Abstract
Introduction
Biomaterials are biological or synthetic substances that are designed to perform, enhance or replace the normal function of different tissues including skin, vasculature, bone, cartilage or tendon by interacting with the biological system. Ideal biomaterial properties vary depending on the tissue being replaced and required function. Ideal biomaterials are highly biocompatible, often with specific functionalisation usually serving as a matrix for cell adhesion that regulates cell processes such as proliferation, migration and matrix synthesis. The ideal biomaterial in orthopaedics would be highly biocompatible, inexpensive and straightforward to manufacture, reproducing the function of the replaced tissue such as stimulating the production of new bone. A great deal of research has been dedicated to developing ideal biomaterials for orthopaedics particularly focussed on osseointegration.
However, any surgical intervention, particularly with implantation of biomaterials, runs the risk of biomaterial-associated infection (BAI) or periprosthetic joint infections (PJI). Bacteria adhere to biomaterial surfaces, where they produce biofilms enhancing their colonisation, preventing phagocytosis and evading the immune response, as well as becoming more resistant to systemic antibiotics.1–4 BAI cause significant patient morbidity and mortality, contributing to implant failure and loosening with an average failure rate of 2%–5%.5–7 Patients often require extensive further surgical intervention and long-term antibiotic therapy. The average economic cost of each patient with PJI is £25,000. 8 An ideal biomaterial would also provide appropriate antibacterial action.
Biomaterial function differs depending upon the indication, and subsequently the infecting organisms may vary. In trauma, for example, skin commensals such as
Treatment of PJI requires a multimodal treatment strategy. Surgery is usually necessary to reduce the localised bacterial infection and remove the biofilm-coated implants, along with long-term systemic antibiotic drug administration.17,18 Local antibiotic treatment strategies such as polymethylmethacrylate (PMMA) antibiotic beads and high-dose antibiotic cement spacers show good clinical results;19,20 however, overdose of local and systemic antibiotics might negatively affect osteogenesis and are not without complications.21,22 Use of surface-modified orthopaedic implants is another option, which may provide clinical advantage. For example, reducing the initial risk of bacterial infection from early or late haematogenous spread, reducing local and systemic antibiotic toxicity effects and improving clearance of infection and subsequent osseointegration following established infection.
Understanding the interaction between host, biomaterial and microorganism is very important for the development of antibacterial orthopaedic implants for clinical applications. This review highlights the relationship between host cells, implant materials and bacteria from an osteoimmunological aspect. We also focus on current surface fabrication techniques of Ti and its alloys for the development of antibacterial surface modifications and their potential clinical applications.
Interaction between host, material and bacteria
The host reacts to microorganisms via innate and acquired immune responses, though some bacteria can evade this by producing a biofilm or by becoming internalised into the osteoblast cells (Figure 1). Planktonic state bacteria are initially attracted to a material surface by different forces, for example, van der Waals or gravitational forces. Once the bacteria have adhered to the surface, they form stronger adhesion using pili. These bacteria then form colonies on the implant surface and secrete extracellular matrix rich in polysaccharides and proteins to form a biofilm layer, hence protecting themselves from the immune system. Bacteria and biofilm formation can reduce osteoblast viability and disturb osteoblast–osteoclast interaction resulting in bone resorption.13,28 A better understanding of the host cellular and bacteria–material surface interactions will help to improve treatment of PJI as well as improve material design. The ideal biomaterial will reduce bacterial colony formation (by anti-adhesive or bactericidal effect), promote osteogenic induction and maintain long-term implant osseointegration.

