Abstract
BACKGROUND:
Higher consumption of phenolic-antioxidant rich berries have been associated with lower risks of chronic diseases, such as type 2 diabetes. In order to target for diet-based therapy it is important to screen and evaluate the right cultivars of berries with optimum phenolic antioxidants and associated anti-hyperglycemic functions.
OBJECTIVE:
Therefore, the primary objective of this study was to screen different rabbit-eye blueberry cultivars extracted in water and ethanol for their phenolic antioxidant-linked anti-diabetic properties using
METHODS:
Water and ethanol extracts of five rabbit-eye blueberry cultivars from two different locations and from two different growing seasons were evaluated for phenolic acids profile, total soluble phenolic content, total antioxidant activity, α-amylase, α-glucosidase, and angiotensin I-converting enzyme (ACE) inhibitory activities using model
RESULTS:
Significant variations in phenolic antioxidant-linked anti-hyperglycemic functions were observed due to differences in genotypes, growing seasons, and locations. High phenolic antioxidant-linked anti-hyperglycemic functions were observed in Tiffblue, Brightwell, and Primier rabbit-eye blueberry cultivars and indicated anti-diabetic potential.
CONCLUSION:
Genotype×environment interactions are most critical factors which determined
Introduction
Blueberries are rich source of dietary fiber, essential micronutrients, and phenolic antioxidants with diverse human health relevant biological activities and functions [1, 2]. Most specifically, blueberries have been known to offer a natural and abundant source of hydrophilic antioxidants such as anthocyanin and other phenolic acids [3]. These natural phenolic antioxidants from plant-based foods are becoming important targets for safe and cost-effective dietary support therapy for oxidative stress-linked non-communicable chronic diseases (NCDs), like type 2 diabetes, cardiovascular disease, and cancer [4–6]. Due to such rich bioactive profile and associated health benefits, blueberries (fresh or frozen) have been highly sought healthy food choice amongconsumers.
The market opportunity of blueberry is rapidly expanding from its geographic origin of America with 10% of the total United States highbush blueberry being exported each year [7]. In the United States, total production of blueberry whether cultivated or wild, in 2014 was around 667.6 million pounds with estimated market value of $824.9 million [8]. Blueberry industry is projecting 25% increase in North American production in next three years with 50% increase in per capita blueberry consumption [7]. There are three different major types of blueberry grown in the United States such as highbush, lowbush, and rabbit-eye with many cultivated varieties from each type being developed in the last few decades for different regions and climatic conditions. Among these blueberry types, hexaploid rabbit-eye blueberries (
Blueberry antioxidant has shown protective functions against chronic oxidative stress associated with NCDs, such as type 2 diabetes, and cardiovascular diseases [13, 14]. Recent studies indicated that phenolic antioxidants from berries also influence glucose and lipid metabolism [14, 15]. Phenolic acids and their metabolites from plant-based foods potentially influence digestion, absorption, and metabolism of dietary carbohydrates such as starch and sucrose and therefore control postprandial blood glucose level [15]. For maintaining glucose homeostasis, α-glucosidase and α-amylase are extremely important enzymes that regulate the rate in which starch are digested and subsequently absorbed as glucose in the intestine [15]. The main function of pancreatic α-amylase is by catalyzing hydrolysis of 1,4-glucosidic bonds, resulting in the release of maltose [15]. Similarly, α-glucosidase, an enzyme found in the small intestine, hydrolyzes the 1,4-linked glucose residues in order to further digest carbohydrates to release glucose [16], before immediate absorption by the intestinal cells. By inhibiting these enzymes, carbohydrate digestion can be slowed in its passage in the intestinal tract resulting in delayed glucose absorption [17]. Previous studies have indicated that berries with highest anthocyanin content, such as blueberry and black currant, exhibited far better inhibition of α-glucosidase than strawberries and red raspberry extracts [18–20]. Inhibition of α-amylase and α-glucosidase have proven effective against early onset of type 2 diabetes and associated complications [19, 20]. But pharmaceutical drugs such as Acarbose with inhibitory activities against these enzymes can cause unwanted side effects including flatulence, abdominal discomfort, and diarrhea due to high levels of α-amylase inhibitory activity [21]. Therefore, finding natural plant-based food sources such as berries with moderate α-amylase and high α-glucosidase inhibitory activities are gaining increasing attention for safe and cost-effective dietary management of early stages type2 diabetes.
