Abstract
Introduction
Cauda equina syndrome (CES) results from compression of the nerve roots emerging below the termination of the spinal cord. 1 Cauda equina syndrome occurs due to prolapsed intervertebral disc in 45% of cases but can result from any compressive pathology. 2 There is no unanimous clinical definition of CES, however common features include perineal sensory disturbance, pain, genitourinary dysfunction, and lower limb motor deficits.1,3 Cauda equina syndrome is a clinically important issue as the physiological, occupational and social ramifications of its sequelae can be devastating for patients. 4 As such, it is a true neurological emergency and requires urgent surgical intervention when compressive pathology is present. 4
A core outcome set (COS) defines an agreed minimum set of outcomes that should be reported by research for a specific health condition or health area. They enable attempts to provide an overview of existing evidence including systematic review and meta-analyses, reduce reporting bias, and ensure investigation of clinically important outcomes. 5 A cauda equina syndrome core outcome set (CESCOS) has previously been defined 6 with an international group of healthcare professionals and patients using consensus methodology. The CESCOS identified the minimum standard outcomes of surgically managed CES which research studies should be reporting. 6 Non-surgically managed CES was not included in CESCOS and as such was not a focus of the present study. 6 The core outcomes from CESCOS are outlined in the methods section of this article.
Outcome measurement instruments (OMI) are tools and frameworks used within research to measure a pre-specified outcome, for example the Landriel Ibañez classification for neurosurgical post-operative morbidity. 7 Outcome measurement instrument’s can include questionnaires, laboratory measurements, and specific clinical definitions. The COnsensus-based Standards for the selection of health Measurement INstruments (COSMIN) initiative is formed by a multi-disciplinary team of researchers with expertise in the field of OMI’s. 8 The initiative has a number of key aims, including the standardisation of research outcomes by the development of COSs. 8 COnsensus-based Standards for the selection of health Measurement INstrument provides guidance on performing reviews of OMIs, including Patient Reported Outcome Measures (PROMs) and Clinician Reported Outcome Measures (ClinROMs). 8 Guidance also exists for identification of OMI’s for the defined outcomes in a COS. 9 Only one third of core outcome set developers proceed to the next step, the development of a Core Outcome Measurement Set (COMS). 10 The lack of COMS is a known barrier to the uptake of COS within clinical trials. 11 Currently, there is no consensus agreement available on which measurement instruments should be used to measure the defined core outcomes in CES. 6 Cauda Equina Syndrome core OMIs are needed to further improve the comparability of similar studies in this area, facilitate meta-analysis and reduce research waste. 12
Rationale and Objectives
The purpose of this study is to perform a systematic review of commonly used PROMs and ClinROMs in the CES evidence base. Additionally, this study also identifies and evaluates studies seeking to develop or validate research instruments for use in CES.
Methods
This is a systematic review of the OMI’s used in the reporting of clinical studies for CES. The methodological approach to identifying research measurement instruments has previously been defined and a similar method was undertaken to pre-existing literature.8,13 No patient consent or ethical approval was required for this study.
Inclusion and Exclusion Criteria
Inclusion Criteria for Eligible Studies.

Database Search and Data Extraction to Identify Outcome Measurement Instruments
To identify studies containing OMIs, 3 databases were searched. Search strategies for Ovid (Medline), Ovid (Embase), and CINAHL Plus were developed. The full searches are outlined in Supplementary Materials 1. Studies published within the inclusion time-period (between January 1, 1990 and April 30, 2022) were extracted. This time period was split into 2 discrete searches. First, a historic search described in Srikandarajah et al provided studies from January 1, 1990 to September 30, 2016. 14 In Srikandarajah et al. the authors provide a review of reported outcomes for surgically managed CES patients, within the CES evidence base. The search strategy results from this study formed the basis of the initial search in the present study, as the evidence base was the same, and only the OMI’s used required extracting from the prior results. Additionally, this search was updated using a qualitative study filter as the historic search had excluded studies of this type. Updating the historic search whilst applying the qualitative filter allowed the original screening results to be used despite the difference in inclusion/exclusion criteria. This search was then updated using an identical strategy to cover January 1, 2015 to the April 30, 2022. An overlap of 1 year was included to reduce the chances of erroneously missed studies. Articles were single screened by 2 authors (G.E.R. and N.S.). Following screening, approximately 10% of all screened records were checked by the senior authors (N.S., M.W. and S.C.).
