Abstract
Introduction
Electrolytic manganese dioxide (EMD) production is a well-established industrial process for obtaining high-purity manganese dioxide, a material of significant importance across various technological and environmental applications. 1 Its superior electrochemical properties make it particularly suitable as a cathode material in both alkaline (AA) and lithium-ion batteries. 2 Beyond energy storage, manganese dioxide is widely utilized in water purification systems, chemical synthesis, and as a catalyst in organic reactions.3–5 In light of the growing global demand for sustainable energy technologies and the rapid expansion of battery manufacturing, the pursuit of more efficient and environmentally friendly methods for EMD production has emerged as a critical research priority.6,7
The conventional electrolytic production of manganese dioxide relies on the anodic oxidation of manganese sulphate solutions. Although effective, this process demands substantial energy input and generates waste by-products, posing environmental and economic challenges. 8 An effective approach to bolster the sustainability of EMD production entails integrating the regeneration of ferrous iron (Fe2+) into the electrochemical system. By simultaneously oxidizing manganese sulphate to manganese dioxide and regenerating Fe2+ at the cathode, overall process efficiency can be improved while reducing energy consumption and waste generation.9–11 Recent studies have demonstrated that this integrated approach not only minimizes operational costs but also supports resource recovery and aligns with broader goals of sustainable hydrometallurgical processing.9,12 These advancements underscore the importance of developing low-cost, environmentally friendly technologies for manganese recovery, particularly in response to the growing demand for battery-grade materials and the depletion of high-grade manganese ores.
The regeneration of Fe2+ during EMD production presents several notable advantages in terms of process efficiency, resource utilization, and environmental sustainability. This integrated approach enables the production of high-purity EMD while simultaneously regenerating Fe2+ at the electrowinning stage. The regenerated ferrous iron, combined with sulphuric acid effluent, is recycled and employed as a reducing agent in the subsequent leaching step. Ferrous iron itself is a widely utilized reductant in various industrial applications, including wastewater treatment, chemical synthesis, and metallurgical operations. By regenerating Fe2+ in situ, the process minimizes reliance on external reductant sources, thereby reducing operational costs and conserving natural resources. Furthermore, the incorporation of iron regeneration enhances the energy efficiency of the EMD process by harnessing electrons from the cathodic reduction of Fe³+ to drive the anodic oxidation of manganese. This synergistic electrochemical interaction contributes to lower overall energy consumption, reinforcing the sustainability of the production method.
The successful integration of Fe2+ regeneration into the EMD production process is contingent upon several critical factors, including the selection of electrode materials, membrane properties, electrolyte composition, and operational parameters. Advances in membrane technology, particularly the development of materials exhibiting high ionic conductivity and chemical stability, have demonstrated considerable potential in improving the efficiency and durability of electrochemical systems. Furthermore, precise control over electrolyte composition and key operating conditions, such as temperature, pH, and current density, is essential for optimizing both the yield and purity of manganese dioxide and the effectiveness of Fe2+ regeneration. Collectively, these considerations play a pivotal role in enhancing the overall performance and sustainability of the EMD process.
Despite its potential advantages, the integration of Fe2+ regeneration into the EMD production process poses several technical challenges. A primary concern is the management of side reactions arising from cross-contamination between the cathodic and anodic compartments during electrolysis, which can compromise product purity and process efficiency. Additionally, maintaining the chemical stability and operational longevity of membrane materials under harsh electrochemical conditions is critical for sustained performance. Overcoming these challenges will minimize energy consumption while ensuring high-quality MnO2 output.
The production of EMD from various sources has been investigated in recent studies. Roriz et al. 13 explored using depleted batteries as a manganese source, finding optimal current densities between 102 and 139 × 10−3s A/m² for EMD production. The EMD produced in all the experiments had the same allotropic form, the α-MnO2 variety. Song et al. 14 proposed a single-membrane double-chamber electrolysis method, simultaneously producing manganese metal and EMD while recovering sulphuric acid. The study achieved high yields of manganese dioxide (75.1%) and electrolytic manganese dioxide (23.6%), as well as a high recovery of sulphuric acid (62.3%) and relatively low energy consumption (5701 kW h t−1). The manganese metal produced at the cathode had a smooth, dense surface with a silver-white metallic luster and uniform growth. The EMD produced at the anode was α-MnO2 with a spherical particle structure and a uniform particle size with a honeycomb structure. Buzatu et al. 15 developed an electrolytic process for simultaneous production of high-quality EMD and commercial-grade zinc from used battery leachates. The paper looks at how the efficiency of the MnO2 deposit changes with anodic current density and how the concentration of sulphuric acid affects the zinc metal and the MnO2 deposit. The results of the study allowed the identification of optimal conditions for the electrolytic process. Önal et al. 11 presented a hydrometallurgical route proposed to valorize waste furnace dust for the production of battery-grade MnO2. Dextrin, a cheap organic reductant, is used to enable the direct and complete dissolution of manganese in the furnace dust without the need for high-temperature pre-reduction. Changing the pH of the leachate cleans it up, and then it goes through direct electrowinning to make electrolytic manganese dioxide (EMD). An overall manganese recovery rate of >90% is achieved. Biswal et al. 1 presented EMD as a critical component of the cathode material in modern alkaline, lithium, and sodium batteries, including electrochemical capacitors and hydrogen production. In terms of environmental and cost considerations, EMD is likely to remain the preferred energy material for the future generation, as it has been in recent decades. These studies demonstrate various approaches to EMD production, utilizing different raw materials and process configurations. While not explicitly addressing ferrous iron regeneration, these papers highlight the potential for resource recovery and process optimization in EMD production.
In alignment with the United Nations Sustainable Development Goals (SDGs), Binnemans and Jones 16 introduced the concept of circular hydrometallurgy, proposing twelve foundational principles aimed at enhancing the sustainability of hydrometallurgical operations. These principles emphasize key strategies such as reagent regeneration, waste minimization, process efficiency optimization, and the integration of material and energy flows. This framework is consistent with broader circular economy models, which, Suárez-Eiroa et al. 17 argue, should be grounded in the principles of circularity, sustainability, and systemic resilience. Rao 18 emphasizes the critical role of resource recovery and recycling from metallurgical waste streams as a fundamental aspect of circular hydrometallurgy, focusing on practices like metal reclamation, slag utilization, and the recycling of water and chemical reagents. Complementing these applied strategies, Burkin 19 outlines the fundamental chemical principles that govern hydrometallurgical processes, including aqueous equilibria, reaction thermodynamics, and leaching kinetics. Together, these scientific and strategic approaches provide a comprehensive foundation for developing environmentally responsible and resource-efficient metallurgical technologies.
This study investigates the production of EMD production of manganese dioxide in conjunction with the simultaneous regeneration of Fe2+, with a particular emphasis on the underlying electrochemical mechanisms, process optimization strategies, and the functional role of membrane materials. The investigation includes a systematic analysis of key operational parameters, such as current density, electrolyte composition, and cell configuration, and their impact on process efficiency and product quality. Furthermore, the scalability of the proposed method for industrial implementation is critically assessed. The findings aim to advance the development of sustainable and energy-efficient techniques for MnO2 synthesis, thereby contributing to the broader fields of green chemistry and resource recovery.
Conventional production of EMD
A typical process for the production of EMD from MnO2 or other mineral resources containing manganese includes a reductive roasting if MnO2 ore is used followed by sulphuric leaching and purification steps as presented in Figure 1. 8

