitations, such as insufficient structure–activity relationship data, inconsistency in extraction and purification methods, and variability in clinical efficacy due to heterogeneous chemical compositions. These challenges underscore the need for standardized production protocols, optimized delivery systems, and deeper mechanistic understanding to fully harness the therapeutic potential of GLPs in cancer therapy.
Currently, independently marketed GLP products. In China do not have a specific molecular conformation, their pharmacological mechanism is unclear. The level of evidence-based medicine is weak, and their international recognition is low, making it harder for them to enter the international market. The anti-tumor effects of GLPs vary significantly depending on the species and origin of
Summary
GLPs as the major bioactive constituents of
Despite these advancements, challenges remain in translating GLPs into standardized and internationally recognized anti-cancer therapeutics. Major limitations include their structural complexity, heterogeneous composition, and the lack of well-defined structure-activity relationships. Differences in extraction sources, processing techniques, and molecular characterization lead to variable biological efficacy and hinder regulatory approval as well as global clinical acceptance. Moreover, most mechanistic evidence remains at the cellular level, with insufficient comprehensive molecular validation and clinical trial support. Therefore, future research should aim to establish standardized production and quality control methods, clarify precise molecular targets through advanced glycomics and bioinformatics technologies, and develop novel delivery strategies to improve bioavailability and tumor specificity. Well-designed, multicenter clinical trials are also essential to provide stronger evidence-based support.
In conclusion, GLPs represent a promising natural resource for cancer therapy owing to their multifaceted anti-tumor functions and excellent safety profile. Further interdisciplinary research integrating structural elucidation, mechanism-based optimization, and well-designed clinical studies is required to facilitate their translation into standardized and evidence-based anticancer therapeutics with broader clinical applicability.
Footnotes
Acknowledgements
This work was supported by Natural Science Foundation of Zhejiang Province (LLSSZ25H280002); National Traditional Chinese Medicine Comprehensive Reform Demonstration Zone Science and Technology Co-construction Project (GZY-KJS-ZJ-2025-065); Zhejiang Medicine and Health science and Technology Project (2025KY1958); Zhejiang Province Traditional Chinese Medicine Inheritance and Innovation Talent Program (2023ZR059); Lishui Innovation Consortium-Key R&D Program Projects (2023LHT02).
Ethical Approval
Ethical Approval is not applicable for this article.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Author Contributions
Mengyuan Wang: conceptualization and writing – original draft; Xiaoju Guo and Jiale Chen: investigation and visualization; Zhang Chen and Qiaoyou Weng: writing – review & editing; Liyun Zheng and Shiji Fang: diagram preparation; Jiansong Ji, Minjiang Chen, and Shimiao Cheng: supervision. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing is not applicable.
Institutional Review Board Statement
Not applicable.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
