Abstract
Introduction
Herpes simplex virus (HSV) is a common infection in humans. Disease caused by this virus ranges from mild to severe; it may even lead to life-threatening conditions, especially in immunocompromised patients.1,2 The most clinical manifestation is herpes labialis and genitalis typically caused by HPV-1 and HPV-2 infection, respectively. 3 This virus can produce latent infection in the host for life and is reactivated to cause recurrent infections and lesions. 4 The incidence of HSV infection has continuously increased in many countries. Treatment of HSV is done using nucleoside analogs such as acyclovir (ACV) and other nucleoside derivatives, famciclovir, penciclovir and valacyclovir. 5 However, there can be side effects from using high doses of these drugs. In addition, some resistant mutants of this virus have been documented, especially in individuals receiving long-term medication. This can result in the recurrence of HSV-associated diseases, especially among immunocompromised patients. 6 Moreover, these drugs may cause the failure of treatment in the recurrence of latent viruses. Therefore, it has become important to find new anti-HSV agents with different mechanisms of action against the virus.
In this study, IPAD cream was prepared and its anti-HSV activity was assessed
Materials and methods
Virus, cell line and cell culture
HSV-1 wild-type strain KOS sensitive to acyclovir was kindly provided by Dr Donald Coen (Biological Chemistry & Molecular Pharmacology, Harvard Medical School, Boston, USA). The virus was propagated and titrated in an African green monkey kidney (Vero; ATCC CCL81) cell line that was kindly provided by Prof. Pilaipan Puthavathana (Mahidol University, Thailand). Viral stock was stored at −80°C until use.
3,19-isopropylideneandrographolide (IPAD)
IPAD was semi-synthesised from andrographolide isolated from
Animals
Bagg albino strain C (BALB/c) mice (female, 6 to 8 weeks old) were obtained from Nomura Siam International Co., Ltd, Thailand. They were maintained at 25°C and exposed to 12 h light/dark cycles with food and water ad libitum at the Animal Unit, Faculty of Medicine, Khon Kaen University, Khon Kaen, Thailand. Animal experiments were performed with the permission of Institute for Animals for Scientific Purpose Development (IAD), National Research Council of Thailand (NRCT), No. U1-01595-2558. This project was approved by the Animal Ethics Committee of Khon Kaen University (AEKKU 55/2556, Reference No. 0514.1.12.2/78).
Skin irritation test
The dermal toxicity of the IPAD creams was tested in BALB/c mice (female, 6 to 8 weeks old). The mice were treated with orally administered prednisolone (4 mg/kg) for 3 days before infection and continually for 10 days thereafter. Hair was removed from the dorsal side of each animal with a chemical hair remover (Veet® cream, Reckitt Benckiser). Skin on the mid flank (both sides) of each animal was scratched with a needle (21Gx1.5, Nipro, Japan). One hour after scratching, the IPAD creams (5 and 15%) were applied to the abraded area of groups of animals (n = 3) three times daily for a 10-day period. The amount of cream applied was 20 mg per lesion. The extent of erythema and edema on mouse skin was assessed and photographed daily using a digital camera (Nikon, D620) for 23-day period after the first topical administration on mouse skin as shown in Figure 1.

Schematic diagram of experiments using a mouse model for testing IPAD cream on anti-HSV infection. (a) Schematic diagram of each experimental step in a mouse model was shown. (b) After HSV KOS infection on both sides of mouse midflank skin, untreated, cream base, 5% acyclovir cream, 5% IPAD cream and 7.5% IPAD cream were divided in to each five group and applied to tropical administration on mice skin lesion in both midflank skin.