Conventional process for the production of electrolytic manganese (EM) and electrolytic manganese dioxide (EMD) 8 .
At pH 3–4, sulphide precipitation typically removes other impurities like Ni, Co, Cu, and Zn while hydroxide precipitation typically removes iron (III). The resultant solution can be used for the production of electrolytic manganese.
Lime is used instead of potassium hydroxide to avoid contamination of the electrolyte by potassium. If potassium is present in the ore, it should be removed. It is necessary to remove potassium in order to make electrolytic manganese dioxide, since potassium helps it form during electrolysis but does not make it battery-active. This is usually achieved by precipitation of jarosite or alunite in the leaching process, normally by addition of ferric sulphate.
8
Equation 1 can represent a jarosite precipitation reaction:
Harris et al. 20 put this procedure into practise in South Africa after Fuller et al. 21 had first developed it. The plant operated at a rated capacity of 16500 t/year at current efficiencies of approximately 68%. The typical plant conditions required for the electrolysis process are summarised in Table 1. 8
Plant conditions for the electrolysis of manganese sulphate.

The process described above is only profitable when using high-grade manganese ores. However, for the manganese ores containing less than 40% Mn and Mn-waste sources, a process treatment was recently developed by Hi-Tech Energy Ltd. 8 In this process, the ore is leached with an acidic solution at a temperature below 60°C with strict control of the potential for the addition of gas and the addition of manganese to minimise the generation of dithionate ions. The manganese in the leach solution was extracted using a carboxylic acid, at pH 5.5–6.5 followed by stripping with spent aqueous H2SO4 electrolyte to obtain MnSO4 feed solution for electrowinning to produce EMD. A schematic diagram of the improved process II vs. the conventional process I is shown in Figure 2.