Bacteria–material–host interaction. (a) Bacteria adhere on material surface and form a biofilm enhancing their proliferation and protecting themselves from immune response and antibiotic drugs. (b) Bacteria interact with host cells such as osteoblasts. Osteoblasts non-professionally internalise bacteria. 23 This mechanism helps bacteria evade the immune system. 24 Bacteria induce osteoblast apoptosis by toxin production.25,26 Infected osteoblasts also induce tumour necrosis factor–related apoptosis-induced ligand (TRAIL) via caspase-8. 27 (c) Immune cells, both innate and adaptive, attack the planktonic bacteria to reduce bacterial numbers. Infected osteoblasts produce cytokines to activate immune response. (d) Infected osteoblasts produce RANKL, CXCL2 and CCL3 which enhance osteoclastogenesis resulting in bone resorption. 28 , 29
Osteoimmunology: how bone cells respond to bacteria
Once bacterial pathogens are introduced into the host via direct surgical site or haematogenous spread, bacteria then present pathogen-associated molecular patterns (PAMPs) such as lipopolysaccharide (LPS), lipoprotein, lipoteichoic acid (LTA) or peptidoglycans.30–32 Bacterial detection results in activation of the complement cascade and attracts inflammatory cells to the infected site. Phagocytic innate immune cells, such as macrophage and neutrophils engulf and kill planktonic bacteria directly. They can recognise bacterial PAMPs via toll-like receptors (TLR), a family of cell surface pattern recognition receptors (PRRs). Ligation of TLRs activates intracellular nuclear factor kappa B (NFκB) signalling cascades, which results in the increased production and release of soluble chemoattractants (cytokines and chemokines). These recruit immune cells to the site of infection. 32 Professional antigen presenting cells such as dendritic cells (DCs) link the host innate and adaptive immune responses, and by activating both cytotoxic CD8+T-lymphocytes and B lymphocytes produce antibodies against bacteria. 33 Dysregulation of this process can result in an attenuated immune response, driving sustained chronic infection.
Osteoblasts respond to planktonic bacteria by several mechanisms. 34 Initially, osteoblasts can internalise bacteria into vesicles; however, some bacteria have adapted to remain quiescent or to secrete toxins such as phenol-soluble modulins (PSMs) to escape internalisation and induce osteoblast necrosis and apoptosis. These bacteria will then continue to infect surrounding cells. Infected osteoblasts also activate innate and adaptive immune cells by producing a plethora of cytokines and chemokines (such as interleukin-6 (IL-6), CXCL2, CXCL8, CXCL10, CCL2, CCL3 and CCL5).33,35 Infected osteoblasts also produce factors such as RANKL, granulocyte macrophage colony stimulating factor (GM-CSF), macrophage colony stimulating factor (M-CSF) and granulocyte colony stimulating factor (G-CSF) to enhance osteoclastogenesis leading to bone resorption.33,36
Osteoclasts originate from haematopoietic stem cells and differentiate from the same precursors as macrophages and dendritic cells. 34 Li et al. 37 demonstrated that mature osteoclasts can function as antigen-presenting cells and can activate CD4 + and CD8 + T cells. Osteoclast precursors are attracted to sites of infection by sphingosine-1-phosphate (S1P). 38 During the host response to a bacterial infection, macrophages become activated by the inflammatory environment and produce pro-inflammatory cytokines, which further promote osteoclastogenesis. 39
Understanding the immunological response to common microorganisms is necessary for treatment of osteomyelitis and PJI.
FnbA and B as well as Cna play important roles in bacterial binding to implants.
43
Testoni et al.
44
suggested Cna and Bbp synergised to drive
Bacterial–material interaction: underlying theories, interaction phases and biofilm formation
There are three main influences on bacterial interaction with material surfaces, material features, bacterial features and the environment (Figure 2).
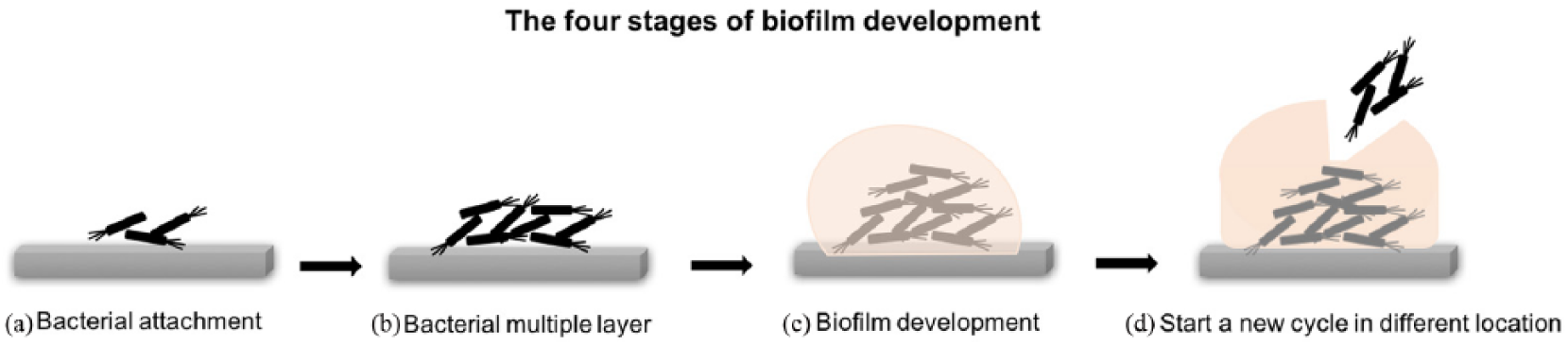
The four stages of biofilm development. (a) Initial bacterial attachment. (b) Bacteria start to produce multiple layers through cell aggregation and accumulation. (c) Biofilm development and matrix elaboration. (d) Bacteria start a new cycle of biofilm formation in different location.