Although berries and berry phenolics have shown high baseline values of α-amylase and α-glucosidase inhibitory activities both in previous
Materials and methods
Materials
A total of five rabbit-eye blueberry cultivars; Tifblue, Climax, Pimier, Brightwell, and Baldwin were obtained from the Auburn University (grown in two locations 245 miles apart: Dothan, AL 36302 and Cullman, AL 35055 for 2009, and 2010). Both Dothan and Cullman have a humid sub-tropical climate with average annual rainfall of 143–146 cm with slightly cooler average night temperature in Cullman. Dothan has fine-loamy soil, while Cullman has sandstone mixed with alluvial soil. Due to disease infestation no fruits of Baldwin blueberry cultivars were collected from Dothan in both years. Frozen sample (4 months after harvest) of blueberry fruits were shipped to the Department of Food Science, University of Massachusetts, Amherst, MA 01002 in ice and then biochemical analysis was carried out in 2010 and 2011 for the respective collection years. Porcine pancreatic a-amylase (EC 3.2.1.1), baker’s yeast glucosidase (EC 3.2.1.20), 2, 2-diphenyl-1-picrylhydrazyl (DPPH), and 3,5-Dinitrosalicylic acid (DNS) were purchased from Sigma Chemical Co. (St. Louis, MO). All other chemicals and solvents were purchased from Sigma Chemical Co. (St. Louis, MO) and of analytical grade.
Preparation of blueberry extracts
Well ripened and frozen blueberry fruits from five cultivars: Tifblue, Climax, Pimier, Brightwell, and Baldwin were selected. Upon even thawing, 40 g of whole blueberry fruits were added to 100 mL of either distilled water to reflect physiological relevance and water-based foods and beverages or 12% ethanol to reflect typical alcoholic beverage levels and homogenized for 5 minutes using a Waring laboratory blender (Winsted, CN) set on HIGH. Two replications were used for each extraction. The homogenate was then collected after two time centrifugation at 10,000
Phenolic acid profiles (HPLC)
The blueberry sample (0.4 g/mL) extracts (2 mL) were filtered through a 0.2 μm filter. A volume of 5 μL of sample was injected using an Agilent ALS 1100 autosampler into an Agilent 1100 series HPLC (Agilent Technologies, Palo Alto, CA) equipped with a DAD 1100 diode array detector. The solvents used for gradient elution were (A) 10 mM phosphoric acid (pH 2.5) and (B) 100% methanol. The methanol concentration was increased to 60% for the first 8 min and to 100% over the next 7 min, then decreased to 0% for the next 3 min and was maintained for the next 7 min (total run time, 25 min). The analytical column used was Agilent Zorbax SB-C18, 250 - 4.6 mm i.d., with packing material of 5 μm particle size at a flow rate of 1 mL/min at room temperature. During each run the absorbance was recorded at 306 nm and 333 nm and the chromatogram was integrated using Agilent Chem station enhanced integrator. Pure standards of chlorogenic acid, protocatechuic acid, gallic acid, ellagic acid and quercetin in 100% methanol were used to calibrate the standard curves and retention times.
Total soluble phenolics assay
The total soluble phenolic content was determined using Folin–Ciocalteu method, modified by Shetty et al. [25]. Blueberry sample (0.4 g/mL) extract (0.5 mL) and 0.5 mL of distilled water were transferred into a test tube and mixed with 1 mL of 95% ethanol, 5 mL of distilled water, 0.5 mL of 50% (vol/vol) Folin–Ciocalteu, and 1 mL of 5% Na2CO3 and the reaction mixture allowed to stand for 60 min. The absorbance was read at 725 nm and results were expressed as mg of gallic acid equivalent per gram of sample in fresh weight (FW) basis.
