Abstract
Introduction
The medical treatment of metastatic renal cell carcinoma (RCC) has been revolutionized in recent years with the approval of several drugs. Previously, only immunotherapy had shown even limited efficacy in a minority of patients. Such revolutionary improvements resulted from advances in the knowledge of RCC's carcinogenesis. 1 Recent research has focused on the importance of two pathways involved in the development and prognosis of clear-cell RCC: the VEGF/VHL/HIF pathway and the PI3K/Akt/mTOR pathway.2,3 At least two thirds of clear-cell RCC exhibit inactivation of the VHL gene, either by mutation or loss of expression (Fig. 1). 4 This inactivation results in a deregulation of the expression of a wide range of genes, including Vascular Endothelial Growth Factor (VEGF). VEGF is the main growth factor involved in angiogenesis. Thus, the development of VEGF inhibitors was successful in many tumor types, including breast, colorectal, lung carcinomas. 5 In fact, RCC shows the greatest expression of VEGF in solid tumors, and the importance of its vascularization is a characteristic that has been recognized by clinicians for decades. In addition, VEGF expression was shown to be an adverse prognosis factor, either in localized tumors or in the metastatic setting. 6 Therefore, this axis was a preferred target for the development of targeted therapies in RCC. Five targeted therapies inhibiting the VEGF axis have already gained regulatory approval due to positive phase 3 clinical studies: sorafenib, sunitinib, bevacizumab, pazopanib and axitinib.7–11 In addition, others are still in development. Sorafenib, sunitinib, pazopanib and axitinib are multi-target tyrosine kinase (TKI) inhibitors that inhibit the VEGF Receptor, as well as other tyrosine kinases. Bevacizumab is a monoclonal antibody that target VEGF specifically. One other class of targeted therapies, mTOR inhibitors, was approved for metastatic RCC, including temsirolimus and everolimus.12,13 Among these treatments, sorafenib was the first TKI to gain approval by regulatory agencies concerned with RCC and is the focus of this review. 14

The VHL/HIF/VEGF Pathway.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Sorafenib is a multi-target TKI. 15 Like most TKI, it binds to the kinase at the ATP-binding site and inhibits the phosphorylation induced by the catalytic site. Sorafenib inhibits a wide range of tyrosine kinases, including BRAF (as well as mutant V600E BRAF), VEGFR-2, VEGFR-3, PDGFRβ, FLT3 and KIT. The concentrations inhibiting 50% (IC50) of activity of these tyrosine kinase are 6 nmol/L for RAF, 22 nmol/L for V600E BRAF, 90 nmol/L for VEGFR-2, 20 nmol/L for VEGFR-3, 57 nmol/L for PDGFRβ,58 nmol/L for FLT3 and 58 nmol/L for KIT. 16 Sorafenib inhibits phosphorylation and activity of the receptors and consecutively inhibits the activity of downstream signaling via the MAPKinase-pathway, as assessed by the inhibition of ERK signaling. 17 It should be noted that sorafenib was first developed as a RAF inhibitor, and was then recognized as a potent VEGFR inhibitor. This inhibitory profile could raise the possibility of a dual action in both endothelial and tumoral cells (Fig. 2). This was shown in cell lines model, but the effect in animal models and in human is more difficult to analyze.18,19 However, most data support a predominant effect as an antiangiogenic agent rather than a direct effect on tumoral cells. 18
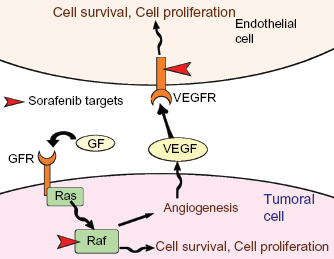
Potential mechanisms of action of Sorafenib in RCC.
Sorafenib is administrated as oral tablets. The bioavailability is about 38% to 49%. 20 This may be reduced by food ingestion and therefore the Food and Drug Administration (FDA) recommends avoiding ingestion concomitantly with a meal. 21 Sorafenib shows a large interpatient variability in pharmacokinetics. 22 However, this variability was not correlated with toxicities in phase 1 studies. Sorafenib is also highly protein-bound (99.5%). Its half-life is between 24 hours to 48 hours. Steady-state concentrations are reached after 7 days of dosing. Metabolism involves two pathways: sorafenib is a substrate of cytochrome P450 CYP3A4 and is also glucorono-conjugated by UDP glucuronosyltransferase (UGT) 1A9.23–25 However, around 50% of sorafenib is eliminated in its unchanged form. Two weeks after a single oral administration, 96% of the dose is eliminated, 77% in the feces and 19% in the urine. It is noteworthy that sorafenib seems to be neither an inducer nor an inhibitor of CYP3A4. However, other drugs can affect the activity of CYP3A4 (Table 1). Such treatments should be avoided during sorafenib treatment.