Bacterial features
Different bacterial species have different adherent behaviour to biomaterial surfaces due to their physicochemical characteristics and preferred environment.46,47
Bacterial hydrophobicity/hydrophilicity
In general, bacterial species with hydrophobic properties prefer binding to hydrophobic surfaces and vice versa. However, the material surface hydrophobicity plays a more important role in bacterial adhesion than the intrinsic bacterial surface hydrophobicity. 46 Bacterial hydrophobicity changes according to bacterial age, surface structure and medium growth. Possible reasons behind this include increased exopolysaccharide production with higher salt concentrations and in aged cells and lower nutrients in the culture media. These factors all lead to a drop in hydrophobicity. 48
Bacterial surface charge
The relationship between bacterial surface charge properties and bacterial adhesion are not clearly understood. However, there is some evidence that bacterial charge is affected by growth medium, environmental pH, the buffer ionic strength, bacterial age and surface structure. Moreover, bacteria in aqueous solutions are usually negatively charged.46,47 Hence, the surface charge for both bacterial and biomaterial should be considered in predicting bacterial adhesion on material surfaces. 49
There are two phases of the bacterial–material interaction. Phase I: this is the initial, instantaneous and reversible physical phase. Phase II: this is the time-dependent and irreversible molecular and cellular phase. 47 Biomaterial surface topography such as pattern and roughness can affect the bacterial adhesion. 50
Normally, bacteria prefer to grow on available surfaces rather than in the surrounding aqueous phase. 51 During phase I, bacterial adhesion starts with surface attraction, followed by cell adsorption and attachment. 52 The bacterial movement occurs by physical interactions such as Brownian motion, van der Waals and gravitational forces; the surface electrostatic charge and hydrophobic interactions.25,29 In addition, physical interactions are classified into long- and short-range interactions. In long-range interactions, the distance between cells and surfaces is not specific (>50 nm). While in short-range interactions the cells have a close contact (< 5 nm) with the surface. Once the bacteria have attached to the surface (long-range interactions), the initial part of adhesion occurs (short-range interactions), allowing phase II to begin.47,53
There are three theories described to date that determine the interaction between bacterial cells and surfaces. First, the Derjaguin–Landau–Verwey–Overbeek (DLVO) theory describes the net interaction between cells and surfaces when the particle adhesion is affected by long-range interactions. These include Lifshitz-van der Waals interactions and overlapping electric double-layer interactions. 54 This explains the reason why some colloidal systems agglomerate while others do not. Second, the thermodynamic theory 55 describes bacterial attachment to the surfaces with different attractive and repulsive interactions like van der Waals, electrostatic or dipole. Generally, the thermodynamic theory works as a closed system where the organism converts the substrate to energy without any energy from the outside. 56 Finally, a combination of the available theories termed the extended DLVO theory was developed. This includes the hydrophobic/ hydrophilic interactions.57,58
In the second phase (adhesion phase), molecular-specific reactions occur between structures on bacterial surfaces with the substratum surfaces. Specific polymeric structures in bacteria such as capsules, fimbriae or pili serve as bridges between the cell and the surface.53,59,60 Clumping factors, proteins and teichoic acid are factors that may play a role in highly viscous masses. 46 Following phase II, bacteria may then begin to form biofilms on the material surface.
