Abstract
Introduction: The Role of Extended Hormonal Contraceptive Regimens
Combination oral contraceptives (COCs) are the most frequently used method of contraception in the United States. Since their introduction in 1960, more than 80 percent of women in the United States born after 1945 have used COCs at some time in their lives. 1 Since 1960, dozens of different COC formulations with varied composition and dosage of estrogen and progestin components as well as different dosing regimens have been marketed. Unfortunately, despite the popularity of COCs, discontinuation rates at one year are approximately 33–30 percent and unintended pregnancy rates have not changed appreciably over the past two decades. While COC discontinuation is most often related to access problems, side effects such as headache, moodiness, and bloating also contribute to this phenomenon. The pregnancy rate with typical use of COCs is estimated to be 8% at the end of one year, while with perfect use the pregnancy rate is less than 1%.2,3
Over the past half-century, most pill formulations consisted of a 21/7 regimen–-21 days of active hormone consisting of an estrogen, most often mestranol or ethinyl estradiol, and one of several progestin components, followed by a 7-day hormone-free interval (HFI). The traditional 21/7 regimen was originally chosen to allow women to experience a monthly withdrawal bleed in a time prior to the availability of rapid immunologic pregnancy testing. Additionally, social and cultural norms at the time made monthly withdrawal bleeding preferable and allowed for acceptability of hormones as a novel form of contraception. For decades, however, providers and women have altered this regimen in an off-label fashion by decreasing the duration and frequency of the HFI. Extended cycling has long been used as a safe, effective means for modifying menstrual patterns for patient convenience as well as for treatment of various menstrual and gynecologic disorders including dysmenorrhea, endometriosis, menorrhagia, and leiomyomata.
Although clinicians have recognized that monthly withdrawal bleeding is not medically necessary and have successfully used continuous or extended-cycle regimens for several decades, it is only in the past decade that the US Food and Drug Administration (FDA) has approved specific COC formulations for extended-use. While many women are reassured by monthly menstruation, more research over the past decade has suggested that women find oligomenorrhea or amenorrhea desirable and acceptable.4,5 A multicenter survey of 1470 United States women observed that fifty nine percent of women would prefer to menstruate less than once per month. 6 Among providers, acceptability is also high with only a small proportion of providers, 11 and 12 percent in two studies respectively, stating that monthly bleeding is important.6,7
Additional benefits of extended cycle hormonal contraceptive regimens may also include an increase in typical use efficacy. Although effectiveness of extended and traditional regimens has been similar in structured clinical trials, real-world typical patterns of use may differ. Women participating in studies may be more willing and able to adhere consistently to their contraceptive regimen than the larger population of women. A large part of the increase in failure rate between perfect and typical use may be attributed to delays in starting the subsequent cycle of pills. 3 With approximately half of all women developing ovarian follicles large enough to ovulate by the end of the traditional 7-day HFI, delays in starting the next pill pack or missed pills predispose women to ovulation and contraceptive failure, particularly in the lowest dose pill formulations. 8 Shortening or removing the frequency of the HFI may lessen the effect of missed pills and the resultant risk of ovulation and contraceptive failure.
