Abstract
Introduction
Metallic materials have been extensively used for biomedical applications can be tracked down to the past three decades. Researchers have been successfully developed various bio-compatible materials and technologies for human body. The materials that are used are inert to body fluids. These body fluids flow over the bone and between the tissues and help in the circulation of oxygen and various other nutrients that is required by the human body. 1 The most commonly used materials that are used in the human body are stainless steel (316L), Ti-based alloys, and Co-based alloys. These metallic materials and alloys are typically used in orthopedic, dental, and cardiovascular applications in the human body. 2 Due to lower costs, stainless steel is the most widely used material. Apart from being cost-effective, they have good resistance to corrosion by body fluids, superior mechanical properties, and good biocompatibility. These properties present in the metallic materials make them a perfect candidate for use in the fabrication of biomedical implants. 3 The movement of human body joints is the final act of a coordinated action of the muscles and the bones where they are inserted. Thus, any undesired interruption of the chronological action may lead to an impairment in the joint. In this light, good biometallic implants used to replace injured bone tissue play a crucial role in the maintenance of the whole physiological and biomechanical scenario. It is also crucial for all the tissue around the joint to have a good adherence to the surface of the implant.4–6
The use of metallic materials as an implant material has several limitations. They have a weak bone-bonding ability. Long-term usage of these metallic materials causes the release of toxic ions into the body fluid. The aggressive body fluids cause a quick degradation of these materials inside the human body.7–9 Various osteoconductive coatings like hydroxyapatite (HAp) or other calcium phosphates (CaPs) are applied over the implant material to enhance their biocompatibility.10–12 After the implant is fixed in the body, the bone tends to form a bonding with the bioactive layer. As a result, no other fibrous interface layers forms. CaPs can exist in various phases, depending on the Ca:P ratio. The crystallinity and the bioactive behavior can be controlled by changing the Ca:P ratio. Hydroxyapatite (HAp) is the most stable phase of calcium phosphate in the physiological environment (pH ≈ 7). There are many other phosphates like brushite [BS, CaHPO4], monetite (CaHPO4), dicalcium phosphate dihydrate (CaHPO4·2H2O), and octacalcium phosphate (Ca8(HPO4)2(PO4)4·5H2O), which are stable in an acidic environment. In body fluids, brushite has a higher solubility than that of HAp. However, HAp has a greater amount of similarity to natural human bone. Therefore, HAp exhibits an excellent re-mineralization ability and bioactive behavior. 13 Likewise, brushite also can be used as an alternative since it acts as an HAp precursor by slowly transforming into HAp while coming in contact with suitable body fluids that are alkaline in nature.14,15
Metallic surfaces can be coated with these bioactive coatings by using various techniques like pulsed laser deposition,16,17 sputtering,18,19 plasma spraying,19,20 electrophoretic deposition (EPD),21,22 sol-gel,
23
and electrodeposition (ED).24–26 Electrodeposited coatings have a uniform thickness can be produced at a relatively low cost. The phase purity of the coatings, degree of crystallinity, and morphology can be tailored by varying the deposition temperature, pH, Ca:P ratio, and the concentration of additives. However, the above mentioned methods employ organic solvents or organic templating agents that are a potential threat to the environment as well as the biological systems.
27
Therefore, this work focuses on the synthesis of HAp-brushite composite coatings by using abundant environmental-free compounds because of their renewability and cost-effectiveness compared with other synthetic organic templates.28–30 There are various environmentally compatible alternatives to traditional reagents used for the production of HAp-brushite composite coatings. Primarily they are based on plant extracts. Locally available apple (
Pioneering work carried out by Gopi et al. demonstrate the use of “green templates” that is, naturally extracted organic compounds for the synthesis of HAp such as pectin derived from banana peels,
31
extracts of banana, grape, and tamarind,
32
and sucrose derived from natural as well as commercial sources.
33
They also reported the use of apple-derived malic acid for the preparation of HAp nanoparticles via a sol-gel route.
34
They compared the effectiveness of naturally extracted malic acid from apples apple (
Hence, this present study focuses on developing a consistently adherent composite layer of HAp (Ca to P ratio 1.67) and brushite (Ca to P ratio of 1) over a surface of stainless steel via electrodeposition technique in the presence of naturally extracted malic acid. The use of electrodeposition allows the direct deposition of the bioactive HAp-brushite composite coating on the metallic bioimplants with good adhesion. The deposited coatings have the required amount of a crystallinity and adequate surface area for initiation of tissue growth. The bioactive behavior of the HAp-brushite composite coatings is also investigated.
Experimental procedure
Substrate preparation
The substrates for the electrodeposition of HAp-brushite composite were prepared from the stainless steel (316L) sheet having a thickness of 180 µm procured from Jiangsu Taigang Puxin Stainless Steel Co., Ltd., China. The elemental composition of the substrate, as obtained from energy dispersive spectroscopy (EDS) analysis, is shown in Table 1. The sample surface was polished with different grits of silicon carbide emery papers (600, 800, 1500, and 2000 grits). After polishing, the sample surface was cleaned thoroughly with distilled water and rinsed with ethanol. An exposed surface area of 2 cm2 was maintained by masking the extra surface with a layer of non-conductive epoxy resin.
Elemental analysis of the SS 316L substrate (weight %).

