Abstract
Keywords
Introduction
The vaginal contraceptive ring is considered a ‘second-tier’ contraceptive method, meaning that it is considered very effective; however, it is user dependent, which decreases its efficacy. 1 The very first Food and Drug Administration (FDA)-approved vaginal contraceptive ring was released in 2001. Since that time, numerous studies have been performed examining the efficacy of the vaginal contraceptive ring, and different versions have been released. There are limited resources that have synthesized this information into a single document. In this article, we will discuss the different types of vaginal contraceptive rings: the etonogestrel/ethinyl estradiol (ENG/EE) ring and the segesterone acetate (SA)/EE ring. The details of dosing and administration, indications, advantages, disadvantages, usage, adverse reactions, efficacy, cost-effectiveness, and newest research available will be reviewed for each method. The purpose of this article is to provide a comprehensive reference on the two vaginal contraceptive rings widely used in the United States for clinicians to guide management.
Methods
This literature review was conducted using PubMed. The search was conducted in June 2022. The search terms included ‘vaginal contraceptive ring’, ‘etonogestrel/ethinyl estradiol ring’, and ‘segesterone acetate/ethinyl estradiol ring’. The search was then sorted by year from 2000 till present in both databases. The search yielded 774 articles. Articles were screened by the first and second authors for relevance. The remaining approximately 72 articles were reviewed by the first two authors. Articles were selected by author’s perceived relevance to clinical practice and understanding of these medical devices (Figure 1). The FDA labels of the devices discussed were also reviewed. Additional websites were reviewed to view company data and recent customer perspectives.
Flow diagram for database review.
ENG/EE ring
Introduction
In the United States, the intravaginal hormonal contraceptive containing ENG/EE is called the
Pharmacology
ENG/EE are rapidly absorbed by the body from the device. According to a study examining serum hormone levels cited in the FDA label, the bioavailability of ENG and EE is 100% and 56%, respectively. 2 This is comparable to oral administration of these hormones. When ENG is distributed in the blood, about 32% is bound to sex hormone-binding globulin (SHBG) and about 66% is bound to albumin. Likewise, about 98.5% of EE is bound to serum albumin and induces an increase in serum concentrations of SHBG. Both ENG and EE are metabolized in liver microsomes by the cytochrome P450 3A4 isoenzyme. EE, specifically, is primarily metabolized by aromatic hydroxylation, forming a variety of hydroxylated and methylated metabolites, which are present as free metabolites and as sulfate and glucuronide conjugates. 2 When these hormones are administered vaginally, gastrointestinal absorption and the hepatic first-pass metabolism are avoided. Despite this, concentrations of EE and ENG levels in the uterus are not elevated compared to combined oral contraceptives (COCs). 3 Indications for the ENG/EE ring include prevention of pregnancy, reduction in menorrhagia, and dysmenorrhea. 4
Mechanism of action
The ENG/EE ring is a combination of hormonal contraceptive. These contraceptives work by suppressing hypothalamic gonadotropin-releasing hormone. This, in turn, prevents pituitary secretion of luteinizing hormone (LH) and follicle-stimulating hormone. Follicle-stimulating hormone is suppressed by estrogen, which stabilizes the endometrium. Therefore, when the ENG/EE ring is removed, the endometrium sheds, causing withdrawal bleeding. 1 LH is suppressed by progesterone, which prevents ovulation. When this occurs, the cervical mucus thickens to restrict the entry of sperm into the uterus. Suppression of LH also causes the endometrium to become unfavorable for implantation. 1
Usage
The ENG/EE ring is inserted inside the vagina and remains in place continuously for 3 weeks. The ring is then removed for 1 week to allow for withdrawal bleeding, then a new ring is placed. This withdrawal bleeding usually begins 2–3 days after the removal of the ring. The bleeding may not have stopped by the time a new ring should be placed; however, a new ring should be placed exactly 7 days after removal to remain effective. 2
To insert the ring, the patient can stand with one leg up, squatting, or lying down. The ring is compressed and inserted. The ring is removed by hooking the index finger under the forward rim, or by grasping the ring between two fingers and pulling it out.