Antioxidant activity by 2, 2-diphenyl-1-picrylhydrazyl radical (DPPH) inhibition assay
The DPPH scavenging activity was determined by an assay method modified from Pinto et al. [26]. 1.25 mL of 60 μM DPPH in 95% ethanol was added to 250 μL of each sample extract (0.4 g/mL), with the decrease in the absorbance monitored after 5 min at 517 nm. The absorbance of a control (distilled water instead of sample extract) was also recorded after 5 min at the same wavelength. The percentage of inhibition was then calculated by the following equation:
a-Amylase inhibition assay
The α-amylase inhibitory activity was determined using an assay method modified from the Worthington Enzyme Manual [26]. A total of 500 μL of each blueberry sample extract (0.4 g/mL) and 500 μL of 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M NaCl) containing α-amylase solution (0.5 mg/mL) were incubated at 25°C for 10 min. After pre-incubation, 500 μL of a 1% starch solution in 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M NaCl) was added to each tube and then incubated at 25°C for 10 min. Next, 1.0 mL of dinitrosalicylic acid color reagent was added. The test tubes were then incubated in a boiling water bath for 10 min and absorbance was measured at 540 nm after cooling and dilution. Additionally, a 1:2 and 1:5 dilution of the sample extract was performed to investigate potential dose dependence. The
a-Glucosidase inhibition assay
The assay method is modified from the Worthington Enzyme Manual for
Angiotensin I-converting enzyme (ACE) inhibition assay
ACE inhibitory activity was determined using an assay method modified by Kwon et al. [27]. A volume of 50 μL of blueberry sample extract (0.4 g/mL) was incubated with 200 μL of 0.1 M NaCl-borate buffer (0.3 M NaCl, pH 8.3) containing 2 mU of ACE solution at 25°C for 10 min. After pre-incubation, 100 μL of a 5.0 mM substrate (hippuryl-histidyl-leucine) solution was added to the reaction mixture. Test solutions were incubated at 37°C for 1 h. Sample blanks (buffer in place of enzyme and substrate), a control (distilled water instead of sample extract) and a blank (buffer instead of sample extract and enzyme) were also included. The reaction was stopped with 150 μL of 0.5 N HCl. The hippuric acid formed was detected by the High Performance Liquid Chromatography (HPLC) method [27]. The percentage of inhibition was calculated considering the area of the hippuric acid peak according to the equation below:
Statistical analysis
Two extractions were performed for each sample and all
Results and discussion
HPLC analysis of phenolic acids profile
Phenolic acids profile provides significant information on potential biological functions and human health relevant properties in plant-based foods including berries [28]. The major phenolic acids found in this study were catechin, gallic acid, protocatechuic acid, and quercetin (Table 1). Similar composition of phenolic acids were observed in previous studies with blueberry cultivars [19, 29]. Protocatechuic acid was not detected in rabbit-eye blueberry cultivars such as Brightwell and Baldwin. Therefore phenolic acids composition of blueberries significantly varies with the choice of cultivars. Among phenolic acids catechin was most prominent phenolics followed by quercetin and protocatechuic acids detected in this study. Ethanol extracts (12%) of blueberry fruits had significantly higher phenolic acids profile when compared with the water extracts of same blueberry cultivar. Similar effect of extraction solvents on phenolic acids profile of blueberry was observed in a previous study [30]. Overall, higher concentrations of phenolic acids were also observed in rabbit-eye blueberry fruits grown in year 1. Therefore, growing and other environmental conditions also have significant impacts on phenolic acids profile of blueberries. This study suggests that blueberry has rich phenolic acid profile with significant biological activities and functions, including human health relevant properties.