Process flowsheet for low-dithionate acid leach process 8 .
In the production of EMD according to the process used by Tekkosha in Greece, 22 the process usually starts with ore containing manganese dioxide, according to Figure 3, which is first reduced to MnO with either coal, heavy fuel oil, hydrogen, or natural gas. The MnO is then leached with sulphuric acid to form a manganese sulphate solution. Alternatively, if rhodochrosite (manganese carbonate) is the starting material (as in Japan), only a leaching step with sulphuric acid is required. The acidic manganese sulphate solution is then purified by oxidation with MnO2 and neutralized with lime to a pH of 4–6. This treatment precipitates any heavy-metal ions present (Fe, Pb, Ni, Co). Hydrogen sulphide may be added for complete purification. After solid-liquid separation and adjustment of concentrations to 0.50 −1.06 M of MnSO4, the manganese sulphate solution is subjected to electrolysis, which raises the acid content to 0.50 −1.00 M.

Production of EMD according to the process used by Tekkosha, Greece. 22
The typical plant process conditions required for the electrolysis process according to the Tekkosha process are summarised in Table 2.
Electrolysis of manganese sulphate according to the Tekkosha process 22 .

Electrolysis of manganese dioxide with regeneration of ferrous iron
Ntunka and Loveday 10 have developed a conceptual design of the electrolysis circuit for producing MnO2 with regeneration of ferrous iron. The electrowinning was designed to operate in bags with pure manganese sulphate inside the bags at the anode and Fe3+ at the cathode. Operating the plant in closed circuit with recycles helps to achieve this. Instead of the hydrogen evolution reaction at the cathode, Fe3+ is reduced to Fe2+. The acid produced at the anode permeates through the membrane bags and is mixed with the catholyte solution. Operating in this manner significantly reduces power consumption during electrolysis and produces a by-product Fe2+ and H2SO4 to be used as leaching reagents.
The purified solution is introduced into the bag, which constitutes the anode compartment. The spent anolyte will permeate through the membrane, be mixed with the spent catholyte, and be recycled back to dissolve the manganese ore at the leaching stage.
The main electrochemical reactions are as follows:
Main anode reaction:
Main cathode reaction: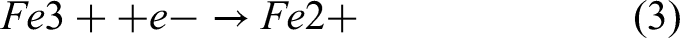
As the electrolysis of MnO2 proceeds only at about 90% current efficiency for modern plants, the following side cathodic reaction can occur:
The side cathodic reaction's production of OH− increased the possibility that pH control in the cathode compartment would fail. A loss of pH would cause Mn precipitation on the cathode surface as Mn(OH)2. However, the catholyte is acidic enough in the proposed flowsheet to prevent any loss of pH. A simple schematic representation of the electrolysis section can be seen in Figure 4. The temperature is kept at around 90–98°C. As it can be expected, there will be evaporation in the electrolysis compartments.

Proposed block diagram of electrolytic manganese dioxide production. 10
The electrolysis circuit designed by Ntunka and Loveday 10 demonstrates an innovative approach to producing MnO2 while regenerating ferrous iron. This process could potentially improve efficiency and reduce costs in manganese production. The process operates in bags with pure manganese sulphate and Fe3+, reducing power consumption and producing a by-product for leaching. The electrowinning process involves main anode and cathode reactions, with pH control in place.
Material and methods
Electrolysis tests
Design of the electrowinning experimental setup for the production of EMD
Electrolytic cell for the production of MnO2
A divided electrolytic cell was employed in all experiments to ensure optimal separation of the anodic and cathodic reactions. The anodic compartment housed a lead electrode, while the cathodic compartment contained a carbon electrode mounted on a stainless-steel rod. The use of a divided cell was crucial for preventing cross-contamination between the compartments, thereby maintaining the high purity of the produced MnO2. This setup ensured that oxidation and reduction reactions occurred independently, allowing for precise control of electrochemical conditions and minimizing undesirable side reactions that could compromise product quality and process efficiency.
Furthermore, the divided configuration facilitated efficient regeneration of ferrous iron at the cathode while simultaneously promoting MnO2 deposition at the anode. This dual-functionality not only improved resource recovery but also optimized the overall energy and material efficiency of the process. The cell design closely simulated industrial conditions used for the electrowinning of nickel metal from nickel sulfate solutions, thereby enabling accurate assessment of electrochemical behavior and energy consumption under controlled laboratory conditions.
Although EMD is commonly produced industrially using titanium anodes—primarily due to the ease of stripping the final product—initial trials with titanium in this study were unsuccessful due to passivation. As a result, lead anodes were chosen, following precedent set by other researchers, to demonstrate the core benefits of the electrochemical process without the complications introduced by titanium passivation.
Polarization curve measurements were conducted using a three-electrode setup, consisting of an Ag/AgCl (3 M KCl) reference electrode and a Luggin capillary positioned near the anode to ensure accurate potential readings. Energy consumption tests were carried out using a similar configuration, with anodic and cathodic voltages monitored through a specialized three-divider cable supplied by Metrohm.
The experimental electrolytic cell was constructed using a 600 mL beaker filled with a catholyte composed of a ferrous-ferric sulfate solution. The anolyte compartment, fabricated from PVC and sealed on one side with finely woven PVC cloth, was filled with a pure electrolyte solution (see Figure 5). This compartment was carefully inserted into the beaker so that the anolyte level was maintained approximately 1 cm above the catholyte level. This arrangement created a gentle flow of anolyte through the cloth, preventing back-diffusion of impurities such as Fe2+ and Fe³+ into the anodic compartment. Additional anolyte was added as needed to sustain the level differential.