Materials used for malic acid assisted HAp-brushite composite coating fabrication
Ultrapure forms of phosphoric acid (H3PO4), calcium chloride dihydrate (CaCl2.2H2O) and aqueous ammonia solution were procured from Merck. All the chemicals were of laboratory use grade, and they were used directly. Locally available apple (
Electrodeposition of HAp-brushite composite coating
The composite layer was electrodeposited over the stainless steel (SS) substrate via pulse electroplating route. A square-wave pulse was applied with 50% duty cycle (Ton = Toff = 10 ms). A typical two-electrode system with SS plates as working as well as the counter electrode was used. The working electrode had an exposed area of 2 cm2, and the counter electrode had an exposed area of 4 cm2. The electroplating bath was prepared by a consistent mixing 0.1 M of CaCl2. 2H2O (Merck, >98% purity), 0.15 M of NH4H2PO4 (Merck, >98% purity) along with 0.1 M of malic acid assisted HAp particles in MiliQ water. 2.0 M of NaCl (Merck, 99.8% purity) was added to the solution in order to enhance the conductivity of the electroplating bath. The deposition was carried out at a current density of 50, 100, and 200 mA/cm2 for 30 min. All coatings were deposited at room temperature. Excessive hydrogen evolution at the cathode occurs if the deposition voltage exceeds 1.23 V. This may lead to over-porosity in the coatings, which may create adhesion issues of the coating. Therefore, the electrodeposition voltage was maintained below 1.23 V.
Material characterization of the HAp-brushite composite coatings
The phase purity of the composite coatings was analyzed by X-ray diffraction (XRD) (Rigaku SmartLab diffractometer). A Cu target was used with CuKα radiation (with a wavelength of 0.15406 nm). Two theta range between 10° and 50° was scanned with a step size of 0.02° and scanning rate of 0.05° per second. The crystallinity of the coating surface was measured from the obtained XRD patterns, using the modified Scherrer equation:

Where β is the full width half maximum (FWHM in radian), K is the Scherrer constant (taken as 0.94 here), λ is the wavelength of the X-ray radiation, θ (radian) is the angle of diffraction, D is the interplanar spacing, and ε is the rms strain present in the sample. Because of peak broadening due to the instrumental errors, the accurate value of β is taken per the following formula:

Where B is the actual measured value from XRD pattern of the peak and b is the full width at half maximum for the Si single crystal from the same 2
The microstructure of the composite coatings at different current densities was characterized by field emission scanning electron microscope (FE-SEM) (Hitachi SU9000). Fourier transform infrared spectroscopy (FTIR) was carried out to confirm the presence of phosphate group in the coated layer. FTIR was carried out using Nicolet 6700 spectrometer in the range of 4000–400 cm−1, with a resolution of 8 cm−1 using the KBr pellet method. The hardness of the coatings was measured using nanoindentation method. A constant load of 2.2 mN was applied with a dwelling time of 10 s. All the experimental measurements were carried out at around 25°C.
Hemocompatibility assessment
As per the guidelines laid down by ASTM F 756-00, the hemolytic assay of the samples was conducted in sterilized physiological saline. Coated samples were immersed in saline solution and incubated for 12 h at body temperature that yielded test extract. In order to remove any particulate matter from the same and to ensure only dissoluted components from the coating remain in in-vivo conditions, the solution was subjected to centrifugation, and the resulting supernatant was used as a test extract. 35 Fresh human blood was collected using liquid EDTA as an anticoagulant from a volunteer and diluted in a 1:5 ratio with physiological saline itself. The diluted blood was added to the prepared test extract and set for incubation at body temperature for an hour. Positive and negative controls were taken as Distilled water and Saline, respectively, and the incubated Blood-Test extract was subjected to absorbance measurement in a UV-Vis scanning spectrophotometer (Shimadzu UV-1800) at 545 nm wavelength. 36 The acquired data was tabulated, and Hemolysis ratio (Z) was calculated using the following formula:

where
Cytotoxicity assessment
Osteosarcoma cell lines MG63 collected from The School of Life Sciences and Biotechnology (SLSB) of Shanghai Jiao Tong University (SJTU) were cultured in DMEM with antibiotic and antimycotic solutions and fetal bovine serum procured from Servicebio Ltd. China. Test extracts as prepared for Hemolytic studies were used to assess the cytotoxicity levels, wherein different volumes (25, 50, 100 µg/ml) were used to determine the optimized release concentration of the samples based on weight measurements of metal samples in coated and uncoated states. Weight measurements of respective coated samples as bare and coated states were used to determine the volume of media to meet weight/volume concentration of test extracts mentioned above. Cultured cells were washed with PBS and incubated with test extracts for 24 h in a serum-free media at 1 × 104 cells/well with cells treated with sterilized physiological saline serving as medium for test extracts as control and undergoing similar treatments as test extracts throughout. The medium was aspirated after the incubation period and treated with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) prepared in PBS and further incubated for 4 h, followed by washing of the cells with PBS. 37 The as-formed crystals that turned purple were dissolved in dimethyl sulfoxide (DMSO), and absorbance was measured at 570 nm. Collected data was put to the following equation, and relative cell viability was measured.

Results and discussion
Phase analysis of coating
Figure 1(a) represents the X-ray diffraction patterns of the composite coatings deposited at various current densities. The XRD of the electrodeposited surface is in good agreement with the standard data of brushite and HAp. Peaks of brushite and HAp can be clearly seen. Therefore, the formation of a HAp-brushite composite layer is confirmed. The coatings deposited at 200 mA/cm2 show high-intensity HAp peaks indicating the more amount of HAp presence in the coating compared to the coatings deposited at 100 and 50 mA/cm2. The average crystallinity of the coatings deposited was found around 71%. The proposed electroplating protocol delivers a mixture of hydroxyapatite and brushite in the resulting coatings instead of a pure hydroxyapatite coating. According to literature, pure hydroxyapatite coatings obtained via electroplating route show relatively inferior adhesion to the underlying metallic substrates. 38 In contrast, we observed that the presence of brushite phase along with the hydroxyapatite phase improved the adhesion of the coatings significantly. This also does not affect the purpose of this technology as the brushite phase slowly transforms into hydroxyapatite when it comes in contact with body fluids. Similar observations are already studied and documented in literature.14,15 Therefore, we allowed some amount of brushite phase to be present along with the hydroxyapatite phase to ensure that the adherence of the coating to the underlying metallic substrate is maintained.

(a) XRD pattern of the electrodeposited sample at various current densities (50, 100, and 200 mA/cm2) shows the presence of both brushite and HAp in the coating for all three depositions. ICDD No. brushite (01-072-0713) and HAp (00-009-0432) and (b) load (mN) versus penetration depth (µm) curve for the coatings deposited at various current densities (50, 100, and 200 mA/cm2).
Hardness and coating adhesion
The hardness of the coatings was measured using nanoindentation method. All the coatings were exposed to a constant load of 2.2 mN and a dwelling time of 10 s. This was repeated three times on each sample to get the average values. The samples deposited at higher current density showed higher resistance to the load and had higher hardness. Figure 1(b) shows the load versus penetration depth curve for the three samples. The adhesion strength of the samples was checked by a cello-tape pull-out test. The sample deposited at 200 mA/cm2 had good adhesion strength, and the coating remained intact with the substrate upon pulling out the cello tape. The samples deposited at 100 and 50 mA/cm2 failed to pass the test. Therefore, the structural and biological characterization of the sample deposited at 200 mA/cm2 was only carried out.
Morphology of coating
The SEM micrographs of the HAp-brushite composite coating deposited at 200 mA/cm2 are shown in Figure 2. The observed composite layer has been found to be less crowded and has a porous, spongy structure. The porous structure is clearly witnessed at higher magnification (Figure 2(b)). Figure 2(c) shows the corresponding EDS spectrum. The presence of Ca, P, and O are evident, and they are present in major quantities. Some traces of Ca2+ and PO43− is also found.