Therapeutic efficacy of IPAD cream on cutaneous HSV-1 infection
Prednisolone (4 mg/kg) was orally administered to BALB/c mice for 3 days before infection and continually for 10 days thereafter. The mid flank on both sides of each mouse was clipped and depilated with a chemical depilatory hair remover. Two days later, the naked skin was scratched using needles and 100 µL of HSV-1 (KOS) suspension of 250,000 PFU/mL was applied to the scarified area. Thereafter animals were divided into treatment groups (2 lesions per mouse and 3 mice per group; n = 6 lesions/group). One control group was not given any additional treatment. Another control group was treated using only the cream base. The remaining groups were administered with IPAD cream (5% or 7.5%) or 5% acyclovir cream. All creams were first administered to the relevant groups one hour after infection and four times daily thereafter for 10 days. The development of skin lesions and mortality were continuously observed and scored for 10 days. The score was set as 0 = no lesion; 2 = vesicles in local region; 4 = erosion and/or ulceration in local region; 6 = mild zosteriform lesion; 8 = moderate and/or severe zosteriform lesion; and 10 = death of mouse (Supplementary Figure 1.). All lesions were scored as above and photographed every day over a 10-day period (Figure 1).
Mouse cell collection and DNA extraction
Mouse cells in each group were collected by brushing the lesion with sterile Dacron swabs (Puritan, Hardwood Products, Guilford, USA). Each cell pellet was collected and washed by centrifugation. Genomic DNA was isolated from the cell pellet by the EZNA Tissue DNA Kit (Omega Bio-Tek, Norcross, GA, USA), according to the manufacturer's instructions. DNA quality and quantity were estimated using a NanoDrop 2000 microvolume spectrophotometer (Thermo Fisher Scientific, Rockford, IL, USA) and by amplification using primers for the house-keeping gene, glyceraldehyde-3-phosphate dehydrogenase (
Estimation of HSV DNA copy number by Rt-PCR
Genomic DNA from mouse cells taken at the skin lesion site was used to determine HSV DNA copy number using real-time polymerase chain reaction (RT-PCR). RT-PCR was performed in 10 µL volumes containing SsoAdvanced Universal SYBR Green Supermix (Bio-Rad, Mississauga, Ontario, Canada), 300 nM of each primer: HSV DNA polymerase (forward 5′-GTGTTGTGCCGCGGTCTCAC-3′ and reverse 5′-GGTGAACGTCTTTTCGAACTC-3′) and 50 ng of DNA from infected cells. Amplification and detection were performed using a CFX96 Touch™ Real-Time PCR Detection System (BioRad, USA). Cycling conditions were 45 cycles of 95°C for 30 s, 55°C for 30 s and 72°C for 30 s. Copy number was calculated by absolute quantification using a standard curve generated from purified HSV-1 (KOS) viral DNA that had been serially diluted into a range from 1 to 106 copies per reaction. Each PCR run contained several controls including two reactions without HSV DNA, a positive amplicon control, and a standard dilution curve of amplicon DNA. Each test group was run in duplicate and reported as the copy number of HSV DNA/femtogram (fg) in a reaction.
Detection of HSV gD expression by Rt-PCR
Total RNA from cells brushed from HSV-infected mouse skin was extracted using Trizol reagent according to the manufacturer's directions. cDNA was generated from this RNA (200 ng) with RevertAid Reverse Transcriptase (Thermo Scientific, Schwerte, Germany). Late gene gD expression was estimated by qRT-PCRs in 10 µL volumes that contained 1× SsoAdvanced Universal SYBR Green Supermix (Bio-Rad), 300 nM of each primer: gD forward 5′-AGCAGGGGTTAGGGAGTTGT-3′ and reverse 5′-CCATCTTGAGAGAGGCATCC-3′) and cDNA. Amplification of
Statistical analysis
The analysis of variance (ANOVA) with Bonferroni post-tests was used to analyze the difference between IPAD cream-treated mice and control mice in mean skin lesions and HSV copy number as well as gD expression for 10 days after infection.
Results
Toxicity of IPAD cream on skin
The dermal toxicity of 15% IPAD cream was assessed by application of the creams to the dorsal skin of the mice. No sign of edema or erythema was observed in any of the animals indicating that the creams have no dermal toxicity over a 30-day period after first treatment (Figure 2). In addition, no sign of edema or erythema was observed after application of 5% IPAD cream (data not shown).