Electrolytic cell with anodic and cathodic compartments.
This design effectively minimized contamination of the anolyte, thereby preserving process efficiency and reducing energy losses. The cell's structure was intended to replicate one side of a diaphragm or bag-type system used in nickel electrolysis, particularly when processing impure solutions containing ferrous ions.
The catholyte solution contained 0.25 M of Fe3+ in 0.5 M H2SO4, was fed into the beaker (cathodic compartment). A 1 M solution Mn2+ prepared in 0.5 M H2SO4 was used for the anolyte. Partly spent anolyte solution entered the cathodic compartment through the diaphragm cloth. The cathodic compartment held about 360 cm3 of catholyte solution and the anolyte compartment held about 80 cm3 of pure manganese sulphate solution.
Since the temperature of the cell solution is an important operating variable, the beaker was suspended in a waterbath fitted with a temperature controller.
Electrowinning experiments
The electrowinning process for producing EMD was tested as an electrolytic batch cell in bags. The system is illustrated in Figure 6. The electrolyte that has been cleaned up and contains ions goes through the main anodic reaction with a plating of inside bags. These bags keep out impurities, especially Fe2+ ions, but let the current flow through them. Figure 7 is a photograph of the frame that was used to support a polyethylene cloth, which had a suitable porosity to allow a small flow of anolyte containing the sulphuric acid produced on the anode, and some Mn2+ ions permeate into the cathodic compartment where the reduction Fe3+ into Fe2+ occurred. The level of anolyte was about 1 cm higher than the catholyte in order to avoid impurities from the catholyte migrating inside the bags and being oxidised.

Schematic diagram of the electrowinning of MnO2 in bags.

Schematic diagram of the polarization experiments.
The electrolytic cell was fitted with lead anodes and graphite cathodes. A three-electrode setup was used for the electrowinning tests, powered by a METROHM PGSTAT as power supplied.
The experimental conditions for the electrowinning tests are summarized in Table 3.
Experimental conditions for the electrolysis test.

Polarization experiments
Polarization curves were plotted by the linear, cyclic voltammetry method using a three-electrode setup on the same PGSTAT 302 (Autolab Eco Chemie B.V., Netherlands) coupled with a saturated silver/silver chloride reference electrode (Ag/AgCl) and a Luggin capillary. Using a unique 3-Divider cable that Metrohm SA supplied, it was also possible to measure the potential of the counter electrode in relation to the working electrode.
The anodic potential was changed from 0 to 2.0 V (vs. Ag/AgCl) at a rate of 20.0 mV/s, and the transient current response was recorded by a computer connected to the PGSTAT302 via NOVA software interface. The anode was made of lead, and the cathode was in lead. Similar to the electrolysis experiment, the reference electrode was placed inside a Luggin capillary closer to the cathode. The polarization tests were conducted in a water jacket vessel heated by a temperature controller connected to a water bath. The vessel was clamped on a retort and was seated on a hot plate with a magnetic stirrer. A schematic diagram of the polarization setup is shown in Figure 7.
The electrowinning tests of EMD were conducted using a divided electrolytic cell with a lead electrode in the anodic compartment and a carbon cathode on a stainless-steel rod in the cathodic compartment. The electrolytic cell was designed to mimic one side of a bag, as used for electrolysis of nickel using impure solution containing ferrous ions. The electrowinning process was tested as an electrolytic batch cell in bags, with the electrolyte cleaned up and containing ions going through the main anodic reaction with a plating inside bags. Polarization curves were plotted using a three-electrode setup on a METROHM PGSTAT 302 coupled with a saturated silver/silver chloride reference electrode and a Luggin capillary. The energy consumption tests were conducted using a similar configuration, monitoring anodic and cathodic voltage using a special 3-Divider cable supplier by Metrohm.
Results and discussion
X-Ray diffraction (XRD) analysis of manganese dioxide product
XRD analysis verified that the synthesized manganese dioxide (MnO2) was of high purity and exclusively composed of the pyrolusite phase (blue line). The diffractogram, acquired using a PANalytical EMPYREAN XRD instrument equipped with a cobalt radiation source operating at 40 kV and 40 mA, exhibited peak patterns corresponding solely to MnO2. No secondary phases, such as other oxides or sulfates, were detected (Figure 8), confirming the successful formation of a single-phase MnO2 product with high structural integrity.