Core Outcomes Defined for Use in CES. 6

Database Search and Data Extraction for Instrument Validation Studies
To identify studies investigating the validity of measurement instruments in CES, a separate search was conducted. The search covered the period between September 1, 1990 and May 30, 2022. The full search strategy is provided in Supplementary Materials 1. To identify validation studies, the sensitive PubMed search filter was employed from Terwee et al. 15 This search filter was developed to identify studies on PubMed that look specifically at the measurement properties of measurement instruments, rather than the OMI’s themselves. 15 No other tool is available to serve this purpose. 15 The search was only conducted on PubMed since the filter was developed specifically for this platform. Screening of articles was performed by one author (G.E.R.). Approximately 10% of all screened records were checked with a senior author (N.S.).
Risk of Bias Assessment for Included Measurement Instrument Validation Studies
To evaluate the risk of bias for measurement instrument validation studies, the 2018 COSMIN risk of bias checklist was used.8,16 For each component of the checklist, studies are scored on a four-point scale from “very good”, “adequate”, “doubtful” to “inadequate”. 16 Evaluation of content validity was the primary step when evaluating instruments and was evaluated according to published guidance. 17 Studies with inadequate content validity were not considered for further evaluation. 8 Next, evaluation of internal structure was performed by considering structural validity, internal consistency, and cross-cultural validity/measurement invariance. 8 Evaluation of the remaining measurement properties was then performed. These include, reliability, measurement error, criterion validity, hypotheses testing for construct validity, and responsiveness. 8 Risk of bias assessment was undertaken independently by 2 authors (G.E.R. and N.S.). Difference in scoring were discussed and any disagreements were settled between the authors.
Results
Studies Containing Outcome Instruments
A total of 112 studies investigating surgical outcomes for CES were included. The PRISMA flowchart for the combined instrument search is demonstrated in Figure 1. PRISMA flow chart for combined instrument identification search.
Frequencies and Percentages of Study Types Included in the Systematic Review.

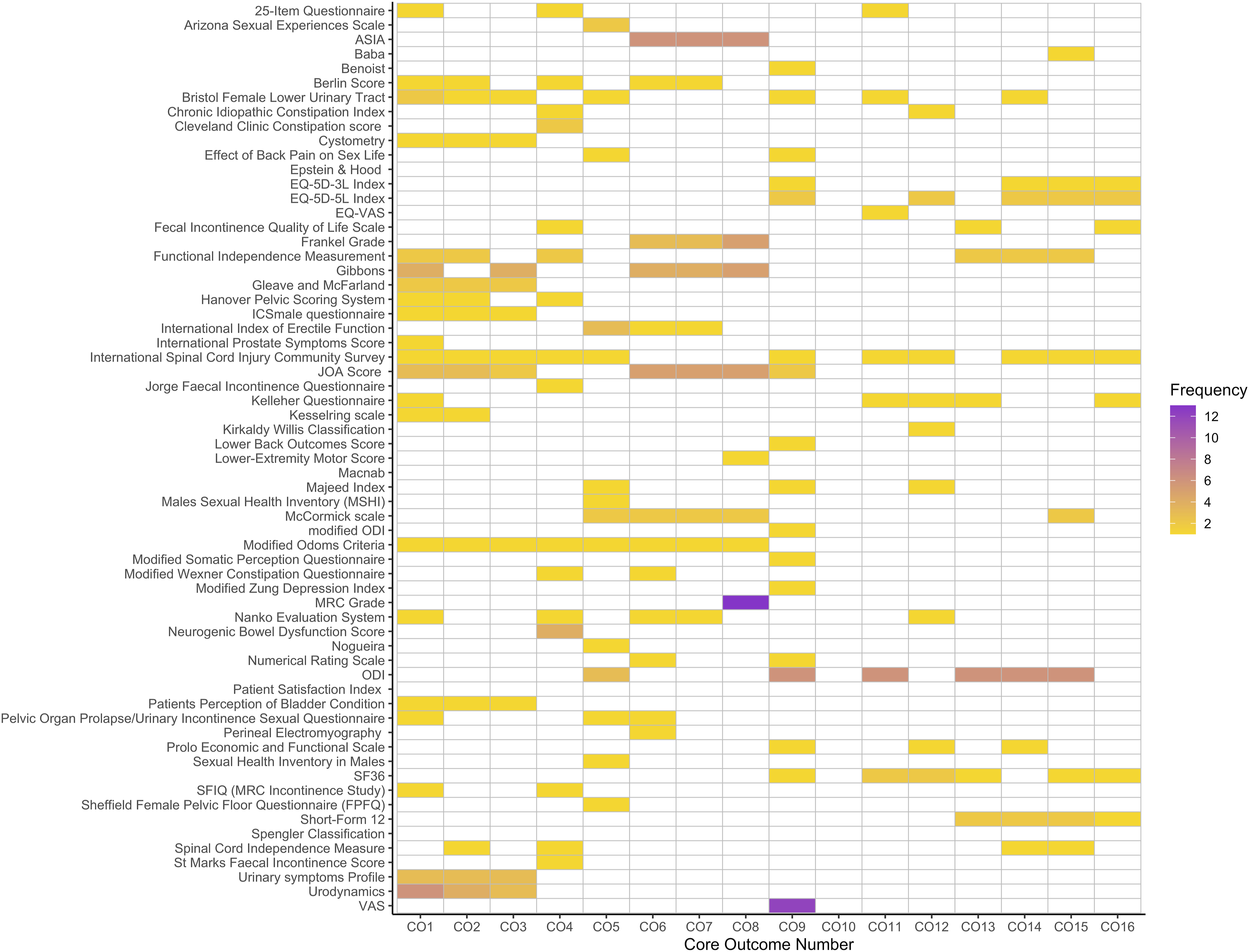
Heatmap demonstrating measurement instrument use frequency by respective core outcome. Core outcomes numbers are defined in Table 2.