Skin irritation test in female BALB/c mice without HSV-1 KOS infection. The cream base and 15% IPAD cream were topically administered on mice skin for 10 days. The skin was observed daily for any signs of erythema and edema.
Effect of IPAD cream on HSV-1 KOS infected mouse skin
Skin lesion was measured among negative control, cream base, 5% acyclovir, 5% IPAD cream and 7.5% IPAD cream. Only 7.5% IPAD cream was found to significantly reduce the lesion score of HSV-1 KOS-infected mouse skin at days 5 to 10 but not found in 5% IPAD cream in contrast to negative control and cream base. Inversely, 5% acyclovir cream significantly reduced the lesion score in day 4 to 10 in comparison with negative control (Figure 3). Interestingly, no significant difference of mean lesion score between 5% acyclovir and 7.5% IPAD was found over the 10-day period: both have similar efficacy.

Effect of topical administration of 5% and 7.5% IPAD cream on mice cutaneously infected with HSV-1 KOS. (A) Lesions on skin of mice caused by HSV-1 KOS were treated by topical administration of base cream, 5% acyclovir, 5% IPAD or 7.5% IPAD creams, applied four times daily as well as untreated cream (negative control with HSV-1 infection). (B) The means of lesion scores in the negative control group, base cream, 5% acyclovir, 5% and 7.5% IPAD cream groups were plotted over a 10-day period using Prism 5 for Windows.
Effect of IPAD cream on viral production and viral genome copy number
To compare the effects of acyclovir and IPAD cream, the HSV DNA copy number from skin lesion cells was examined. The HSV DNA copy numbers at day 10 of treatment were 0.02 and 21.7 per fg for 5% acyclovir cream and 7.5% IPAD cream, respectively. These values were not significantly different (P > 0.05) as shown in Figure 4. Copy numbers at day 10 in other treatment groups ranged from 271 (5% IPAD cream) to 694 (base cream group) per fg. This study suggested that the topical effect of 7.5% IPAD cream against HSV-1 skin infection was comparable to that of 5% acyclovir.

Effect of topical administration of 5% and 7.5% of IPAD cream on HSV DNA copy number in mice cutaneously infected with HSV-1 KOS. HSV-1 polymerase gene was amplified to determine viral copy number in DNA extracted from brushed mouse skin in the negative control group, base cream, 5% acyclovir, 5% and 7.5% acyclovir cream groups at days 3, 5, 6 and 10. The graph was created using Prism 5 for Windows.