XRD patterns of the electrolytic manganese dioxide product.
Polarization behaviour
The polarization curves presented in Figure 9 illustrate the electrochemical behavior of both anodic and cathodic processes within the system. The anodic polarization curve demonstrates a marked increase beginning at approximately 0.8 V, followed by a plateau between 2.5 and 3.0 V. This plateau region is indicative of a limiting current condition, primarily governed by mass transfer constraints associated with the oxygen evolution reaction (OER), rather than by kinetic limitations alone.

Comparison between polarization curves of the anodic oxidation of Mn2+ and the cathodic reduction of Fe3+ and H+, respectively.
In contrast, the cathodic polarization curves exhibit negative current values characteristic of reduction reactions. When considering the reduction of ferric iron (Fe3+), the curve deviates from that of the hydrogen evolution reaction (HER), particularly at cell voltages below 1.4 V. Within this voltage range, the regeneration of ferrous iron (Fe2+) is favored due to its lower energy requirement compared to HER. Nonetheless, the presence of significant hydrogen evolution in this region suggests competitive interactions between Fe3+ reduction and HER. At elevated current densities corresponding to voltages between 1.5 and 2.5 V, the anodic polarization curve reflects enhanced OER activity, consistent with the observed increase in cell voltage.
For the proposed electrolysis process, the polarization curves of Mn2+ oxidation in yellow and brown are shown in Figure 9.
Experimental cell voltage for the electrolysis of MnO2 with membrane
It is well known that cell voltage during the electrolysis of MnO2 is influenced by several key factors, including electrode material selection, current density, electrolyte composition, and the inter-electrode gap. Pilot-scale experiments conducted at MINTEK by Te Riele 23 demonstrated that current efficiencies ranging from 85% to 95% could be achieved by adjusting the cell voltage between 2.1 and 2.4 V. These tests utilized graphite rods and various titanium anodes, along with electrolytes containing 0.5–1.5 M manganese and 0.5–1 M sulphuric acid. 23 These findings highlight the necessity of optimizing electrode configurations and operating conditions to enhance energy efficiency and process performance. Future research should focus on mitigating mass transfer limitations to further improve overall system efficiency.
The cathodic polarization curves presented in Figure 9 reveal negative current values, indicative of reduction reactions. At low current densities, the polarization behavior for Fe3+ reduction diverges from that of the HER, suggesting that Fe2+ regeneration occurs preferentially at lower cell voltages. Specifically, when the applied voltage falls below 1.4 V, Fe2+ regeneration is accompanied by significant hydrogen evolution, indicating competitive interactions between the two cathodic processes. This overlap implies that Fe2+ regeneration is not the sole reaction occurring under these conditions, as HER contributes substantially to the observed current.
Overall, the polarization data suggest that the incorporation of Fe2+ regeneration alters the electrochemical dynamics relative to conventional MnO2 electrowinning. The distinct polarization profiles highlight the potential of Fe2+ recycling to improve process efficiency and reduce energy consumption, offering a promising pathway for more sustainable and cost-effective manganese dioxide production.
Comparison between cell potential of the conventional electrolysis and the electrolysis with the regeneration of Fe2+
As illustrated in Table 4, the cell voltage during MnO2 electrolysis is influenced by the process configuration, specifically whether Fe2+ recycling is incorporated at the cathode. Compared to the conventional method, the proposed approach operates at a lower current density, below 30 A/m², rather than the optimal range of 65 to 108 A/m² reported by. 24 Under these conditions, the modified process achieves a reduced cell voltage, resulting in lower energy consumption and operational costs. Moreover, the regeneration of Fe2+ at the cathode provides an in-situ reductant for the leaching stage, enhancing overall process efficiency. This dual benefit, cost reduction and improved resource utilization, demonstrates the economic and technical advantages of integrating Fe2+ recycling into MnO2 electrolysis, offering a more sustainable alternative to traditional electrowinning methods.
Comparison between cell potential of the conventional electrolysis and the electrolysis with the regeneration of Fe2+.

