Abstract
Introduction
The era of hydroxyapatite (HA) in regenerative science dates back to 1950s 1 when bioceramics were used as an inert scaffold for filling of the bone defects. The history of calcium orthophosphates dates back to 1770; 2 the early history till 1950 can be read elsewhere in the published literature.3,4 On the basis of the exhaustive literature available on HA from 1950 in relation to its composition, properties, production, and its uses, this article comprehensively reviews landmark studies on HA properties and its use in clinical sciences with changing trends in the understanding of material interaction with living tissues. 5 In the beginning, HA was used for grafting, which had no reaction with adjacent living tissues. Later, the trend changed to the reactive nature of the material so that it is categorized as second generation, where the material acts as a conductive scaffold for bony ingrowth. 4 Recently, emerging production technology with the advent of nanotechnology and understanding of regenerative science has changed the face of bioceramics to a different dimension.6–11
HA is one of the bioceramics that represents the large quantity of regenerative graft material available in the market. HA is very closely associated with the bony apatite structure. It is one of the inorganic components of the bone. It is bounded in the organic matrix, so that it exists with other mineral trace elements in the normal bone. 2 Diseases associated with the bone and ablative surgery the resections or removal of part of the bone, which ultimately requires reconstruction through various available measures. Because of the nature of the HA, it is gaining increasing importance in regenerative science as a potential substitute material next to autograft. This paper comprehensively reviews the role of HA in regenerative science since the beginning of the history.
Chemical and Physical Properties of HA
HA is used as a bone substitute because of its chemical similarities with the natural bone. The major composition of bone is a mineral phase (69 wt%), an organic matrix (22 wt%), and water (9 wt%). 4 Bone is the major calcified tissue present in mammals 2 and is a ceramic–organic bionanocomposite that has a complex structure. HA with a general formula of Ca10(OH)2(PO4)6 is much similar to an inorganic component of bone matrix. 4 Because of this close similarity, extensive research is ongoing to use HA as a bone substitute. HA is one of the most stable and less-soluble calcium phosphate bioceramics with Ca/P ratio of 1.67.1,4 The pure HA powder is white, whereas naturally occurring HA can also have brown, yellow, or green colorations, comparable to the discolorations of dental fluorosis. In biological systems, HA occurs as the principal inorganic constituent of normal (bone, teeth, fish enameloid, and some species of shells) and pathological (dental and urinary calculus and stones) calcifications. 2 The mechanical properties of HA depend on porosity, density, sinterability, crystal size, phase composition, and so on. The bending, compressive, and tensile strength values of HA ceramics lie in the range of 38–250, 120–150, and 38–300 MPa, respectively.1,4 Young's modulus of dense HA ceramics varies from 35 to 120 GPa, depending on the residual porosity and impurities.1,2,4 Weibull's modulus of dense HA ceramics lies in the range 5–18, characteristic of brittle materials. The Vicker's hardness of dense HA ceramics is 3–7 GPa. The mechanical properties of HA bioceramics strongly depend on the microstructure and sintering ability; densely sintered bodies with fine grains are tougher and stronger than porous ones with larger grains.2,4
Biological Properties of HA
HA bioceramics have been widely used as artificial bone substitutes because of their favorable biological properties, which include biocompatibility, bioaffinity, bioactivity, osteoconduction, 1 osteointegration, 2 and osteoinduction 3 (in certain conditions). HA contains only calcium and phosphate ions and therefore no adverse local or systemic toxicity has been reported in any study. When implanted, newly formed bone binds directly to HA through a carbonated calcium-deficient apatite layer at the bone-*-implant interface.4,5 HA surface supports osteoblastic cell adhesion, growth, and differentiation, and new bone is deposited by the creeping substitution from the adjacent living bone. HA scaffolds can also serve as delivery vehicles for cytokines with a capacity to bind and concentrate bone morphogenetic proteins (BMPs) in vivo. 6
The interaction of apatite with biological tissues is an important aspect for regeneration. The concepts in the mineralization and tissue interaction are changing because of the change in production technology, size, nature of material, and so on. The beginning of bone regeneration has started with scaffold. The term biomimetics was coined by Otto Herbert Schmitt in 1950s. 4 This biological process induces the biological process of generation of highly ordered materials with hybrid composition and begins by designing and synthesizing molecules that have the ability to self-organize spontaneously to higher order structures. 4