Measurement Instrument Validation Studies
One study validating a measurement instrument in CES was identified. The PRISMA flowchart for the validation study search is shown in Figure 3. A search of the COSMIN database for the OMI’s identified in the initial search (Figure 1) identified that 13 of the instruments had been included in prior COSMIN reviews. These 13 OMIs were included in reviews relevant to spinal cord injury, however there were no OMIs included in reviews relevant to CES. PRISMA flow chart for instrument validation study search.
The study identified did not exclusively validate an instrument in a population of CES patients and instead formed part of a composite cohort. In the study, the authors sought to validate a foreign language version of an instrument in patients with multiple sclerosis and spinal cord injury, the latter of which contained a smaller cohort of CES patients. 18 The instrument was not included in any of the studies from the initial search strategy (Figure 1).
In Noordhoff et al the authors sought to validate the Multiple Sclerosis Intimacy and Sexuality Questionnaire (MSISQ-15) in a cohort of Dutch language speaking participants. 18 Six Cauda Equina patients were included alongside forty-three spinal cord injury patients as one composite cohort. Participants were asked to complete the MSISQ-15 questionnaire at their initial enrolment and complete it a second time 2 weeks later at home. The questionnaire set included the MSISQ-15, the EQ visual analogue scale (EQ-VAS), Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ-12), and International Index of Erectile Function (IIEF-15). Results were compared to a reference cohort recruited from a single general practitioner’s patient group. Content validity was deemed to be adequate during the linguistic validation process and found the translated version of the questionnaire to be understandable. Internal consistency of the MSISQ-15 was deemed to be good, with a Cronbach’s alpha of >.8 for test-retest in all cohort groups. Of note, within the spinal cord injury cohort (containing Cauda Equina patients), the primary domain of the MSISQ-15 demonstrated only moderate internal consistency with a Cronbach’s alpha of .53 and .55 (for test and retest, respectively). Reliability analysis demonstrated adequate (>.7) intraclass correlation coefficients for the MSISQ-15 and the 3 domains in all participant cohorts. Pearson’s correlation coefficients demonstrated significant relationships between the MISISQ-15 and PISQ-12/IIEF-15 in female/male cohorts respectively.
COSMIN Assessment of Methodology
Results From COSMIN Analysis of Validation Study.

Discussion
Key Findings
This review identified a total of 63 unique measurement instruments within the current CES evidence base. The most frequently used measurement instruments were the MRC grade of muscle power and the VAS of pain. One study was identified seeking to develop or validate instruments for use in CES populations. This study was not specifically validating for use in CES alone and the OMI was not used within the wider CES evidence base (e.g. identified during the initial CES OMI search).