Voltage savings diminish as current density increases. While operating at higher current densities can reduce capital costs by enabling smaller cell designs and faster production rates, lower current densities and cell voltages are generally more energy-efficient. This is primarily due to the suppression of competing secondary reactions, such as the OER at the anode and the HER at the cathode, which become more pronounced at elevated voltages. As previously demonstrated by, 23 optimal performance, characterized by enhanced energy savings and high current efficiency, can be achieved at a current density of 100 A/m² and a temperature of 90 °C, irrespective of the anode material used. These findings highlight the importance of balancing operational intensity with electrochemical efficiency to optimize process economics and sustainability.
Electrode potentials versus time for the deposition of EMD
A controlled electrolysis experiment was conducted to investigate the temporal evolution of electrode potentials during the deposition of EMD and to assess the quality of the manganese oxide formed at the anode. The test was performed at a constant current density of 30 A/m², and the resulting electrode potential profiles over time are presented in Figure 10. The electrochemical cell comprised a lead anode and a carbon cathode, with the anodic compartment containing an electrolyte solution of 1 M MnSO₄ and 0.5 M H2SO₄, while the cathodic compartment was filled with 0.25 M Fe2(SO₄)₃. The experiment was conducted at a temperature of 25 °C. The potential of the working electrode (anode) was measured against a Silver/Silver Chloride reference electrode (Ag/AgCl, 3 M KCl), and the counter electrode (cathode) potential was recorded relative to the working electrode using the ADC164 module of the PGSTAT Autolab system.

Electrode potentials vs. time for the plating of EMD onto lead anode and carbon cathode in the divided electrolytic cell at 25 °C.
During the electrodeposition of MnO2, the anodic plating potential remained relatively stable at approximately 1.60 V. Similarly, the cathodic potential associated with Fe2+ regeneration was consistently maintained at around 0.045 V relative to the anode. The observed anodic potential closely aligns with the theoretical value predicted by the Nernst equation (Equation 6). However, discrepancies between the experimental and theoretical electrode potentials can be attributed to the influence of various overpotentials, including activation, concentration, and ohmic losses, as well as the use of molar concentrations rather than thermodynamic activities in the calculations. These factors contribute to deviations from ideal electrochemical behavior under practical operating conditions.
The application of the Nernst Equation to the anodic reaction is represented in Equation 6.
Equation 6 can be employed to predict the variation in anodic potential during the electrolytic deposition of MnO2, particularly as Mn2+ ions are progressively depleted from the electrolyte. Assuming equimolar activities of Mn2+ and H+ ions at the onset of electrolysis, the logarithmic term in Equation 6 effectively cancels, resulting in an anodic potential approximating the standard electrode potential
Figure 10 illustrates a slight decrease in anodic potential toward the end of the electrolysis period, suggesting dynamic changes in the electrochemical environment. It is important to note that variables such as manganese concentration, acid concentration, electrolyte impurities, and diaphragm characteristics were not examined in this study. Considering the reference potential of the Ag/AgCl electrode is 0.1976 V above the standard hydrogen electrode at 25 °C, the observed anodic potential of 1.60 V is reasonable. The deviation from the theoretical value of 1.280 V can be attributed to cumulative overpotentials, including activation, concentration, and ohmic losses.
Regarding the cathodic process, the standard potential for the Fe3+/Fe2+ redox couple is 0.77 V versus the standard hydrogen electrode. Equation 7 applies the Nernst equation to this cathodic reaction. The experimentally measured cathodic potential was approximately 0.045 V relative to the working electrode. Assuming equimolar activities of Fe2+ and Fe3+, the cathodic potential should align closely with the standard value; however, the observed deviation is primarily due to the overpotential associated with the reduction of Fe3+ to Fe2+ at the graphite cathode.
The experimental cathodic potential is reading 0.045 V with respect to the working electrode potential. In the same way, assuming equimolar activities of both Fe2+ and Fe3+ will lead to the cathodic potential approximately equal to the standard electrode potential for the ferric /ferrous couple. The higher value cathodic potential is due to the overpotential of reduction of Fe3+ into Fe2+ at the graphite cathode.
The overall cell voltage is defined as the difference between the anodic and cathodic potentials. Under the experimental conditions of 25 °C and a current density of 30 A/m², the cell voltage for MnO2 deposition coupled with Fe2+ regeneration was approximately 1.55 V, as shown in Figure 9. In contrast, literature reports a cell voltage of 2.32 V for an electrolytic bath containing 0.5 M H2SO₄ and 1.2 M MnSO₄ using graphite anodes and lead cathodes at an average temperature of 77 °C. 24 Comparable cell voltage values have also been documented under similar conditions in more recent studies.23,25 These findings highlight the potential for energy savings through process optimization and the integration of Fe2+ regeneration.
Polarization curve of a conventional EMD electrolyzer
A potentiodynamic polarization curve was recorded using a lead anode and a carbon cathode in an electrolyte solution comprising 1 M MnSO₄ and 0.5 M H2SO₄, without the use of a membrane. The results, depicted in Figure 11, reveal distinct electrochemical regions. The curve exhibits a charge transfer-controlled region between 1.5 and 2.0 V, followed by a mixed control region, governed by both charge transfer and mass transport, spanning 2.0 to 2.5 V. Beyond 2.5 V, the system enters a mass transport-limited regime. Operating the electrolyzer at voltages just below the onset of the mixed control region is recommended to mitigate the occurrence of oxygen evolution at the anode, thereby enhancing process selectivity and energy efficiency.