Phenolic acid content (ug/ mL) of water (A) and ethanol (B) extracts of five blueberry cultivars from two different locations (Cullman and Dothan) and two growing seasons
Phenolic acid content (ug/ mL) of water (A) and ethanol (B) extracts of five blueberry cultivars from two different locations (Cullman and Dothan) and two growing seasons
Total soluble phenolic content of five rabbit-eye blueberry cultivars was determined using Folin-Ciocalteu method [25]. Significant variations in total soluble phenolic content was observed among five rabbit-eye blueberry cultivars, between two locations, two growing seasons, and cultivar×location×growing season interactions (

Total soluble phenolic content (mg/g F.W.) of water (A) and ethanol (B) extracts of five different rabbit-eye blueberry cultivars grown in two locations (Dothan & Cullman) and in two growing seasons. Different capital letter represents significant differences between cultivar×location×growing season interactions at
Blueberries are rich source of natural antioxidants and many health benefits of blueberry have been attributed to this characteristic [34]. Therefore to understand the potential role of rabbit-eye blueberry cultivars to counter oxidative stress-induced human diseases, antioxidant activity was determined using DPPH free radical scavenging assay. Similar to the total soluble phenolic content significant variations in total antioxidant activity was observed among cultivars, between two growing seasons, two locations, and cultivar×location×growing season interactions (

Total antioxidant activity (DPPH % inhibition) of water (A) and ethanol (B) extracts of five different rabbit-eye blueberry cultivars grown in two locations (Dothan & Cullman) and in two growing seasons. Different capital letter represents significant differences between cultivar×location×growing season interactions at
Correlation coefficient (
Cooperation between α-glucosidase and α-amylase is crucial in the control of the postprandial hyperglycemic state in diabetic individuals [16]. Inhibiting α-glucosidase can result in a delayed breakdown of starch and absorption of glucose in the small intestine, which aids in the controlling of the postprandial increase in glucose. Alpha-amylase plays a similar initial role in controlling the rate at which starches are digested into shorter chain length rather than how they are broken and absorbed in the final step as glucose. Therefore, to understand the potential anti-hyperglycemic functions of rabbit-eye blueberry cultivars for α-glucosidase and α-amylase inhibitory activities was determined using
Overall, significantly high α-amylase and α-glucosidase inhibitory activity was observed in all undiluted water and ethanol extracts of rabbit-eye blueberry cultivars, irrespective of the growing seasons, locations, and extraction methods. Due to high baseline value (98–100% inhibition) of α-amylase and α-glucosidase inhibitory activity (78–96%) in undiluted water and ethanol extracts, no significant differences was observed among cultivars, growing seasons, and locations and therefore data of undiluted sample was not presented. When diluted in half significant variations in α-amylase inhibitory activity among rabbit-eye blueberry cultivars, locations, growing seasons, and cultivar×location×growing season interactions were observed (Fig. 3A, B). Similar to total soluble phenolic content and antioxidant activity, significantly higher α-amylase