Outcome Measurement Instrument Validation and Heterogeneity
The number of validation studies identified in this review was very low, even in comparison to similarly rare diseases. For example, COSMIN database reviews have identified 23 measurement instruments or methods validated for use in Huntington’s disease.19-22 Both conditions have a similar prevalence, and the disparity between number of available instruments for CES should trigger a call for further work to validate instrument for use in ongoing research. Moreover, of the validation studies that were highlighted by this review, none were specifically focused on cohorts of patients with CES and the instrument being developed or validated were not implemented within the wider CES evidence base. As previously stated, consensus methodology should be used to decide if identified measurement instruments are suitable to quantify the core outcomes. Suitable instruments would then require validation for use within surgically managed CES, as highlighted above by the lack of currently validated instrument. The pragmatic short-term solution to this is to adopt multiple pre-existing measurement instruments, a combination of which would address all the defined core outcomes in CESCOS. Such a combination of instruments could then be validated to ensure their adequacy. The only foreseeable drawback to this approach is that no instrument identified for the purpose of quantifying morbidity. This could possibly be rectified by seeking outside the CES specific literature and validating a pre-existing complications grading system, such as the Therapies-Disability-Neurology grade. 23
Within this review a large number of unique OMI’s were identified. The outcomes measured by these instruments varied greatly, as did the degree to which they were implemented. The MRC grade and VAS were the most frequently used measurement instruments.24,25 Their high frequency of use is likely related to their ubiquitous nature in other topic areas, the simplicity with which they can be applied, and their low burden of implementation. Many of the other instruments identified were used only a single time. The result is reduced comparability between similar CES studies and limited opportunities for meaningful pooled meta-analyses. Consensus methodology should be utilised to obtain an agreement among key stakeholders on how each core outcome should be measured, and if the identified measurement instruments are appropriate to do so.
For complications (core outcome 10), no CES specific instrument was identified within any of the current evidence base. This is because studies presented data on morbidity and mortality in its raw format. For example, limb weakness following surgery would be reported as mentioned, rather than within an established complication grading system (such as the aforementioned Therapies-Disability-Neurology grade). 23 This raises the issue of whether all core outcomes require a specific measurement instrument, for example mortality can be easily defined clinically, however such omissions from COMS would still require consensus agreement. The instrument identified with the highest utility in this regard was the International Spinal Cord Injury Community Survey (ISCICS), addressing a total of eleven core outcomes defined in the CESCOS, however this was used in only one study of CES patients and no studies were found validating this for use specifically in cauda equina. The ISCICS itself was developed with the aim of investigating the lived experiences of spinal cord injury patients on an international scale, of which CES was included. 26 The ISCICS was an amalgamation of existing measurement instruments, including the SF-36, Model Disability Survey, and the General Self-Efficacy Scale. 26 The world health organisation’s international classification of functioning, disability and health (ICF) was used as a framework against which the outcomes of interest were selected. 26 The higher degree of utility related to the core outcomes defined relates to breadth of outcomes included within the ICF. 27
This study represents a single step in the ongoing work to improve the reporting quality of surgically managed CES. The next step would be consensus agreement on which OMIs are appropriate for each outcome in the CESCOS. Then, using the recommended CESCOS and the appropriate OMIs for them, a study can be designed to adequately investigate the short- and long-term outcomes for patients with CES after surgery.
Limitations
Studies in this review were subject only to single author review. This was felt to be appropriate as similar methodology was employed for the previous study on which the original search strategy was based. 14 It is possible that measurement instruments identified in the initial search may have been validated in a broader population still of relevance to CES but were not included as the validation search focused solely on CES patient cohorts.
Conclusion
This review highlights the current measurement instruments utilised in the surgically managed CES literature, within the context of the defined core outcomes. Further work is needed to validate adequate OMI for the measurement of these outcomes specifically in CES populations. As with the development of COSs, a consensus approach involving key stakeholders should take place to decide which measurement instruments best achieve the requirements of ongoing surgically managed CES research.
Supplemental Material
Supplemental Material - Identification and Assessment of Outcome Measurement Instruments in Cauda Equina Syndrome: A Systematic Review
Supplemental Material for Identification and Assessment of Outcome Measurement Instruments in Cauda Equina Syndrome: A Systematic Review by George E. Richardson, Christopher P. Millward, James W. Mitchell, Simon Clark, Martin Wilby, Anthony G. Marson, Paula R. Williamson, and Nisaharan Srikandarajah in Global Spine Journal
Supplemental Material
Supplemental Material - Identification and Assessment of Outcome Measurement Instruments in Cauda Equina Syndrome: A Systematic Review
Supplemental Material for Identification and Assessment of Outcome Measurement Instruments in Cauda Equina Syndrome: A Systematic Review by George E. Richardson, Christopher P. Millward, James W. Mitchell, Simon Clark, Martin Wilby, Anthony G. Marson, Paula R. Williamson, and Nisaharan Srikandarajah in Global Spine Journal
Footnotes
Declaration of Conflicting Interests
Funding
Data Availability Statement
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