Linear polarization of the lead anode in 1 M MnSO4 + 0.5 M H2SO4 at 25°C, scan rate 2 mV/s.
Electrolysis of MnO2 with regeneration of Fe2+
Electrolysis experiments were conducted to evaluate the simultaneous production of EMD at the anode and the regeneration of Fe2+ at the cathode. The electrochemical cell was operated at a constant current density of 30 A/m² and a temperature of 25 °C for a duration of two hours. The cathodic compartment contained 360 mL of an electrolyte solution comprising 0.25 M Fe2(SO₄)₃ and 0.5 M H2SO₄, while the anodic compartment was filled with 80 mL of a synthetic solution of 1 M MnSO₄ and 0.5 M H2SO₄. A membrane was employed to separate the catholyte from the anolyte, thereby preventing cross-contamination.
Analytical control tests were performed on both compartments before and after electrolysis to assess changes in Fe2+ concentration and total iron content. Ferrous iron was quantified via titration using potassium dichromate (K2Cr2O₇) as the titrant, and total iron was measured using inductively coupled plasma optical emission spectrometry (ICP-OES). As shown in Table 5, no Fe2+ was detected in either compartment prior to electrolysis. Post-electrolysis analysis revealed that the Fe2+ and total iron concentrations in the anolyte remained unchanged, confirming the integrity of the membrane and the absence of contamination. In contrast, a significant increase in Fe2+ concentration was observed in the catholyte, indicating successful reduction of Fe3+ to Fe2+ at the cathode. Specifically, a 100% increase in Fe2+ concentration and a 10% variation in total iron content were recorded, the latter attributed to sample preparation inconsistencies and calibration errors.
Analytical test control of the catholyte and catholyte content after 2 h electrolysis at 25°C.