Biofilm formation on implants
Biofilm formation is an advantageous process for bacteria. The majority of the world’s bacterial populations are found in the form of a biofilm at various stages of development. 61 A biofilm is a structured group of bacteria that cover themselves in an exopolysaccharide matrix, which results in firm adhesion on the implant. Inside biofilms, intercellular bacterial communication regulates gene expression and adaptation including phenotypic variation and survival during nutrient starvation.62,63 In addition, the bacteria are protected from antibiotics and dynamic environments.64,65 There are four steps for biofilm formation (Figure 3). Step 1: The bacteria initially attach on the substrate. Step 2: Accumulation of multiple cell layers through cell aggregation and accumulation. Step 3: matrix elaboration and biofilm development. Step 4: Bacterial release to start a new cycle of biofilm formation in a proximal location.43,66,67 Following biofilm development, bacteria work in groups rather than as individual cells in a process called ‘quorum sensing’. A number of methods have been developed to prevent biofilm formation, including inhibition of quorum sensing, anti-adhesion drugs and macromolecules.43,68–72 However, we will focus on material surface adaptations.
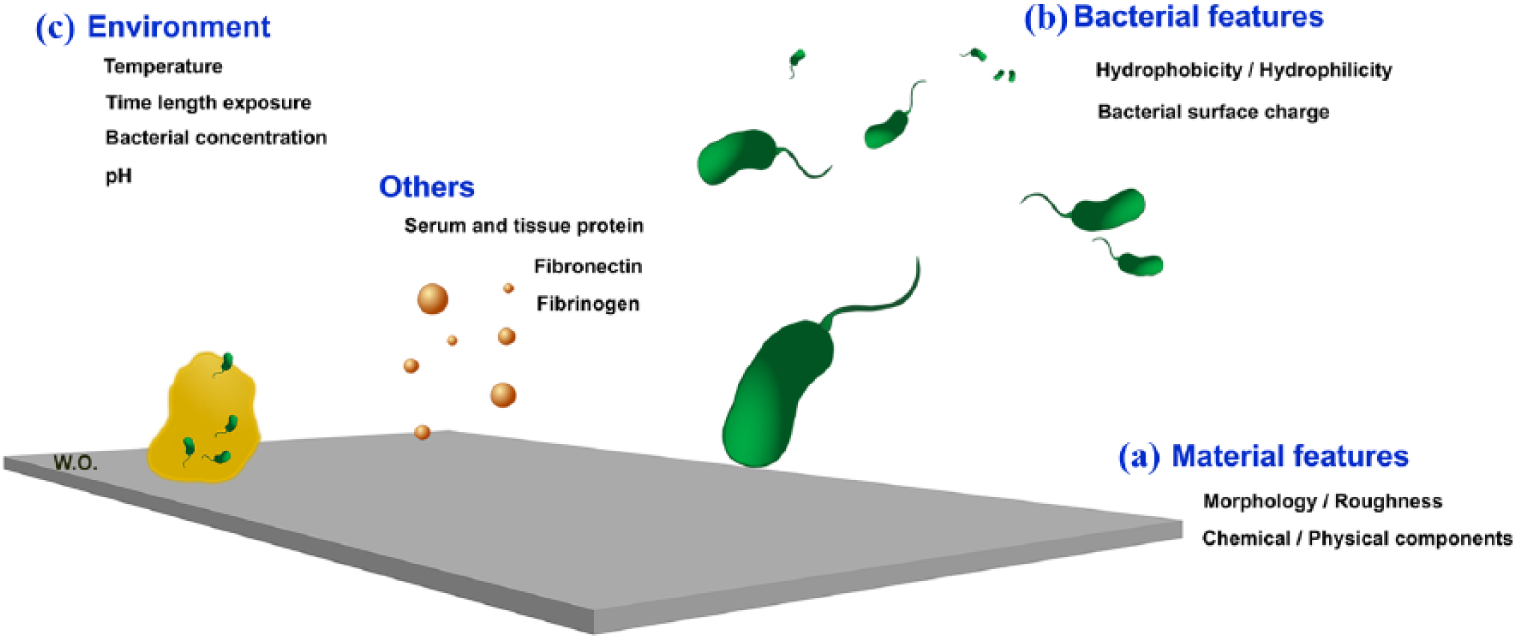
Three main features affect bacterial–material interaction. (a) Material features such as morphology and physicochemical cues. (b) Bacterial features including surface charge and hydrophobicity/hydrophilicity. (c) Environments such as temperature, pH, bacterial concentration and contact time as well as other factors such as serum and protein.