Additional benefits of extended cycle COC regimens may result from greater suppression of ovarian follicular activity as compared to 21/7 regimens. Degree of ovarian suppression has been associated with side effects women commonly experience with the various available regimens; more follicular development during and just after the HFI has been associated with increased unscheduled bleeding 9 as well as with symptoms such as mood changes, pelvic pain, and headaches. 10 Approaches to limiting follicular development with lower-dose COCs include decreasing the frequency of or completely eliminating the HFI, deceasing the duration of the HFI, and supplementing low dose estrogen in lieu of a completely hormone-free interval. Additionally, because COCs containing 0.02 mg EE tend to have more ovarian activity during the HFI than 0.03 mg EE formulations in 21/7 regimens, limiting the duration and frequency of the HFI may improve the side effect profile of these lower dose formulations.11,12
While scheduled bleeding days are decreased in COC regimens with fewer hormone-free intervals per time period, most women do experience some degree of unscheduled bleeding with extended-cycle regimens. Studies of the first United States FDA approved 91-day cycle regimen, consisting of 0.15 mg levonorgestrel and 0.03 mg EE for 84 days followed by a 7-day HFI (84/7), demonstrated fewer scheduled bleeding days but a greater incidence of unscheduled bleeding in the first cycles as compared to a 21/7 regimen of the same medication. At the end of one year, however, rates of unscheduled bleeding and spotting with the extended regimen decreased and was comparable to the 21/7 regimen. At the completion of four 13-week cycles of this formulation, the median number of unscheduled bleeding days was 1.5 and 41% had no unscheduled bleeding. 13
Although bleeding patterns improved with time in the above study, unpredictable bleeding is one of the leading causes of dissatisfaction with COCs. 14 A modification to both 21/7 and extended-cycle regimens that may limit unscheduled bleeding and decrease follicular development includes addition of low-dose estrogen during the HFI. Addition of EE to the HFI was originally studied in monthly regimens and is associated with greater ovarian follicular suppression than with a completely hormone-free week. Two randomized studies have demonstrated decreased rises in inhibin-B, follicle-stimulating hormone (FSH) and estradiol level during the HFI.15,16 Levels of ovarian activity during the HFI, as measured by follicular size and estradiol and progesterone levels, have also been directly correlated with the unscheduled bleeding. 9 When bleeding profiles in an 84/7 regimen with the addition of 0.01 mg EE to the HFI were compared to a similar 84/7 regimen without EE supplementation, scheduled bleeding was similar over time, although the two regimens were not compared directly. 13 Unscheduled bleeding, however, was noted to decrease more quickly with the regimen containing low-dose EE in place of placebo during the HFI. 17 A third FDA approved and studied extended COC regimen consists of 0.09 mg LNG with 0.02 mg EE and was also noted to have unscheduled bleeding that decreased over time in a fashion similar to the 84/7 regimens, with close to 59% of women achieving full amenorrhea at the end of 12 months. 18
The remainder of this review will focus on the newest addition to the list of FDA approved extended-cycle COCs–-a 91-day regimen consisting of 84 days of 0.02 mg EE plus 0.1 mg LNG followed by 7 days of 0.01 mg EE during what would traditionally be the hormone-free interval. This is the first dedicated 84/7 regimen consisting of a 0.02 mg EE pill.
Pharmacology
Mechanisms of action
The 84/7 regimen of 0.02 mg EE and 0.1 mg LNG plus 0.01 mg EE works through several mechanisms, similar to other COC formulations, to prevent pregnancy. The primary mechanisms of action include preventing ovulation through suppression of the hypothalamic-pituitary-ovarian system and thickening of cervical mucus to prevent sperm penetration into the upper genital tract. Other proposed mechanisms include slowing fallopian tubal motility and alterations of the endometrium, which would reduce the likelihood of implantation of the fertilized zygote.
Pharmacokinetics
While no studies have directly evaluated the absorption of the active hormones in the current regimen, levonorgestrel is a gonane progestin derived from 19-nortestosterone and is rapidly and completely absorbed with nearly 100% bioavailability and no first pass metabolism. Chemical structures of the active hormone components in this formulation are shown in Figure 1.

Structural formulas for levonorgestrel and ethinyl estradiol.
Levonorgestrel and its metabolites are excreted primarily in urine and feces. Ethinyl estradiol is nearly completely absorbed from the gastrointestinal tract and has a bioavailability of approximately 43% secondary to first pass metabolism in the gut and liver.
During the initial evaluation of this formulation, a subpopulation of 30 women were evaluated for pharmacokinetic parameters. Measures were collected following a single dose of 3 tablets of 0.02 mg EE with 0.1 mg LNG under fasting conditions, as shown in Table 1. 19
Mean pharmacokinetic parameters after 3 tablets of 0.02 EE and 0.1 mg LNG. 19

Levonorgestrel
LNG circulates as 97%–99% protein bound, primarily to sex hormone binding globulin (SHBG) and, to a lesser extent, albumin. EE circulates 95%–97% bound to albumin and induces SHBG synthesis. This increase effectively decreases the clearance and increases the plasma concentrations of LNG over repeated dosing. Once absorbed, LNG is metabolized by conjugation at the 17 beta-OH position to form sulfate and glucuronide conjugates. Metabolic clearance rates are variable among individuals, accounting in part for variation in LNG concentrations among users. LNG and its metabolites are excreted about 45% in the urine and 32% in feces, most of which is reabsorbed and undergoes recirculation.
Ethinyl estradiol
Most EE is metabolized first by the gut wall to ethinyl-3-sulfate. A portion of the remaining untransformed EE undergoes hydroxylation via hepatic metabolism with cytochrome P-450 3A4 (CYP3A4). CYP3A4 levels vary among individuals and may account for the varying rates of EE metabolism.