Effect of IPAD cream on viral late gene, gD expression
The HSV gD gene functions in virus entry and fusion and could be inhibited by IPAD treatment according to our previous
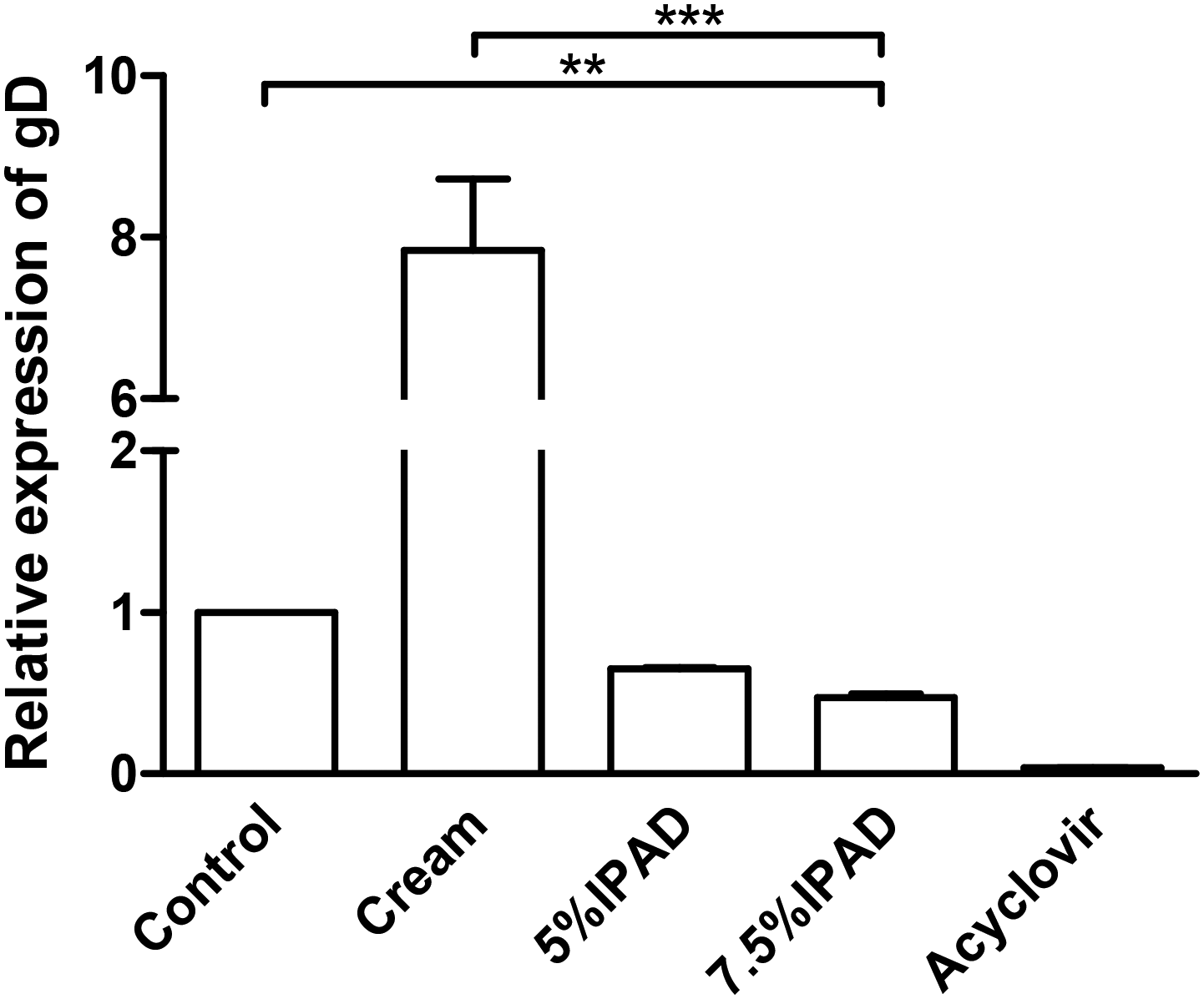
Effect of topical administration of 5% and 7.5% IPAD cream on inhibition of HSV gD expression in mice cutaneously infected with HSV-1 KOS. HSV-1 gD expression was investigated by qRT-PCR in the negative control group, base cream, 5% acyclovir, 5% and 7.5% acyclovir cream groups at day 10. The graph was created using Prism 5 for Windows.
Discussion
In this study, the anti-HSV activity of IPAD cream was evaluated. The 7.5% IPAD cream was shown to exhibit anti-HSV activity
In the mouse cutaneous infection model, first lesions of HSV-1 in the untreated and base cream-treated groups were apparent by day 3. IPAD (5% and 7.5%) delayed the appearance of lesions until day 4 and 5% acyclovir until day 5. Although IPAD and acyclovir were initially topically administered at one hour post-infection, HSV-1 lesions still developed on mouse skin. Both acyclovir and IPAD act on stages in the viral life cycle but cannot prevent the initial infection step by HSV-1. However, topical administration of 7.5% IPAD cream led to significantly lower lesion scores during days 5 to 10 relative to controls (P
Previous study suggested that application of antiviral compounds four to five times a day was the most effective in delaying the skin lesion development and in protection against mouse death. This was why we used four applications daily. 2
ACV is a nucleoside analog that exhibits anti-herpetic activity after phosphorylation by viral thymidine kinase (TK) and then contributes to acyclovir triphosphate which competes with the natural nucleotide, deoxyguanosine triphosphate (dGTP), for incorporation into viral DNA, resulting in the selective inhibition of viral DNA polymerization and prevention of continued extension of the viral DNA chain. 11 Our previous study demonstrated that IPAD exerted anti-DR HSV-1 (ACGr4) activity using a different mechanism. This viral strain contained mutations in its DNA polymerase genes and consequently conferred resistance to acyclovir. 12 Therefore, IPAD cream may be useful to inhibit acyclovir-resistant HSV strains. This will be further investigated in the future.