The tissue interactions of HA are important. It is necessary to understand the in vivo host responses for HA. In general, the mechanism of action of a biomaterial is considered to be biocompatible, bioinert, biotolerant, and bioactive, and includes bioresorbable materials. These shifts in understanding have occurred due to the changes in the properties and production technology and the deeper understanding of material interaction with the tissues. The advanced front of nanotechnology results in cutting edge production of HA in a much bioactive or bioresorbable manner. The tissue reaction to any foreign body, even though it is biocompatible, will form a capsule thus it will be isolated. Bioinert materials will not show any positive interaction nor release any toxic constituents. The body or host tissue will separate such materials through encapsulation, which measures the bioinertness of material. 4
A bioactive material will dissolve slightly, but it forms a biological apatite before it interacts with tissues at the atomic level; this results in the formation of chemical bonds directly with bones. This phenomenon provides good stabilization for the materials that are subjected to mechanical loading. Bioresorbable material dissolves over a period of time so that new tissues will grow into surface irregularities. The bioresorbable materials are used as scaffolds that allow substitution and act as filling material. These reactions depend on the nature of the material, such as porosity. Recently, concepts of bioactive material made into bioresorbable and bioresorbable into bioactive. The use of HA with Ca/P ratio of 1.0–1.7 is nontoxic and neither has it induced any foreign body reaction. The nature of healing mimics fracture healing. 4
HA has displayed an ability to directly bond with bone. Sometimes, the micromovement of implants may lead to inflammation because of disruption of large microvessels that have grown into the pores of the implant. HA also exhibits the property of osteoinductivity.5,10,12–25 The mechanism of bone induction by a synthetic material is still not clear, but various factors such as microporosity, surface area, geometry, and topography are important,25–27 of which microporosity has a positive effect on increasing ectopic bone formation. Some studies have shown that osteoinduction is brought about by the concentration of bone growth factors from circulating biological fluids. 28 Other studies have shown that the geometry of HA is a critical parameter in bone induction. Nano structured HA, rough surfaces are found to cause asymmetrical division of stem cells into osteoblasts which is important for osteoinduction. 26
Biodegradation of HA is usually initiated by changes in the surrounding biofluids and adsorption of biomolecules. The physicochemical dissolution process depends on the surface area to volume ratio, fluid convection, acidity, and temperature.29,30 The dissolution is usually inversely proportional to the Ca/P ratio, purity, crystal size, and surface area. Usually, HA is more stable than other calcium orthophosphates such as TCP. Bioresorption is usually mediated by osteoclast cells,31–33 sometimes by macrophages. The biodegradation kinetics depend on the Ha/TCP ratio. The higher the ratio, the lower the degradation. The incorporation of ions either increases or decreases the solubility of HA and CDHA. 4
Bioactive materials form a chemical bond. The roughness and biomaterial porosity are considered to be important factors for bonding.34–36 The interfacial reactions of bioactive materials were introduced by Prof. H Larry.37–40 Soon after implantation, the proteins will be adsorbed on the HA surface. The extent and interconnectivity of pores influence bone ingrowth and blood vessel formation.41–44 A minimal pore size of approximately 50 µm has been estimated for blood vessel formation and approximately 200 µm for osteoid growth. The pore dimension, approximately 100 µm and 50 µm, also showed bone ingrowth. 45 The pore size and effects are as follows: <1 µm is responsible for bioactivity, interaction with proteins, and attraction of cells; 1–20 µm leads to the orientation and directionality of cellular and bone ingrowth; 100–1,000 µm helps in mechanical strengthening and functionality; >1,000 µm influences the shape and esthetics of the implant. 46
The degree of porosity regulates the bioactivity of graft substitutes, which controls the rate of bone regeneration, local environment, and equilibrium of new bone at the repair site. The pore interconnectivity, geometry, topography, and porosity modulate osteogenesis, which synergistically promotes the osteoconductivity or the inductivity potential of bone graft.47–49 The excellent biocompatibility, possible osteoinductivity,5,10,12–17 and high affinity for drugs, protein, and cells make these tissue engineering applications very much functional. 50
In the beginning, the bioceramics were bioinert, but the trend changed to responsive bioceramics.