(a) BSE SEM micrographs of the coating deposited at 200 mA/cm2 showing a porous structure, (b) coating morphology at higher magnification, and (c) EDX spectrum of the coating.
FTIR and Raman spectroscopy
The FTIR spectra of coating deposited at 200 mA/cm2 are shown in Figure 3(a). It shows the various absorption regions. The bands at ((i) in insert) 3143 and (ii) 668 cm−1 are originated from

(a) FTIR spectra of the coating deposited at 200 mA/cm2 showing various absorption regions and (b) Raman spectrum of the coating deposited at 200 mA/cm2.
Hemolytic assay
Hemolysis, an indication of the destruction of red blood cells (RBCs), can lead to Anemia or Jaundice that makes its assessment against the prepared coatings necessary. 39 The absorbance values dependent on the changes in color intensity show negligible hemolysis as a virtue of the excellent biocompatibility of HAp coatings. 40 The additional layer of coatings forms a physical barrier for the corrosive free ions such as Cl− to come in contact with the implant surface, thereby preventing physiological leech out of toxic components from the alloy. However, a slight but still biocompatible increase was found when coated alloys were subjected to prolonged immersion in physiological saline-based test extract (Table 2). Such long-term exposure enables fluids to seep through porous coatings given time, and hence the additional components from the alloy affect overall favorable levels of hemolytic activity. Optical micrographs confirm the claims made over absorbance-based measurements and show proper structural integrity of around 80% visible RBCs (Figure 4). A few damaged cells were also observed that could be justified given less than ideal in-vitro working conditions as compared with in-vivo environments, which has also led to agglomeration of cells at preferable areas over slides. 41 Porosity is an essential feature of bioactivity as it increases the available surface area not just for fluids such as saline but rather a cocktail of other components making up the body fluid which is almost impossible to replicate in vitro. Hence it can be stated successfully that even with prolonged exposure, the coatings have hemocompatible features that are necessary for an implant as blood is the most important connective tissue, which not only transports necessary enzymes to bone regeneration sites but also flushes out metabolic by-products.
Absorbance and hemolytic ratio of coated sample calculated with respect to OD values.


Micrographs of blood samples treated with test extracts after (a) 6 h, (b) 12 h, (c) 24 h, and (d) 48 h.
Cytotoxicity evaluation
Quantifying the cell viability using MTT assay has been elaborated in Figure 5. Human osteosarcoma cell lines were used to assess the toxic nature of coated implants over healthy cells. The assay gives an idea about how the material will interact with healthy cells under given circumstances in-vitro. Assessment of percent viable cells is performed over the change in color intensity of purple formazan from blue colored MTT. The change in color is by virtue of treatment of the cells, but the sustenance is dependent on live and healthy cells as only live cells can uptake and maintain formazan; hence the color intensity can be established as a parameter of healthy cells measured against a UV spectrophotometer. 42 The absorbance and the cytotoxicity of the coated sample after a different time frame of treatment at different concentrations are presented in Table 3. It can be seen that most of the samples show favorable cell proliferation rates, but the high mass to solution ratio favors it the most. Within the first 12 h of treatment, the cells proliferate at the highest rate ever with an increase in release concentration as HAp is readily available to cells to stimulate their proliferation levels. An increase in treatment time further elevates the proliferation levels, which faces a slight setback with solutions containing low mass to volume ratio wherein it is speculated that ionic components from the alloy are leeched out in the test extract thereby affecting cell growth. Even with such a slight setback at higher release rates, the overall cytotoxicity levels are well within admissible levels, confirming positive biocompatibility of the coatings in-vitro.