Material features
There are many factors influencing bacteria adherence to biomaterials surfaces including surface morphology and roughness, surface chemical composition and surface hydrophobicity or hydrophilicity.
Surface morphology and roughness
It has become clear that the micro/nano-topography of a biomaterial plays an important role in bacterial adhesion.73–79 This concept originates from observations made on the unique features of Cicada (
Various studies have published the effects of topographical modifications on bacterial adhesion and survival. Tsimbouri et al.
84
showed that TiO2 nanowires produced by hydrothermal oxidation reduce the
Surface chemical/physical composition
Surface chemistry can play a role in bacterial adhesion and proliferation. Materials with different functional groups change bacterial adhesion depending on material hydrophobicity/hydrophilicity and charge state.46,47 Water contact angle (WCA) measurements have been used to reveal the hydrophobic (high) or hydrophilic (low) nature of material surfaces. Metals tend to have a high surface energy, negative charge and hydrophilic features as confirmed by WCA, whereas polymers have low surface energy, less charge and hydrophobic features. 47 In addition, the hydrophilicity of materials may change with time, for example, Ti(OH)4 hydrophilicity may decline overtime due to air oxidation or carbon contamination and become TiO2 (hydrophobic).94–96 Thus, it is important to monitor the chemical changes in the surface features over time to determine the bacteria survival time.97,98
The environment
General environment factors such as temperature, time length exposure, chemical treatment, pH, antibiotic presence and bacterial concentration may play a role in bacterial adhesion.46,47,99 Optimum bacterial temperature allows bacterial enzyme activity, bacterial growth and biofilm formation.99,100 Temperature changes may also affect the physical properties, for example, at 35°C bacteria have a single flagellum, while at 21°C they have 2–3 flagella and at 10°C they have multiple flagella. At lower temperatures, the ability of biofilm adhesion increases if properties of polysaccharides are uniform. 101 This suggests that by raising the temperature, adhesion will reduce between the bacteria and the substrate. However, despite increasing temperature to 80°C–90°C, biofilm removal is not improved due to ‘baking effects’. 102
Changing pH and environmental chemical concentrations such as NaCl, HCl and KCl affect bacterial growth.99,103–105 Bacteria have the ability to respond to changes in internal or external pH by adjusting their activity and protein synthesis. 106 This allows the bacteria to adapt to small changes in their environment.99,107 Some bacteria have the ability to modify their metabolism to lower growth rate under specific drug pressures until they find favourable conditions for multiplication. 108 This can be favourable for bacteria as some antibiotics act to decrease bacterial adhesion.109,110
Other factors: serum or tissue proteins
Serum or tissue proteins such as fibronectin (FN), fibrinogen (Fg) and albumin may promote or inhibit bacterial biofilm accumulation on biomaterial surfaces.46,47
Fibrinogen (Fg) is a protein that plays a role in blood coagulation, platelet adhesion and aggregation and hemostatic processes.118,119
Titanium surface material modification
A wide variety of different materials are used as biomaterials; however, we will specifically focus on the surface modification of titanium in this review. Ti and its alloys (Ti-6Al-4V, Ti-5Al-2·5Fe and Ti-6Al-7Nb) are one of the most widely used materials in orthopaedics, both in trauma and elective practice. Ti has good corrosion resistance, high strength, low weight and modulus of elasticity much closer to that of bone than other metals. The benefit of using Ti-based alloys is their non-reactivity due to auto-passivation. However, the bioinert nature of Ti also means that as a flat surface, it shows no osteoinduction. Titanium is a very adaptable material, and many techniques have been used to modify surface roughness and create interconnecting porous architecture in order to promote osseointegration. Common microscale surface modification techniques for improved osseointegration can be divided into surface roughening (such as blasting, plasma spray, meshing, etching and anodisation) and surface coating (such as plasma-sprayed hydroxyapatite (HA) coating). Currently marketed implants include porous coating (e.g. Zimmer, CSTi); plasma-sprayed HA coating (e.g. DePuy Synthes, PureFix; Stryker); HA coated on porous plasma spray titanium alloy (BoneMaster; Biomet) and Sintered–titanium bead with plasma-sprayed HA coat (ROUGHCOAT; Smith & Nephew). Clinical trials have reported good clinical outcomes and prosthetic longevity with cementless fixation.50,120 However, the effect of macro- to microscale surface roughness on bone ingrowth remains inconclusive.121,122 Many of these techniques create bone on-growth rather than ingrowth and suffer from weak bonding between the surface layer and the underlying implant, with shearing and failure of the surface. 123 Furthermore, the effect of these surface modifications on bacterial adhesion and biofilm formation has been poorly studied.