51
These have been divided into different generations, which depict evolution and properties of HA biomaterials. Osteoconduction and osteoinduction properties of HA scaffolds are well known. Osteoinduction occurs because of the stimulation of the host mesenchymal stem cells. These stem cells then differentiate into bone-forming osteoblasts. Extensive studies have been conducted over the past several years to understand the osteoinduction potential of HA. Osteoinduction has been observed in several independent studies in various hosts such as dogs, goats, and baboons.7–10 Ripamonti et al have conducted extensive work on the long-term use of HA implants in the nonhuman primate
The rationale in using HA coatings as a means of fixation for orthopedic and dental implants has been known as early as the 1980s. HA as a surface coating attempts to improve bone fixation to the implant and thus increases the lifetime of metallic implants. Higher osteoblast activity and in vitro increased collagen levels seen in cells growing on HA-coated Ti, 52 in vivo HA coat resulted in higher bone–implant contact area. 53 Enhancing the ingrowth of mineralized tissue improved the biological fixation, biocompatibility, and bioactivity of dental implants. 54 The deposition can be achieved through plasma spraying, sputter coating, pulsed laser deposition, dynamic mixing method, dip coating, sol–gel, electrophoretic deposition, electrochemical deposition, and biomimetic coating with various advantages and disadvantages. 55 Studies have suggested that both amorphous and crystalline phases in the coatings are desirable to promote a more stable interface with the biological environment.56,57 Thinner HA layers, in the nanometer range, revealed increased cellular response than thicker HA layers.58–60 Biomimetic nano-apatite coatings of porous titanium scaffolds resulted in an enhanced human osteoblast culture as well as greater bone formation in a canine bone in a growth chamber. 61 Recently, biomimetic HA-polymer composite scaffolds have been widely explored for bone regeneration.62,63
Recent Advances in HA
It is well established that nano-sized HA can mimic the dimensions of constituent components of calcified tissues such as bone and teeth. Thus, recent development of HA-based biomaterials for biomedical applications will obviously stand to benefit most from nanotechnology, which offers a unique approach to overcome the shortcomings of their conventional forms due to their large surface to volume ratio and unusual chemical/electronic synergistic effects. 1 Nanocrystalline HA is expected to have better bioactivity and dissolution than coarser crystals.41,42 Nanostructured biomaterials promote osteoblast adhesion and proliferation, osseointegration, and the deposition of calcium-containing minerals on the surface of these materials.43,44
Nanocrystalline HA powder exhibits improved sinterability and enhanced densification due to a greater surface area, which could improve the fracture toughness as well as other mechanical properties.21,23,45,46 In addition, nanostructured ceramics can be sintered at a lower temperature, thereby problems associated with high temperature-sintering processes can also be eliminated. It is possible to enhance both the mechanical and biological performance of HA by controlling the characteristic features of powders such as surface area, crystallinity, morphology, particle size, particle distribution, and agglomeration. 47 Even though nano-HA offers much improved performance than their conventional counterparts, it is not sufficient for wide applications. Artificial biomaterials with different properties depending upon the application are needed for hard tissue replacement. Properties such as density, porosity, thermal stability, bioactivity, resorbability, and mechanical properties should be tailored to make functional biomaterials.