Hemolysis % of blood samples treated with test extracts after 6, 12, 24, and 48 h.
Absorbance and percent cytotoxicity of the coated sample after a different time frame of treatment at different concentrations.

A qualitative assessment of the above results was performed to check morphological features of cells by observing the same under a digital microscope. The presence of interfering mediums reduced proper visibility that called for the assistance of color filters to view structural features of the cells (Figure 6). All the samples showed intact structural morphology of MG-63 cell lines, which can be attributed to metabolic stimulation by Hydroxyapatite residues released in the test extract. The micrographs were used only to confirm any kind of structural damage to cells. The methodology used to place cells over the observable platform is such inconsistent that cell density observed in the images cannot be taken into account of the proliferation levels. With an increase in treatment time, as discussed earlier, the porous nature of coatings allows the introduction of toxic components from the alloy to come in contact with the cells that cause reversible damage to the same as observed in Figure 7(h) and (i). These changes are brought by the compositional change in environment by those free ions in the form of pH fluctuation or deactivation of a cell wall structure and morphology by change in protein motive force or alteration over binding sites. 43 However, the changes do not seem apoptotic and can be recovered in the due process hence supporting the quantitative claims of biocompatibility against MG-63 cells.
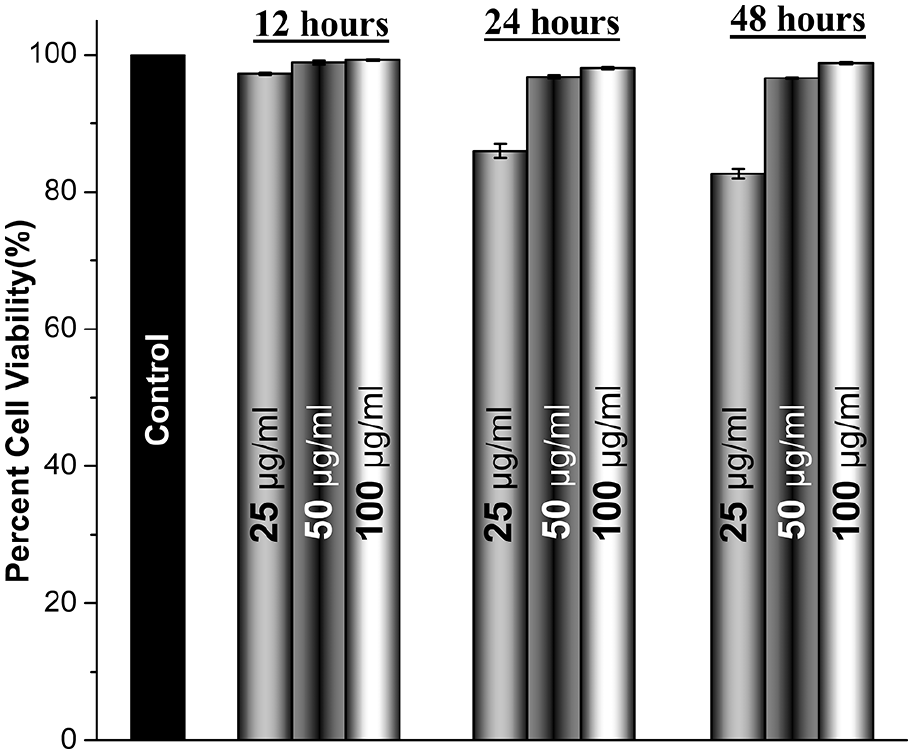
Cytotoxicity of the coated sample after a different time frame of treatment at different concentrations.

Optical micrographs of MG-63 cell lines after 12 h: (a) 25 µg/ml, (b) 50 µg/ml and (c) 100 µg/ml, 24 h (d) 25 µg/ml, (e) 50 µg/ml, (f) 100 µg/ml) and 48 h (g) 25 µg/ml, (h) 50 µg/ml and (i) 100 µg/ml.
Conclusion
A composite coating of HAp and brushite is deposited successfully on stainless steel surface via pulsed electrodeposition route. The electroplating of the HAp-brushite composite coating is carried out in the presence of malic acid extracted from commercially available apple (