The design of prostheses with surfaces that enhance osseointegration and osteoinduction, while also giving an antibacterial effect without cytotoxicity would be ideal, though it remains challenging. Nanotopographical surface modification is an interesting candidate for orthopaedic implants. The current success of nanoscale surface feature design is due not only to promotion of osteogenesis but some surface features also prevent bacterial adhesion. Therefore, understanding the difference between surface patterns and their interaction with osteoblasts and bacteria is crucial for nanotopographic design. Puckett et al. studied bacterial adhesion on different nanotopographic patterns on titanium including nanotubular, nanotextured and nanorough. Nanorough created by electron beam evaporation decreased adhesion of
Topographical modification for improved osteointegration
There are various techniques for patterning material surfaces at the nanoscale such as photolithography, polymer demixing, electron beam lithography and anodisation, the more common examples are summarised in Table 1. 136 Many of these show induction of osteogenesis including nanotubes, 126 nanopits, 137 nanopores 138 and nanopillars. 132
Examples of nanopatterning on titanium surface and fabrication techniques.

Surface antibacterial modification
Ideal antibacterial implant coatings should be biocompatible with no local or systemic toxicity, easy to use with proven antibacterial effects, as well as inexpensive and easy to manufacture. 139 The currently available products in the market are shown in Table 2. The most commonly used involve either antibiotic loading or silver ions; however, these products are expensive, cause local cell toxicity and the long-term side effects and clinical outcomes require further study.
Examples of available antibacterial techniques and orthopaedic implants in the market.

MIC: minimum inhibitory concentration.
Current techniques for reducing bacterial attachment and biofilm formation include anti-adhesive function and bactericidal function. Examples of available strategies for antibacterial treatment including surface coating, nanotopography, as well as dual-function are shown in Figure 4 and Tables 3 and 4.

Planktonic bacteria attach on material surface and form biofilms. (a) Various techniques were used as antibacterial strategies. Anti-adhesive surface coats using concepts of surface chemistry and functionality including ions 145 and polymer coats. 146 (b) Material surface can be coated with bactericidal substances such as antibiotics 147 and silver. 148 (c) Nanotopographic surface modifications were also effective strategies used as either anti-adhesives or bactericidal. (d) The examples of nanotopography, such as nanowires promoting osteoblastogenesis and have bactericidal effects. 84 Other bactericidal topographies include nanotubes (permission from Yu et al. 149 ) and cicada wings (permission from Ivanova et al. 80 ).
Examples of surface coating.

EHDA: electrohydrodynamic atomisation; MEVVA: metal vapor vacuum arc.
Examples of topographic surface modification.

MRSA: methicillin-resistant Staphylococcus aureus; MSSA: methicillin susceptible Staphylococcus aureus.
Anti-adhesive function
Topographic modification
The effects of microscale topography on bacterial adhesion remain controversial. Whitehead et al. 90 demonstrated that bacteria can be retained in pits (substratum layer) depending on bacterial size and pit size, however, other authors have suggested that microscale surface roughness does not affect bacterial adhesion.171,172 Interestingly, some nanoscale topographic features can promote cell differentiation while decreasing bacterial adhesion. Ploux et al. 168 created nanoscale pattern on silicon wafers by UV-photolithography. This surface reduced bacterial adhesion and allowed human osteoprogenitor cell adhesion.
Surface chemistry modification/surface coating
Ionic implantation/element coating
Silver ion coating of biomaterials has been widely studied. Della Valle et al.
173
used anodic spark deposition (ASD) treatment to incorporate chemical elements such as silver nanoparticles, calcium or silicon into titanium oxide. They showed that the silver nanoparticle coating reduced bacterial adhesion, as well as being biologically safe. Combinations of nanotopographic surfaces and silver coating have also been studied. Das et al.