Interestingly, there are many studies that develop and apply natural products to inhibit HSV-infected lesions in both animals and humans. Cream containing 12.5% and 25% resveratrol (35,4-trihydroxystilbene) significantly inhibited HSV-1-induced skin lesions in mice. 13 Unsurprisingly, a higher proportion of resveratrol cream at 25% effectively suppressed the development of lesions topically applied two, three, or five times a day whereas 12.5% resveratrol cream effectively inhibited lesion formation when applied only five times a day. In addition, mice mortality rates of 0%, 6.25%, 12.5% and 19% resveratrol cream-treatment were 37%, 40%, 24%, and 3%, respectively. 14 This information may suggest that the more concentration of bioactive compounds was even more effectively inhibited HSV-1-infected lesions. In humans, the product from Kanuka honey included 90% kanuka honey and 10% glycerine cream had been demonstrated in participants with herpes simplex labialis and there was no difference in compared to 5% acyclovir topical cream. This product consisted of a very high amount of Kanuka honey. 15 Simultaneously, the treatment of herpes labialis with 2% olive leaf extract (OLE) cream is more effective than 5% acyclovir cream. 16 As mentioned above, the proportion of several natural compounds in cream base used in animal models and clinical trials have varied depending on concentration of bioactive compounds, the condition of drug release in the cream and topically applied times a day. In summary, IPAD cream provided therapeutic efficacy equivalent to that of acyclovir against HSV infection. Therefore, IPAD was verified to be a promising anti-HSV drug candidate for the topical treatment of HSV infections. Further investigations will focus on the development of topical formulations with, hopefully, higher effectiveness of IPAD as an anti-HSV agent.
Supplemental Material
sj-docx-1-avc-10.1177_20402066221089724 - Supplemental material for Activity of 3,19-isopropylidinyl andrographolide against herpes simplex virus type 1 in an animal model
Supplemental material, sj-docx-1-avc-10.1177_20402066221089724 for Activity of 3,19-isopropylidinyl andrographolide against herpes simplex virus type 1 in an animal model by Jureeporn Chuerduangphui, Thawaree Nukpook, Chamsai Pientong, Chantana Aromdee, Supawadee Suebsasana, Watcharee Khunkitti, Charinya So-in, Kanisara Proyrungroj and Tipaya Ekalaksananan in Antiviral Chemistry and Chemotherapy
Supplemental Material
sj-docx-2-avc-10.1177_20402066221089724 - Supplemental material for Activity of 3,19-isopropylidinyl andrographolide against herpes simplex virus type 1 in an animal model
Supplemental material, sj-docx-2-avc-10.1177_20402066221089724 for Activity of 3,19-isopropylidinyl andrographolide against herpes simplex virus type 1 in an animal model by Jureeporn Chuerduangphui, Thawaree Nukpook, Chamsai Pientong, Chantana Aromdee, Supawadee Suebsasana, Watcharee Khunkitti, Charinya So-in, Kanisara Proyrungroj and Tipaya Ekalaksananan in Antiviral Chemistry and Chemotherapy
Footnotes
Acknowledgements
Authors’ contributions
Declaration of conflicting interests
Funding
Supplemental material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