inhibitory activity was observed in Tiffblue and Brightwell rabbit-eye blueberry cultivars. Rabbit-eye blueberry cultivars grown in year 1 and in Cullman location was also resulted in higher α-amylase inhibitory activity when compared with rabbit-eye blueberries grown in year 2 and in Dothan locations. This result indicates that α-amylase inhibitory activity of rabbit-eye blueberry cultivars has positive correlations with total soluble phenolic content and total antioxidant activities (Table 2). Therefore, the α-amylase inhibitory activity of rabbit-eye blueberry cultivars may bedependent of phenolic antioxidant profiles and has significant relevance to potentially improve glucose metabolism when consumed as a diet and provides strong biochemical rationale for further clinical studies. Similar levels of α-amylase inhibitory activity in berries were reported in previous studies [19, 26].
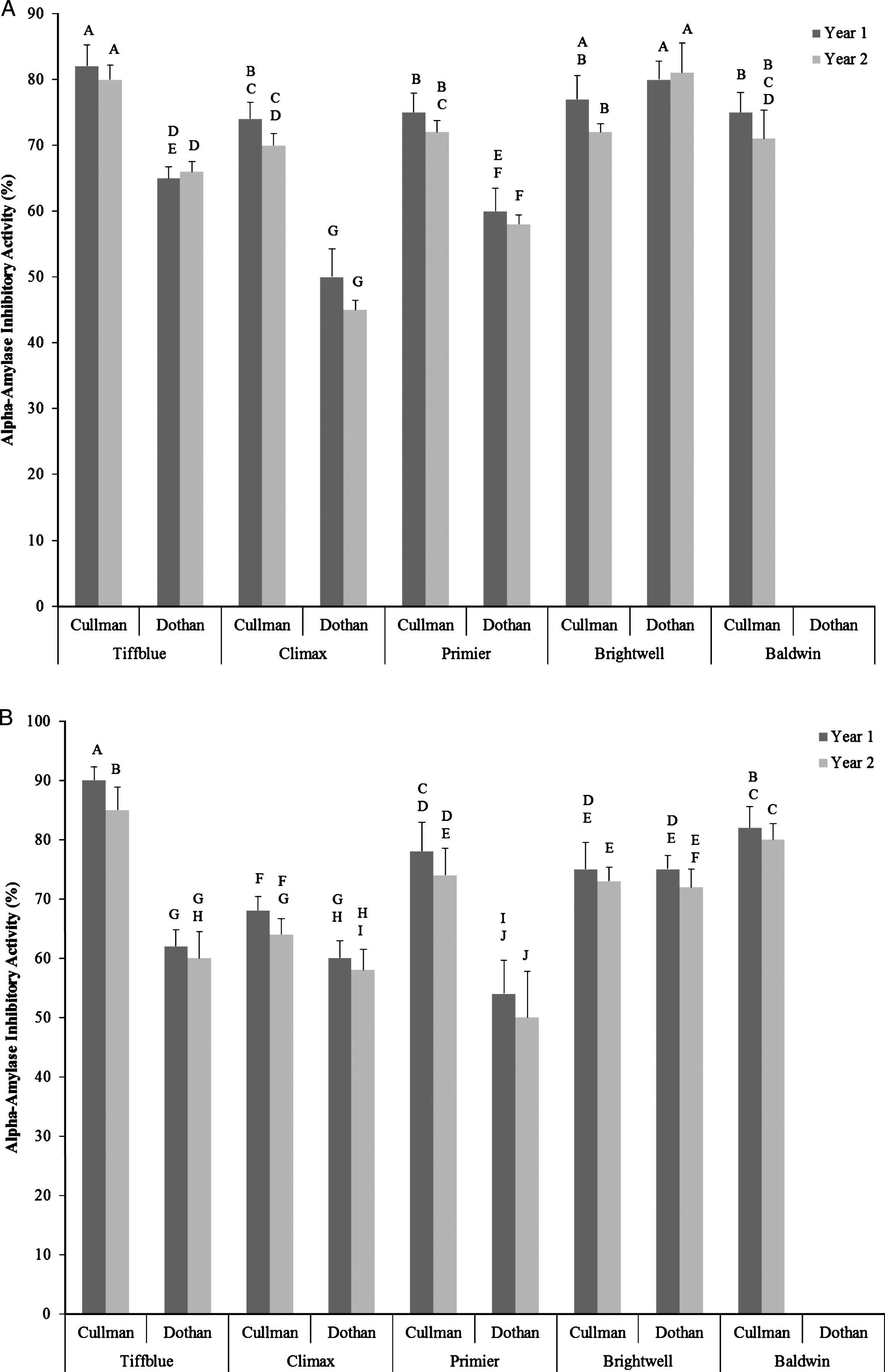
Alpha-amylase inhibitory activity (%) of half-diluted water (A) and ethanol (B) extracts of five different rabbit-eye blueberry cultivars grown in two locations (Dothan & Cullman) and in two growing seasons. Different capital letter represents significant differences between cultivar×location×growing season interactions at
Like α-amylase inhibitory activity significant variations in α–glucosidase inhibitory activities among rabbit-eye blueberry cultivars, between growing seasons, locations, and cultivar×location×growing season interactions were also observed when blueberry extracts was diluted in half (Fig. 4A, B). Higher α–glucosidase inhibitory activity was observed in Brightwell (82%) and Climax (80%) blueberry cultivars, even in half-diluted sample of ethanol extracts (12%). In general, similar to the total soluble phenolic content and total antioxidant activity, higher α–glucosidase inhibitory was also observed in rabbit-eye blueberry cultivars grown in year 1 and in Cullman when compared with blueberry fruits of same cultivars from year 2 and from Dothan location. Similar level of α–glucosidase inhibitory activities has been reported in previous studies [19, 26]. This

Alpha-glucosidase inhibitory activity (%) of half-diluted water (A) and ethanol (B) extracts of five different rabbit-eye blueberry cultivars grown in two locations (Dothan & Cullman) and in two growing seasons. Different capital letter represents significant differences between cultivar×location×growing season interactions at
The angiotensin I-converting enzyme (ACE) converts angiotensin II from angiotensin I [37, 38]. Once angiotensin II is produced it can form a vasoconstrictor in the pancreas, which functions by constricting arteries and veins causing an increase in blood pressure. By inhibiting this vasoconstrictor it is possible to improve blood flow and insulin secretion through beta cells [38, 39]. Therefore, to determine the potentials of rabbit-eye blueberry cultivars to manage hypertension associated with type 2 diabetes,
Conclusions
Plant-based foods rich in phenolic antioxidants such as berries are excellent choice to be utilized in developing functional foods and functional ingredients to prevent and manage oxidative stress-linked diseases such as type 2 diabetes, CVD, and cancer. Such dietary strategy involving plant-based foods are safe and cost-effective as many pharmaceutical drugs have long term consequences and health care costs are also rising rapidly worldwide. But before utilizing in dietary strategies or in expensive clinical studies, it is important to screen and determine appropriate cultivars and suitable growing conditions for obtaining the optimum levels of human health relevant functional properties in plant foods. In this
Conflict of interest
None to report.