List of modifiers of CYP3A4.

Clinical Studies
Major clinical studies of sorafenib are summarized in Table 2
Sorafenib was studied in 4 phase 1 studies, exploring different schedules: 7 days on, 7 days off; 26 21 days on, 7 days off; 27 28 days on, 7 days off; 20 and continuous dosing. 22 These 4 phase 1 studies were reviewed by Strumberg in 2007. 28 Due to the half-life of 24 to 48 hours, most administration schemes involved twice-a-day administration. The maximum doses studied were 800 mg bid. Dose-limiting toxicities were consistent across the 4 studies, and dose-limiting toxicities were usually seen at 600 mg bid or higher. 28 In these 4 phase 1 studies, a total of 137 patients were evaluable for response and two responses were seen in this heavily pre-treated population. 28 However, in the continuous dosing study, 1 RCC patient had disease stabilization lasting more than 2 years, and 50% of patients with Hepatocellular Carcinoma were stable for more than 6 months, already showing interesting activity in the 2 settings with subsequent positive phase 3 studies. Some signs of activity were also seen in colorectal cancer patients. Detailed results of toxicity are reported in the corresponding section of this article.
Sorafenib's clinical studies.

Sorafenib was studied as monotherapy in a phase 2 study.
29
Interestingly, the design of this phase 2 was innovative: a randomized discontinuation trial. Patients included were initially treated with sorafenib open-label in a 12 week runin period at 400 mg bid. Disease was evaluated at 12 weeks using unusual criteria of response: bidimensional tumor measurements of responses were compared with baseline measurements, shrinkage of ≥25% was considered a response, ≥25% tumor growth or new lesions were considered progression. Patients with response continued open-label sorafenib, patients with progressive disease discontinued treatment. Patients without either response or progression were randomized to either sorafenib or placebo in a double-blind fashion. The primary endpoint was the percentage of patients randomly assigned who remained progression-free 12 weeks after randomization (24 weeks after study entry). In total, 502 patients were enrolled, but the publication focused on the 202 RCC patients. It should be noted that randomized discontinuation trials were developed to ensure rapid accrual–-patients are prone to accept the trial because they are sure to receive the studied drug–-but that in this particular case an important number of patients had to be included before completion of the study. At the end of the run-in phase, according to the study criteria, 36% were responders, 34% were stable, and 25% were progressive. The objective response rate using modified WHO criteria was 4%. Sixty-five patients were randomized, and progression occurred at a median of 6 weeks after randomization in the placebo arm, as compared with 24 weeks after randomization in the sorafenib arm (
Results of other studies are described in the safety and efficacy section of this article.7,11,30–33 After results of the pivotal TARGET trial, and before approval by regulatory agencies, two expended-access studies were launched, one in Europe and another in the United States and Canada: the European Advanced Renal Cell Carcinoma Sorafenib (EU-ARCCS) 31 and the US-ARCCS studies. 32
Sorafenib is studied in the adjuvant setting in 2 trials: 33 the SORCE trial (clinicaltrial.gov identifier NCT00492258), sponsored by the UK Medical Research Council, which compared 2 durations of sorafenib (1 year and 3 years) to placebo in patients at risk for relapse; and the ASSURE trial (NCT00326898), sponsored by the ECOG, in collaboration with the CALGB, SWOG, NCI and NCIC, which randomized patients at risk for relapse either to placebo, sorafenib, or sunitinib during a 1 year period. Results of these trials should be available in the next few years.
Safety
Specific side effects
The major side effects of sorafenib reported in published studies are summarized on Table 3.7,22,29–32 The US-ARCCS study reported only grade 2 or grade ≥ 3, but not all grade toxicities. 32 Most frequent toxicities are consistent between studies, with small differences in reported incidences. In the TARGET trial, the incidences of side effect are compared between sorafenib and placebo for grade 2 and grade ≥ 3 respectively, but not for all-grades toxicities. 30 Grade 2 toxicities significantly more present in the sorafenib group were diarrhea (incidence ranging between 43% and 58% across studies), rash (26% to 66%), hand-foot skin reactions (HFSR) (23% to 62%), hypertension (17% to 43%), alopecia (16% to 53%), weight loss (10% to 33%), pruritus (11% to 19%) and headache (6%).