2
The placement of the ring does not have to be perfect to be efficacious. In an
The ENG/EE ring may be removed from the vagina and not lose contraceptive efficacy if it is reinserted within 3 h. To initiate the use of the ENG/EE ring, it should be inserted on the first day of menstrual bleeding. 2
Off-label continuous use is recommended for those who have issues with abnormal bleeding patterns. It is recommended for those who do not desire to take pills. Continuous use is achieved by immediately inserting a new contraceptive ring after 3 weeks, instead of waiting 7 days for withdrawal bleeding. The ring was found to be effective and tolerable with continuous use; however, women were more likely to experience breakthrough bleeding. 6
Efficacy and advantages
A widely accepted way to statistically report contraceptive efficacy is called the ‘Pearl Index’. The Pearl Index is defined as the expected number of pregnancies per 100 woman-years of exposure. This index can be calculated for the intent-to-treat (ITT) population and the per protocol (PP) population. The ITT population is defined as all patients who used a certain treatment. The PP population is defined as all patients who used a treatment without any protocol violations. 7 The Pearl Index demonstrates the effectiveness of the contraceptive method in terms of how well it can prevent pregnancy. The lower the index, the better the contraceptive is at preventing pregnancy. 8
In a study performed by Roumen
Another appealing factor of the ENG/EE ring is the favorable bleeding profile. In a 1-year, multicenter study in which 1156 patients were exposed to the vaginal ring for 12,109 cycles, irregular bleeding occurred in only 1.6–6.4% of the cycles, which mostly consisted of spotting.
7
Breakthrough bleeding was reported in only 0.4–1.1% of the cycles. Withdrawal bleeding occurred in 97.9–99.4% of cycles.
7
In a study by Dieben
There have been various studies to examine the effects of concomitant vaginal products on the efficacy of the ENG/EE ring.4,9–12 The use of spermicide (specifically nonoxynol-9) with the ENG/EE ring was studied. Furthermore, it was demonstrated that the spermicide had no effect on the absorption, serum levels, and efficacy of the vaginal contraceptive device. 10 The use of tampons with the ENG/EE ring was also found to have no effect on hormone levels, and is therefore not expected to compromise its efficacy. 11
Adverse reactions
The most serious adverse reactions observed with the
The most common adverse reaction to lead to discontinuation of the ENG/EE ring was device-related events. This was cited by approximately 2.7% of patients. 2 Other common causes of discontinuation include mood changes, headache/migraine, and vaginal symptoms. 2 Close follow-up is recommended for those affected by vaginal irritation, toxic shock syndrome, constipation, and uterovaginal prolapse. 4
A study was performed by Etminan
Patients should be counseled regarding the risk of VTE. A large prospective observational study called the Transatlantic Active Surveillance on Cardiovascular Safety of NuvaRing (TASC) compared the risk of VTE between the NuvaRing and COCs. The results were similar, with an incidence of 8.3 per 10,000 women-years for the NuvaRing and 9.2 per 10,000 women-years for COCs. 2 There have also been case reports describing the development of life-threatening ailments including portal venous thrombosis and cerebral sinus venous thrombosis.16,17
In a study conducted by Roumen
Contraindications
Absolute contraindications of the ENG/EE ring include history of cardiovascular disease, venous thromboembolism, hypertension, diabetes, liver disease, headaches with neurological symptoms, and smokers older than 35. It should also not be used in those with known or suspected breast, endometrial, vaginal, cervical cancer, and undiagnosed abnormal vaginal bleeding. Patients should also be counseled against the use of ENG/EE ring if they have a family history of benign breast disease, breast cancer, hyperlipidemia, liver disease, heart disease, hypertension, and migraines. 4 In addition, the ENG/EE ring should not be initiated within 30 days of planned surgery, as the estrogen component may lead to increased risk of thrombosis. 4 Some relative contraindications of the ENG/EE ring include uncontrolled dyslipidemia and non-migraine headaches. 18
Cost-effectiveness
Crespi
SA/EE ring
Introduction
Another common vaginal contraceptive ring newly released in the United States is called
Pharmacology
A study was performed by Liu
Mechanism of action
The mechanism of action of the SA/EE ring is the same as the ENG/EE ring because they are both combination of hormonal contraceptives. These methods of contraception reduce the risk of pregnancy by suppressing ovulation, which is described in detail above (see ENG/EE Ring
Usage