The aim of tissue engineering is to help the body heal naturally by implanting a resorbable and porous scaffold to serve only as a temporary matrix that would degrade over time, while allowing the regeneration of the host tissue at the implant site. Degradation depends on the particle size, crystallinity, porosity, the composition and preparation conditions, as well as the environment at the implantation site. Recent advances in composite materials for bone engineering are based on nanotechnology and involve the development of nanocomposites containing nanofibers, HA nanoparticles, carbon nanotubes (CNTs), and so on. Bone is structurally divided into nanostructure, microstructure, and macrostructure that include cortical and cancellous bone.64–66 Nanotechnology is the creation of functional materials, devices, and systems through the control of matter on the nanometer length scale (1–100 nm) and exploitation of novel phenomena and physical, chemical, and biological properties at that length scale. 67 Engineering of nanomaterials can thus meet current challenges in bone replacement therapies.68,69 There are various techniques to manufacture nano-HA particles. These methods include wet chemical precipitation, 70 sol-gel synthesis, 71 co-precipitation, 72 hydro-thermal synthesis, 73 mechano-chemical synthesis, 74 mechanical alloying, 75 ball milling, 76 radio frequency induction, 77 electro-crystallization, 78 microwave processing, 79 hydrolysis of other calcium orthophosphates, 80 double step stirring, 81 and other methods. Nano-HA demonstrated rapid bone ingrowth and accelerated bone formation within and around the implanted material. 82 In recent years, several studies are focusing on the development of HA-CNT nanocomposites with improved mechanical properties, 83 and reports are also available for the processing of HA-CNT composite coatings for orthopedic implants through plasma spraying,84,85 laser surface alloying, 86 electrophoretic deposition, 87 and aerosol deposition. 88 In addition to conventional sintering89,90 and hot isostatic pressing, 91 spark plasma sintering (SPS) 92 has also been employed to fabricate freestanding HA–CNT composites.
In recent years, extensive studies have been conducted to develop biomimetic materials for bone tissue engineering applications. These materials should be three dimensional, have a high volume of open and interconnected pores, be a bioresorbable scaffold with controlled resorption, and have suitable mechanical properties, biocompatibility, and bioactivity. Porous HA implants can be manufactured using a variety of methods including processing of natural bone, ceramic foaming, sintering with porogens, starch consolidation, microwave processing, slip casting, and electrophoretic deposition. Microporosity results in a larger surface area that is believed to contribute to higher bone-inducing protein adsorption as well as to ion exchange and bone-like apatite formation by dissolution and re-precipitation. Over the past 15 years, a variety of 3D HA and biphasic scaffolds with various porosities and surface topographies have been developed.93–95
Forms and Uses of HA
Since its introduction in the mid-1980s, HA has been investigated for its clinical viability in various bone defects. Many researchers have demonstrated a better initial osseointegration and a high short-term success rate.96–98 HA-coated implants showed varying results of survival.99–105 The literature review revealed many studies revolving around these materials.106–149 Different forms of HA have been derived from different origins for various uses. Bovine HA124–135 and synthetic HA136–149 are major sources of HA grafts. These have shown varying success rates. Few studies have compared the efficacy of one form of HA over the other.106–123 HA use in the enhancement of bone regeneration in cystic defects has been selected for discussion. Many published studies have discussed about the implants and HA, which has been described briefly in previous sections.11,53,54 Available studies are divided mainly into bovine, synthetic, and comparative studies for the convenience of discussion and enumeration of the drawbacks and advantages collectively, as well as to discuss the emerging technology in the production with a note on recent materials over old ones.
Synthetic HA exhibits good properties as a biomaterial, such as biocompatibility, bioactivity, and osteoconductivity; hence, it has been widely used as a bone substitute, coating on metallic implants, scaffold for tissue engineering, and carrier for drug delivery. The different forms of HA employed for biomedical applications include porous and dense blocks, granules, paste, cement, and coatings.64–67 Despite its numerous useful characteristics, a major drawback of HA is its low strength resulting in the inability to make high load-bearing implants entirely out of HA. 68 Nanocrystalline HA is the main building block of bones and teeth. HA is used for grafting in sinus augmentation, ridge reconstructions, reconstruction of bone defects. A summary of the various forms of HA with different sources of origin has been enumerated in Tables 1–3106–149 The authors agree upon the use of HA for early bone regeneration in various maxillofacial surgical, orthopedic and neurosurgical, and facial esthetic procedures in an economic way without morbidity of second surgery to harvest the autogenous bone. 4 HA reconstruction of cystic cavity achieved superior bone quality with early regeneration without any complications. It also prevented dead space and fibrous healing of bone.130,150–155 All of the studies showed osteoconduction and few studies recently are in favor of osteoinduction.8–11 The published studies have many limitations. Randomized blind clinical trials are necessary to produce good clinical evidence, which are lacking in the existing literature. In vitro laboratory and animal histomorphometric analysis demonstrated HA as a valuable graft material (Tables 1–3). The changing scenario in the production technology has made HA osteoinductive.6–11
Comparative studies in published literature.