Toxicities reported in clinical trials of continuous dosing of sorafenib.

Most deceivingly, precise descriptions of hemorrhage are frequently lacking in the report of those trials. A meta-analysis was recently published on fatal adverse events in studies involving VEGFR TKIs, including studies of sorafenib in RCC and other tumoral types.
34
Fatal adverse events were more frequent in the treatment arm than in the comparator arm of these studies. For sorafenib, the risk of fatal adverse events was 1.4% (95% CI, 0.6% to 3.2%), not different from others VEGFR TKIs. The relative risk was 2.68 (95% CI, 1.11 to 6.45;
Other toxicities described in subsequent studies were hypothyroidism36,37 (incidence of 18% in one study, 67% in another), hypophosphatemia, and cardiac dysfunction. Drops in left ventricular ejection fraction were reported under sorafenib and other TKIs.38–40 However, these cardiac dysfunctions rarely have clinical consequences. 41 Long-term toxicities were reported for the TARGET trial, in patients treated ≫ 1 year. 42 There was no new toxicity reported, but incidence of side effects seemed higher than those reported for the whole population, with diarrhea in 74% of patients, rash in 51%, HFSR in 49%, alopecia in 39% and fatigue in 38%. However, grade ≥ 3 adverse events were rare, the most frequent being HFSR (7%) and hypertension (5%). Cardiac ischemia was reported in 2%, left ventricular dysfunction in 1%.
Safety in different populations
Safety in elderly patients was addressed in the US-ARCCS study.32,43 Incidence of reported adverse events were not increased in patients aged more than 70 years. In the EU-ARCCS study, the same conclusion can be drawn, with the exception of fatigue, which was reported in 31% of patients aged < 70 year but in 44% of patients aged ≥ 70 years. 31 Results of the TARGET trials were comparable in the young and elderly populations. 44 The safety of sorafenib in patients with brain metastases seemed acceptable in the US-ARCCS and the EU-ARCCS studies, as well as other specific studies, with no increase of bleeding reported.31,32,45
Sorafenib was studied by the CALGB group in a phase 1 trial that included patients with renal or hepatic dysfunction. 46 This study involved patients with different abnormalities and was able to include 138 patients. In case of hepatic dysfunction, the recommended dosage was 400 mg bid for patients with bilirubin ≫ upper limit of normal (ULN) but ≤1.5x ULN and/or AST ≫ ULN, 200 mg bid for patients with bilirubin between 1.5 and 3x ULN, but not even 200 mg every third day was tolerable if bilirubin was >3x ULN. When albumin was below 2.5 mg/dL, recommended dosage was 200 mg each day. In case of renal dysfunction, recommended dosage was 400 mg bid if creatinine clearance was ≤40 mL/min, 200 mg bid if creatinine clearance was between 20 mL/min and 39 mL/min, could not be defined if creatinine clearance was below 20 mL/min without dialysis and was 200 mg each day in case of dialysis.
Management of toxicity
The peculiar toxicity profile of sorafenib (as well as those of other TKIs), prompted the need for proper management. Most side effects appear soon after the beginning of treatment, in the first two months, and many appear in the first 4 weeks. This timeframe emphasized the need for early visit of patients, possibly with the help of trained nurses. 47 Some side effects could be prevented by appropriate measures. Hypertension should be controlled before initiation of treatment. 48 Development of hypertension may require use of combination of antihypertensive drugs, with the help of a cardiologist if needed. However, sorafenib may, with proper management, be continued in the case of hypertension. HFSR could be prevented by proper counseling, urea-based topics, use of emollient and prompt intervention when symptoms appear. 49 Treatment of HFSR involves anti-inflammatory and keratolytics topics. No specific recommendations could be made about treatment of sorafenib-induced diarrhea.
Efficacy
First-line metastatic setting
Sorafenib was compared to interferon alpha-2a in untreated patients with metastatic RCC in a randomized phase 2 trial including 189 patients.