The SA/EE ring is placed inside the vagina and remains in place for 21 days. The device is removed during the next 7 days to allow for withdrawal bleeding. During this time, the ring is cleaned and stored in its case. After the 7-day period, the same ring is replaced inside the vagina for the next cycle. The SA/EE ring was designed to provide contraception for 1 year or 13 cycles. The method of insertion of the SA/EE ring is identical to the ENG/EE ring. The important point to emphasize with patients is that the device should be entirely inside the vagina, behind the pelvic bone. 21
If a patient has regular periods, the SA/EE ring can be initiated between Day 2 and Day 5 of the menstrual cycle without the need for back-up contraception. If the patient has irregular periods or if the ring is inserted at any other time of a patient’s regular cycle, the ring will not be effective for the first 7 days of use and a back-up method is required. 21
Efficacy and advantages
The efficacy of the SA/EE ring was studied in two multicenter, open-label, single-arm, phase III trials with identical protocols performed by Archer
The efficacy analysis included 2265 participants and about 57.5% of participants completed the 13 cycles. The Pearl Index for the primary efficacy group was 2.98, which was within range of efficacy for a patient-controlled contraceptive system. An ITT life table analysis was performed which showed 97.5% efficacy in preventing pregnancy. Furthermore, there was no change in efficacy across 13 cycles. The Pearl Index for good use was 2.98 (95% CI 2.13–4.06) per 100 woman-years. If participants removed the vaginal ring for longer than 2 h, the Pearl Index increased to 5.89, which indicates reduced efficacy with improper use. The youngest participants in the study had the highest rate of contraceptive failure. 23
Another consideration when prescribing this vaginal contraceptive ring are medications that can be used in combination with this product. A study done by Simmons
Adverse reactions
The most serious adverse reactions with
A safety evaluation was recently published by Gemezell-Danielsson
The most common adverse reaction to lead to discontinuation was metrorrhagia or menorrhagia, which was reported by 1.7% of patients. 21 Other adverse reactions that lead to discontinuation of the SA/EE ring is headache (1.3%), vaginal discharge or mycotic infections (1.3%), and nausea/vomiting (1.2%). 21 The rate of partial expulsion of the device is 19.8% and the rate of complete expulsion is 7%. 25
It is important to counsel patients regarding the risk of venous thromboembolism (VTE). In a clinical safety study that combines data from four large datasets from patients using the SA/EE ring, four (0.2%) of 999 women who completed 13 cycles of use experienced VTE. Of note, three had other risk factors for thrombosis including Factor V Leiden mutation (one patient) and two with a BMI greater than 29 kg/m2. 25
Contraindications
The SA/EE ring is contraindicated in those with a hypersensitivity to any of the components of the ring. It is also contraindicated in those who are at high risk of arterial or venous thrombotic events. This includes individuals above the age of 35 who smoke, and those with a history of deep vein thrombosis, pulmonary embolism, cerebrovascular disease, coronary artery disease, inherited or acquired hypercoagulopathies, and uncontrolled diabetes mellitus or hypertension. In addition, individuals with headache with focal neurological symptoms and migraine headaches with aura are also at high risk for thrombotic events. 21
This product should not be given to patients with a history of breast cancer or other estrogen- or progesterone-sensitive cancers. In addition, this product should be avoided in patients with abnormal uterine bleeding of unknown origin, due to the risk of undiagnosed endometrial hyperplasia. 21
The SA/EE ring is contraindicated in those with liver tumors, acute hepatitis, or severe cirrhosis. This is due to the risk of liver damage which could lead to elevated liver enzymes and jaundice. The use of Hepatitis C drug combinations with the SA/EE ring is contraindicated due to the risk of alanine transaminase elevations. Development of hepatic adenomas is a known risk of hormonal contraceptive use. These tumors have the potential to grow with SA/EE rings, which may lead to death by intra-abdominal hemorrhage. 21
Cost-effectiveness
In 2018, the cost of a single
Conclusions
Clinical implications
The nuances that differentiate the ENG/EE ring and the SA/EE ring should be discussed with patients who are considering a vaginal contraceptive ring and are summarized in Table 1. When counseling patients on the use of each vaginal contraceptive ring, proper usage should be emphasized to ensure efficacy. Patients should be instructed to check ring placement before and after sexual intercourse. The device may be expelled accidentally while removing a tampon, during intercourse or straining with a bowel movement. In this case, the device can be rinsed with lukewarm water and re-inserted as soon as possible.
Vaginal contraceptive rings comparison.