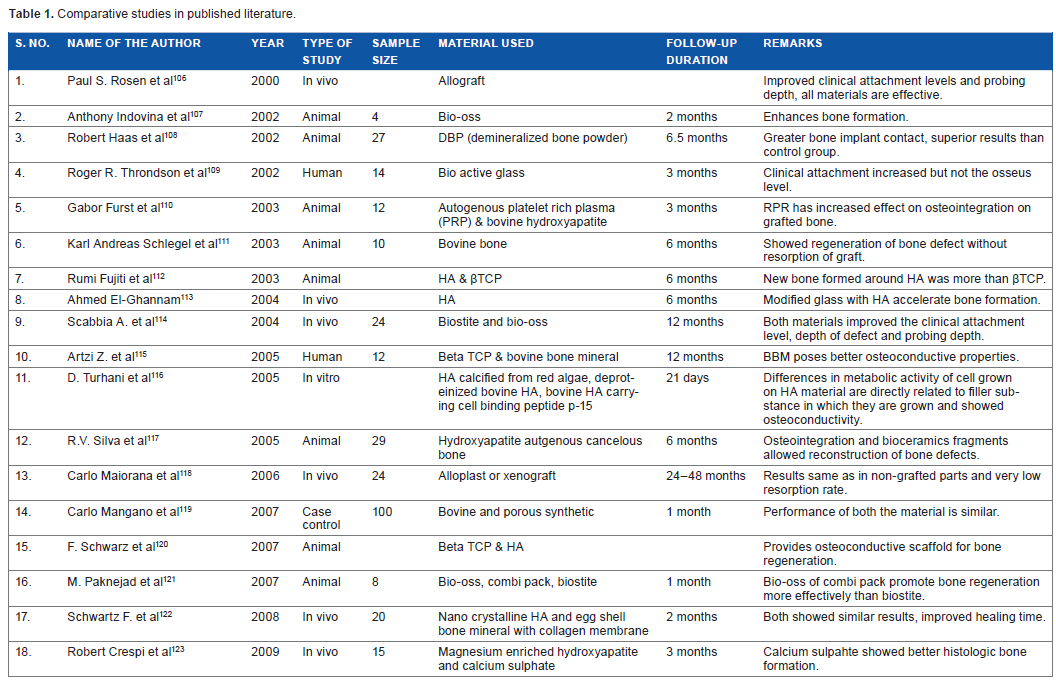
Bovine derived hydroxyapatite studies.

Synthetic hydroxyapatite studies.

Future Insights
In the near future, the production of HA is going to be economic because of the use of natural raw materials such as an egg shell. The production of HA from eggshell is an already established fact. 111 However, commercial production of the material is under trial. HA production is cost effective in a small scale. Pioneering reports on the properties of HA derived from egg shell have shown promising results.153,154 The nano-HA particles derived from egg shell are used in various maxillofacial reconstruction procedures with good results.155,156 The histomorphometric results of egg shell-derived HA have shown equal efficacy to that of commercially available materials. 154 Although only few comparative studies are available, they have demonstrated good performance. The use of nanotechnology is a boon for the production of HA. The material exhibited antibacterial properties, with the substitution of Ag, Sn, and other elements.4,156 The spatial rotation of HA particles makes HA as osteoinductive even in heterotrophic sites.6–11 Other interesting trends for HA include applications in drug delivery, cell culture, purification of antibodies on an industrial scale, as an artificial blood vessel or trachea, as well as a catheter made of an HA composite. 4
Author Contributions
Conceived and designed the experiments: VSK, KPL. Wrote the first draft of the manuscript: VSK. Contributed to the writing of the manuscript: SK. Agree with manuscript results and conclusions: VSK, SK, KPL. Jointly developed the structure and arguments for the paper: VSK, KPL. Made critical revisions and approved final version: VSK, SK, KPL. All authors reviewed and approved of the final manuscript.