30
The primary endpoint was PFS. In this trial, patients progressing while receiving sorafenib had a dose-escalation to 600 mg bid and patients progressing while receiving interferon crossed over to sorafenib 400 mg bid. The PFS was not different between sorafenib and interferon: median PFS 5.7 months vs. 5.6 months, respectively, Hazard ratio (HR) = 0.88 (
The TARGET trial included 161 patients who were not previously treated by cytokines. 7 In these patients sorafenib achieved similar results to patients pretreated with cytokines, with a significant improvement in PFS, suggesting that sorafenib benefit over placebo is independent of the previous treatment with cytokines. In addition, in the EU-ARCCS and US-ARCCS studies, PFS was similar in patients pretreated by cytokines and patients not previously treated.31,32 Median PFS of non-previously treated patients was 24 weeks in the US-ARCCS study, close to results of the TARGET trial, again suggesting sorafenib's efficacy is independent of line of treatment.
The combination of sorafenib with interleukin-2 (IL2) was tested in a randomized phase 2 study comparing it to sorafenib alone. 50 Median PFS was 33 weeks with the combination vs. 30 weeks for sorafenib alone. Grade 3–4 adverse events were reported more frequently in the combination arm. Thus, the combination of IL2 with sorafenib did not show improvement in PFS and was associated with more severe adverse events. Other studies of the combination of sorafenib with interferon did not provide evidence of benefit in adding cytokine to sorafenib.51–54
Results of a phase 3 study comparing sorafenib to tivozanib in the first-line setting were recently presented at the 2012 ASCO annual meeting (Motzer et al, abstract 5401). Tivozanib is a potent inhibitor of VEGFR 1, 2 and 3. The results were in favor of tivozanib, with a median PFS of 12.7 months compared with 9.1 months for sorafenib. The toxicity profile also appeared to be better in the tivozanib arm.
Sorafenib in patients pretreated with cytokines
The TARGET trial included 903 patients pretreated for metastatic RCC, 82% of them being pretreated with cytokines.
7
Fifty-one percent of patients were classified as low-risk by the Memorial Sloan Kettering Cancer Center (MSKCC) classification, 49% as intermediate risk, and 94% were previously treated by nephrectomy. In this trial, sorafenib demonstrated superiority over placebo. The trial was designed to detect a difference of overall survival (OS). However, an interim analysis showed a significant improvement in PFS; consequently, the trial was prematurely stopped and cross-over was allowed for patients in the placebo arm. In the initial publication in 2007, OS was higher in the sorafenib arm, with a Hazard Ratio of 0.77 (95% CI, 0.63 to 0.95), but the
Other efficacy endpoints were in favor of sorafenib: median PFS was 5.5 months for the sorafenib arm vs. 2.8 months in the placebo arm, HR = 0.44 (95% CI, 0.35 to 0.55,
Sorafenib in patients pretreated with targeted therapy
The approval of sorafenib was close to those of bevacizumab and sunitinib in RCC. Soon after came the approval of temsirolimus, then everolimus and pazopanib. Despite the lack of proof of efficacy in randomized trials, clinicians were prone to offer access to sorafenib after progression under other targeted therapies. In fact, data from retrospective studies tends to support the use of second-line anti-VEGF treatment after prior antiangiogenic agents. Tamaskar et al studied 14 patients treated with sorafenib (as well as 16 patients treated with sunitinib) after receiving prior antiangiogenic treatment.
58
Ten out of 14 patients treated with sorafenib experienced some degree of shrinkage, including a partial response. Median time to progression was 10.4 months, with no evidence of difference between sorafenib and sunitinib. Di Lorenzo et al studied 34 patients treated by sorafenib after previous treatment with sunitinib and everolimus.
59
Median PFS was 4 months and median OS was 7 months. Partial responses were seen in 23.5% of patients. Vickers et al analyzed data of 216 patients receiving second-line treatments after first-line anti-VEGF therapy.
60
These patients showed better baseline Karnofsky performance status than patients not receiving second-line treatments. Median time to treatment failure after second-line anti-VEGF therapy was 4.9 months, compared with 2.5 months for patients receiving anti-mTOR therapy (
On the basis of these data, sorafenib is frequently proposed after progression under sunitinib. A phase 3 study compared sorafenib to axitinib in second-line treatment.
11
The AXIS trial randomized 723 patients between the two anti-VEGFR TKIs. Fifty-four percent of patients included were previously treated with sunitinib, 35% with cytokines, 8% with bevacizumab and 3% with temsirolimus. The results were in favor of axitinib, with a median PFS of 6.7 months in patients receiving axitinib vs. 4.7 months in patients receiving sorafenib (HR = 0.67; 95% CI 0.54 to 0.81,
Sorafenib was studied in a retrospective analysis in the third-line setting, after previous treatment with sunitinib and mTOR inhibitor. 61 Thirty-four patients were analyzed, showing median PFS of 4 months and median OS of 7 months in this heavily pretreated population, suggesting activity of sorafenib. However, the retrospective nature of the study and the likely highly selected nature of the population leads to difficulty in drawing firm conclusions.