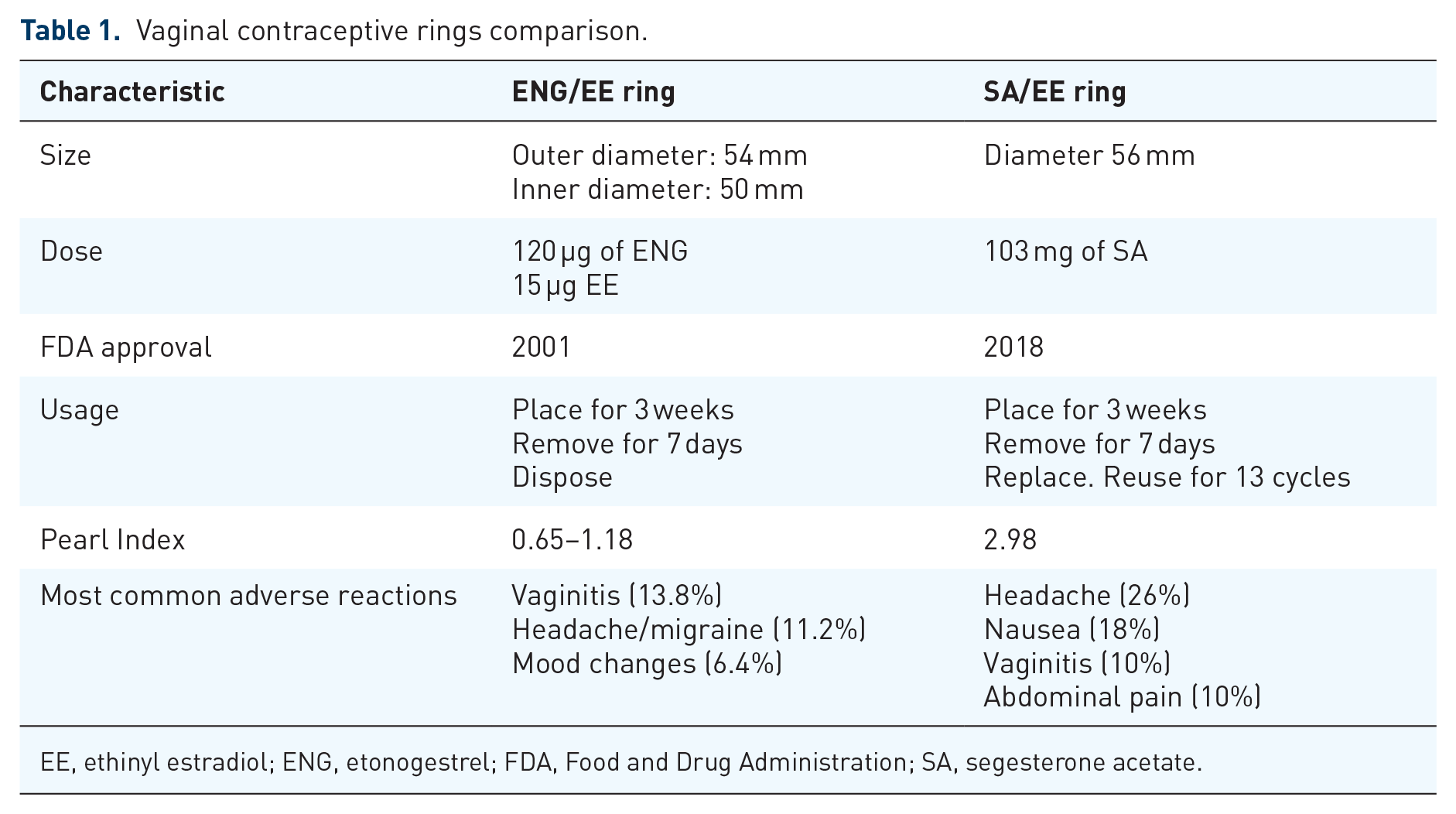
EE, ethinyl estradiol; ENG, etonogestrel; FDA, Food and Drug Administration; SA, segesterone acetate.
A study was performed by Stifani
If the ring has been out of place for greater than 3 h, then after the ring is replaced, the patient should use a backup contraceptive method (condoms or spermicide) for the next 7 days. 2 When removing the ring, patient should also try to keep similar times in which it was inserted. 30
It is also important to consider the appropriate time to initiate usage of the vaginal contraceptive ring. A patient may start using both vaginal contraceptive rings within the first 5 days following a complete first trimester abortion or miscarriage. Following childbirth, the ENG/EE ring should not be initiated until 4 weeks postpartum due to the increased risk of thromboembolism.2,21 If patients desire to change their contraceptive method, the vaginal contraceptive ring can be placed on the day of Depo Provera, Nexplanon, and IUD renewal. 21
Patient satisfaction
In two multicenter open-year study questionnaires by Szarewski, patients had a greater than 90% satisfaction rate with vaginal ring contraceptive. 31 A qualitative analysis was performed after in-depth interviews with 32 patients of various racial/ethnic minoritized groups. Overall, the patients initially had concerns about insertion and removal, leaving the device inside the vagina, interference with sex, and the size of the device; however, many patients were able to overcome their concerns with counseling from providers and positive experiences. Most patients stated that they would recommend it to friends.32–34
Summary
There are two widely used vaginal contraceptive rings: ENG/EE estradiol ring and the SA/EE ring. The two rings have similar side effect profiles and efficacy. Overall, the vaginal contraceptive ring is well tolerated and liked by patients. Patients should be well counseled on known severe adverse reactions. The vaginal contraceptive ring is more expensive than other forms of contraception and this should be an important point of discussion with the patient.