174
fabricated titania nanotubes by anodisation and subsequently treated with silver deposition. These nanotubes showed good osteoblast adhesion and proliferation, while
Selenium is another element that can inhibit bacterial attachment. Holinka et al.
155
studied the effect of sodium selenite coating of titanium discs on bacterial adhesion and showed that it can reduce
Surface functionality (receptor/ligand interaction)
Polymer brush is a technique attaching polymer chains such as polyethylene glycol (PEG), polyethylene oxide (PEO)156,175 onto surfaces to prevent bacterial adhesion and protein adsorption. Nejadnik et al.
156
showed that PEO can reduce
Bactericidal function
Topography modification
Ivanova et al.
80
first noted the antibacterial effect of cicada wings (
Surface chemistry modification/surface coating
Antimicrobial polymers
Timofeeva and Kleshcheva 179 described the use of positively charged polymers (such as quaternary ammonium or phosphonium polymers) to attack bacterial surfaces, which are negatively charged. These polymers act as surfactants which can damage bacterial cell walls and cell membranes, resulting in cell lysis. 180
Organic origin , for example, antibiotics, anti-infective peptides and chitosan
Antibiotic coating
There are many ways to deliver antibiotic drugs such as loading antibiotic in bone cement or degradable materials and superficial modification of materials through covalently binding antibiotics or composite materials consisting of antibiotics embedded in gel or solid matrix.
181
Examples of antibiotic elution are gentamicin-loaded poly-
Chitosan coating
Chitosan itself has bactericidal effects
183
and can be used as a drug-eluting coating. Ordikhani et al.
153
coated titanium surfaces with chitosan–vancomycin composite by a cathodic electrophoretic deposition technique. Lin et al.
184
and Yang et al. fabricated quaternized chitosan derivative (hydroxypropyltrimethyl ammonium chloride chitosan)-loaded titania nanotubes produced by titanium anodisation.
184
Other mechanisms
Competing interaction molecules
Potential adverse effects from surface modification
Wear particles, from both bearing and implant materials, may have local and systemic implications. Wear particles have been found in liver, spleen and lymph nodes, 187 and silver nanoparticles have been identified in brain astrocytes. 188 Wear particles, particularly polyethylene, are one of the primary causes of periprosthetic osteolysis resulting implant loosening and failure. Particle size plays an important role in determining the cellular reaction. Particle sizes >500 nm tend to be engulfed by professional phagocytes using an actin-dependent mechanism, 189 while small particles are endocytosed by non-professional phagocytic cells. Ti wear particles (1.5–4 µm) have negative effects on osteoblast proliferation and viability, 190 induce fibroblasts to release matrix metalloproteinases (MMP) resulting in osteolysis 191 and increase MMP2 and 9 activity, resulting in reduction of bone formation. 192 Micrometric Ti particles impaired Saos-2 adhesion strength, migration and proliferation. 193 Furthermore, Ti wear particles stimulate production of inflammatory cytokines, induce lymphocytes to start a type IV immune reaction 194 and increase vascular endothelial growth factor (VEGF) expression and p44/42 mitogen-activated protein kinase (MAPK) activation in monocytes and macrophages. 195
Ti dioxide (TiO2) is widely used for nanoscale surface modification; however, wear nanoparticles specific to TiO2 may have adverse effects. TiO2 nanoparticles have been shown to disseminate to heart, lung and liver and can cross the placenta.196,197 They have been shown to transfer to offspring and affect the cranial nerve systems in a mouse model, 198 have multiple immunomodulatory effects and may be associated with genotoxicity. 199 In the local environment, TiO2 nanoparticles have been shown to adversely affect cell migration and MSC differentiation in rats. 200 However, no long-term clinical studies have shown any adverse effects from the dissemination of TiO2 particles.
Conclusion and future perspective
Prosthetic and bone infections are devastating to patients and healthcare services. We have reviewed the manner in which bacteria interact with implants and host cells and developing surface modification strategies using titanium implants to prevent bacterial adhesion, while maintaining or improving implant osseointegration. Many surface modification strategies have been developed over recent years with some promising success in vitro, but many have yet to find in vivo or clinical use. We would anticipate the adoption of these promising surface modifications to help prevent bacterial colonisation of implants in the future and provide better treatment options.