Sorafenib use in different histological subtypes
Patients included in the TARGET trial had exclusively clear-cell RCC.
7
However, due to a lack of effective treatment in other histological subtypes, sorafenib was used for patients with different types of RCC. Choueiri et al were the first to describe efficacy of TKIs in other histological subtypes.
62
They studied 53 patients (41 with papillary RCC, 12 with chromophobe RCC), of whom 33 were treated with sorafenib. Their study tends to show some efficacy of TKIs, with a median PFS of 10.6 months for the chromophobe RCC, and 7.6 months for the papillary RCC. Median PFS was significantly higher in papillary RCC treated with sunitinib vs. those treated with sorafenib (11.9 vs. 5.1 months,
Difficulties in evaluation of response
The emergence of targeted therapies in RCC has raised questions about the proper endpoints of clinical trials. For instance, the Response Criteria in Solid Tumors (RECIST) were criticized as potentially underestimating the true efficacy of the treatment. Sorafenib is a good illustration of this point, with a modest response rate of 10% in the TARGET trial contrasting with a significant improvement in PFS and arguments of efficacy in terms of OS. 7 Indeed, if response evaluated by RECIST had been used in the phase 2 setting, sorafenib development could have been stopped in RCC. However, the phase 2 was designed as a randomized discontinuation trial, with time-to-progression as the primary objective. 29
Alternatives to RECIST were tested in RCC. Dynamic-contrast enhanced CTscan, MRI or UltraSound could help to better evaluate the effect of treatment on tumoral vascularization.64–69 Criteria using density were also proposed, as well as FDG-PET.64,70 All these methodologies report increase in the proportion of responding patients. However, data are still lacking about the clinical benefit of such evaluation criteria, and the consensus remains on using RECIST for better inter-trial comparisons. This area will certainly progress in the next few years.
Prediction of response
With the development of targeted therapies comes the hope of a “tailored treatment”.
71
Indeed, these therapies are associated with a high economic burden and significant toxicity. Selection of patients most likely to benefit from treatment would clearly be interesting. Prognostic factors included in the MSKCC classification are still valuable in the area of targeted therapies.
72
However, despite intensive research in the field, no predictive factor of response to antiangiogenic treatment has emerged. In the final report of the TARGET trial, Escudier et al reported that patients with high baseline plasma VEGF tended to derive more benefit from sorafenib, but even patients with the lowest level of VEGF still derive some benefit from sorafenib over placebo.
55
Thus, VEGF could not serve as a predictive factor. Pena et al published the complete analysis of biomarkers from the TARGET trial.
73
They found that some biomarkers were prognostic on univariate analysis, but that only TIMP-1 remained significant in multivariate analysis, together with the ECOG performance status and MSKCC score. However, no biomarker was found to be predictive of benefit from sorafenib.
Patient Preference
Patient-reported outcomes are increasingly considered of first importance for the evaluation of benefit from anticancer therapies. However, data on patient-reported outcomes or quality of life continue to be frequently missing in the literature. Concerning sorafenib, the TARGET trial and the phase 2 randomized trial of first-line sorafenib vs. interferon alpha 2a reported such results.30,56 In the TARGET trial, quality of life and symptoms were significantly improved in the sorafenib arm when compared with placebo. 56 In the phase 2 of sorafenib vs. interferon alpha 2a, a statistically and clinically significant difference of 5.9 points in the FKSI-15 scales, as well as an 11 point difference in the FACT-BRM scale, were shown in favor of sorafenib. 30 This data suggest that sorafenib was better than interferon in preserving quality of life. The authors also report a greater satisfaction in the convenience of treatment.