The analytical control test results of the catholyte and anolyte for Fe(II) and total iron confirm that there are no ferrous ions present in both the catholyte and anolyte before the electrolysis. After the electrolysis, the anolyte content in Fe(II) and total iron is unchanged. This result is important to note because the experimental setup was designed to prevent contamination of the anolyte. Significantly, Fe(II) was detected in the catholyte compartment, demonstrating the reduction of Fe(III) to Fe(II) at the cathode. It was found that there was a 100% change in Fe(II) and 10% change in the total iron content of the catholyte. The total change in total iron is due to the sample preparation and error due to the calibration of the standard solution.
Additionally, manganese was detected in the catholyte following electrolysis, likely due to the migration of Mn2+ ions through the membrane. The remaining Mn2+ was oxidized at the anode, corresponding to the reduction of an equivalent amount of Fe3+ to Fe2+. Based on the quantity of Mn2+ oxidized, the current efficiency of the cell was calculated to be 67%. For comparison, Qiang et al. 26 reported current efficiencies ranging from 96% to 98% for Fe2+ regeneration in Fenton oxidation processes at an ideal current density of 84.8 × 10−3 A/m². These results provide compelling evidence for the effective production of EMD at the anode and the concurrent regeneration of ferrous ions at the cathode under the tested conditions.
When the cathodic reaction is the regeneration of Fe2+ instead of the hydrogen evolution reaction, the standard voltage of decomposition was much lower. This was established in the theoretical background. We would like to show how the cell voltage may vary with the current density. Here, the cell voltage calculations are presented at a temperature of 25 °C, but the value at a higher temperature can easily be determined.
Significance of the integration of Fe2+ regeneration
The principal innovation of this study lies in the integration of Fe2+ regeneration within the electrochemical framework for EMD production. The incorporation of Fe2+ recycling into the electrolysis process significantly enhances operational efficiency and offers the potential for reduced energy consumption. Electrochemical data indicate that Fe2+ regeneration occurs preferentially at low electrode potentials, preceding the onset of substantial hydrogen evolution. This preferential reaction pathway contributes to energy savings by lowering the overall cell voltage required for MnO2 electrowinning, as demonstrated in comparative analyses between conventional and Fe2+ integrated systems.
This approach addresses a key limitation of traditional EMD production methods, which typically depend on external reductants and often result in environmentally detrimental waste streams. Although previous studies have acknowledged the role of ferrous iron in MnO2 synthesis, the direct electrochemical regeneration of Fe2+ within a divided cell, facilitating continuous MnO2 deposition, represents a novel advancement. When combined with recent progress in membrane technology and electrolyte optimization, this method holds promise for enabling industrial-scale, sustainable EMD production.
The experimental findings suggest that, with further refinement, particularly in enhancing the current efficiency of Fe2+ regeneration, this integrated strategy could transform EMD manufacturing into a more environmentally responsible and economically viable process. Future research should prioritize improvements in electrode stability, mitigation of side reactions such as Fe2+ oxidation by atmospheric oxygen, and the development of scalable systems suitable for industrial deployment.
Scalability and industrial application
Future research will focus on both the experimental validation and economic evaluation of scaling the electrolytic production of electrolytic manganese dioxide (EMD) and ferrous iron Fe2+ regeneration from laboratory to pilot-scale operations. Key technical challenges to be addressed include optimizing electrolyte circulation, maintaining membrane integrity, and improving overall energy efficiency.
To enable a successful transition from laboratory-scale experiments to commercially viable processes, extensive testing and process optimization are required to resolve chemical engineering constraints and ensure operational reliability. Preliminary assessments suggest that incorporating Fe2+ regeneration, alongside robust process control strategies, could reduce energy consumption by up to 30%, thereby enhancing the economic competitiveness of the method relative to conventional approaches.
Additionally, the adoption of modular reactor designs may support incremental scaling, allowing for reduced initial capital expenditure and greater flexibility in deployment. Nonetheless, critical issues such as membrane fouling and waste management must be systematically addressed to maintain long-term process stability and ensure compliance with environmental regulations.
Conclusion
In summary, XRD results confirm that the main solid phase species in the material recovered by electrolysis was manganese dioxide. Overall, the cell voltage is directly proportional to the current density and decreases as temperature increases. Additionally, the results indicate that higher current densities lead to lower cell voltages due to the increased rate of Fe2+ regeneration. The electrolytic production of manganese dioxide offers a low cell voltage and reduced costs compared to conventional methods. It also provides a reductant for the leaching stage and significantly decreases cell voltage by recycling Fe2+ at the cathode. The savings in mV are reduced as the current density increases. A short electrolysis test was conducted to examine the effect of time on electrode potentials for the deposition of EMD and the quality of manganese oxide deposited at the anode. The plating potential at the anode remained relatively constant during deposition, while the potential at the cathode for the regeneration of Fe2+ remained fairly constant. This relationship between current density and cell voltage can be further explored in future studies to optimize electrolysis processes. Furthermore, it is important to consider the impact of temperature on cell voltage when designing electrolysis systems for specific applications. By understanding how current density and temperature affect cell voltage, researchers can develop more efficient and cost-effective electrolysis processes. This finding can lead to advancements in green energy technology and help reduce the environmental impact of industrial processes.
Highlights
This manuscript highlights a promising and sustainable approach to EMD production that could significantly impact the battery industry.
This study presents a novel electrolytic process for producing high-purity manganese dioxide while simultaneously regenerating ferrous iron, enhancing overall process efficiency. The integrated regeneration of Fe2+ reduces reliance on external reductants and lowers operational costs, contributing to more sustainable manganese dioxide production. Characterization confirmed that the recovered solid was exclusively manganese dioxide, demonstrating the effectiveness of the electrochemical deposition method. Findings indicate that increasing current density and temperature can decrease cell voltage, leading to reduced energy consumption and improved process economics. The process aligns with principles of sustainability by minimizing pollutant emissions and supporting resource recovery, advancing sustainable practices in electrometallurgy. The process is suitable for industrial-scale production and has the potential to revolutionize the production of cathode materials for batteries.
Footnotes
Acknowledgements
I acknowledge Professor Brian Loveday for guidance on this work. I thank the University of KwaZulu-Natal for their support during this research project. I would also like to express my gratitude to the National Research Foundation of South Africa for their financial assistance. This research was made possible through their funding.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work the authors used QuillBot and Grammarly language and grammar checks. After using these tools, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Author statement
This statement accurately reflects that MGN was primarily responsible for the hands-on experimental work and the initial drafting of the manuscript, while BL provided the overarching conceptual framework, supervised the project, and secured the necessary funding. Both authors contributed to the review and editing process.