Patient preference between different targeted therapies used in RCC is more difficult to analyze. In a collaborative work, Bellmunt et al tried to select the patients’ characteristics that could benefit from sorafenib treatment. 76 However, they did not discuss patients’ preferences, as data are clearly lacking. Very few head-to-head comparisons in a clinical trial were performed. In the publication of the AXIS trial, no data on quality of life under axitinib or sorafenib were presented. 11 However, the authors proposed a composite endpoint including progression, death and deterioration of symptoms, which favored the axitinib arm. The results of the phase 3 study comparing sorafenib to tivozanib were recently presented, with a toxicity profile apparently favorable to tivaozanib over sorafenib. However, this should be taken with caution, as no results on quality of life or patient preference were reported. In the TARGET trial, quality of life was improved as compared with placebo, but comparison with other trials of targeted therapies would be questionable, as the populations included differ greatly. Thus, no conclusion can be drawn about patient preference for the selection between sorafenib and others targeted therapies in RCC. Appropriately on this matter, the PISCES trial compared sunitinib to pazopanib in the first-line setting, the primary objective being patient-reported preference between the two treatments. The results were presented at the 2012 ASCO annual meeting and were in favor of pazopanib. Such data are still lacking for sorafenib, but would be of great interest.
Place in Therapy
Guidelines
There are different guidelines for treatment of metastatic RCC. They are all based on the results of large trials, but differ somewhat on the interpretation of data concerning sorafenib use. While the National Comprehensive Cancer Network (NCCN) 77 guidelines recommend use of sorafenib in the first-line setting, the European Society of Medical Oncology (ESMO) 78 and the European Association of Urology (EAU) 79 guidelines do not. However, all guidelines agree in recommending the use of sorafenib in the second-line setting after treatment by cytokines (which is the main population included in the TARGET trial but a waning practice in current clinical methods), and agree in considering sorafenib as an option for treatment for non-clear-cell RCC.
Sequential or combination treatment
With the clinical use of targeted agents, other practices have emerged, even if strong evidence does not exist supporting them. Sorafenib is commonly used after pretreatment with sunitinib or mTOR inhibitors. 80 Questions were raised whether targeted therapies should be used in combination or in sequence. Indeed, mTOR acts downstream of VEGFR signaling, and mTOR inhibitors could increase the effects of VEGF-targeting agents. Moreover, mTOR induced stabilization of HIFα protein, increasing its pathway and VEGF secretion. Preclinical data suggest synergism between VEGFR TKIs and mTOR inhibitors. 19 Sorafenib was also tested in combination with chemotherapy. 81 However, most phase 1 trials of combination showed excessive toxicity, with the exception of the sorafenib-everolimus combination.82–86 Moreover, sequential treatments appear feasible in clinical practice, and data on efficacy in the clinics have emerged, albeit partial and prone to biases. Reintroduction of sorafenib after previous failure was also associated with efficacy in some cases, as well as dose escalation.87,88
Some studies raised the possibility of better efficacy in the sorafenib-sunitinib sequence when compared to the sunitinib-sorafenib sequence.89–95 However, additional studies did not report the same results. Another debate was the best sequence after failure of a VEGFR TKI–-should the patient be treated with an mTOR inhibitor, then another VEGFR TKI, or in the reverse sequence? Once again, conflicting results were published.60,80 To date, no recommendations can be on best sequence of treatment. Only trials of different strategies may help answer the question, such as the SWITCH trial (NCT01481870), which compares the sunitinib-sorafenib sequence with the sorafenib-sunitinib sequence.
A final setting where the use of targeted therapies is debated is the neoadjuvant setting. 96 One phase 2 trial discussed the opportunity to use sorafenib before surgery. 97 However, other VEGFR TKIs associated with higher response rates would probably become the treatment of choice in the rare indications where downstaging of locally advanced RCC is needed.
Conclusions
Sorafenib was the first VEGFR-TKI to be approved for treatment of RCC. Its efficacy was demonstrated in metastatic clear-cell RCC refractory to cytokines, but its clinical use exceeds this indication. Sorafenib is currently used as second-line or third-line treatment of metastatic clear-cell RCC, as well as in the treatment of non-clear-cell metastatic RCC. Questions remain about the best sequences of therapies in RCC, as well as about the development of predictive factor for benefit and appropriate evaluation of response. However, sorafenib will likely remain as one of the mainstays of RCC treatment over the next few years. Results on efficacy in the adjuvant setting are eagerly awaited.
Author Contributions
Wrote the first draft of the manuscript: JE. Contributed to the writing of the manuscript: JE, EV, NRL, CP, CV, KB, BL. Agree with manuscript results and conclusions: JE, EV, NRL, CP, CV, KB, BL. Jointly developed the structure and arguments for the paper: JE, EV, BL. Made critical revisions and approved final version: JE, EV, NRL, CP, CV, KB, BL. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
CV has received payment from Roche for board membership and lectures, from Novartis for board membership and consultancy, and from Amgen for consultancy. All other authors disclose no competing interests.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest. Provenance: the authors were invited to submit this paper.