Clinical Studies
Clinical studies with the 0.02 mg EE and 0.1 mg LNG plus 0.01 mg EE 91-day COC regimen have been limited to a single industry sponsored trial. 20 This initial study was a multicenter, open-label, single-treatment study of 2185 women aged 18–41 years desiring contraception. Participants were excluded only if they met any contraindications for COCs. Women were not excluded on the basis of weight or body mass index (BMI), as long as they met all inclusion and exclusion criteria, and BMI range was from 15.7–62.8. A total of 1249 women completed the study. Subjects were provided study medicine for four 91-day cycles, for a total of 12 months and were instructed to start their study regimen on the first Sunday of their menses after enrollment.
The findings of this study included a Pearl Index for participants ages 18–35 years of 2.74 among all subjects and 1.77 among subjects who were compliant with the study regimen, as detailed in Table 2. Compliant use in this trial was defined as skipping fewer than 2 consecutive pills and having few missed pills overall. Life table analysis demonstrated pregnancy rates for women who completed at least one treatment cycle to be 2.19% among all users and 2.14% among compliant users. These numbers are based on 36 pregnancies that occurred after the onset of treatment and within 14 days after the last combined contraceptive pill. Both measures of contraceptive effectiveness are comparable to other combined hormonal contraceptive regimens. Post-hoc data analysis suggests no effect of weight or BMI on efficacy of the regimen. 21
Summary of pearl index calculations. Adapted from Kroll et al. 20

ITT, intent-to-treat of all subjects who completed at least one 91-day cycle.
PITT, pregnancy intent-to-treat; results in participants age 18–35 only
Although the study was not powered to detect specific side effects from hormonal contraception, the most commonly reported side effects included headache (33%), nasopharyngitis (16%), dysmenorrhea (11%), upper respiratory tract infection (8%), metrorrhagia (8%), nausea (8%), back pain (8%), sinusitis (7%) and urinary tract infection (5%). Five serious adverse events occurred over the course of the study and included headache and syncope, gastroenteritis, and three cases of gallbladder related illness in women with risk factors for gallbladder disease. No venous thromboembolic events were reported. Eighty-nine women reported bleeding and/or spotting as the reason for study discontinuation.
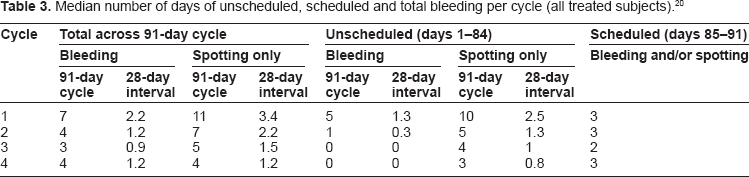
Women in this study also kept daily diaries to track their bleeding pattern. Table 3 provides the median numbers of scheduled and unscheduled bleeding days per 91-day cycle. Median scheduled withdrawal bleeding, defined as bleeding during the 7-day EE-only interval, lasted 2 to 3 days per cycle, with a significant proportion of women experiencing no bleeding requiring protection. In the first cycle of use, however, study participants had a median of 5 days of unscheduled bleeding. In cycle two, this median value decreased to one day, and to zero by cycles three and four. Unscheduled spotting, bleeding not requiring sanitary protection, decreased from a median of 10 days in cycle one to 3 days in cycle four.
Median number of days of unscheduled, scheduled and total bleeding per cycle (all treated subjects). 20
Place of Product in Therapy
The 91-day regimen of 84 days of 0.02 mg EE and 0.1 mg LNG plus 7 days of 0.01 mg EE provides a safe, effective option for women choosing COCs for contraception or other non contraceptive benefits. Although the regimen has not been directly compared in head-to-head trials, this formulation has similar effectiveness and side effects as other extended COC regimens. The current regimen is the only FDA approved 91-day contraceptive regimen containing 0.02 mg EE. COC formulations with 0.02 mg EE are associated with somewhat greater risk of bleeding disturbances than 0.03 mg or greater EE formulations, including both amenorrhea and irregular or unscheduled bleeding. Lower doses of estrogen and progestin, however, have the advantage of decreased hormone-related side effects. While safety is likely similar between formulations 0.02 mg EE and low-dose COCs with greater than 0.02 mg EE daily, lower doses COCs may also be associated with decreased nausea, breast tenderness, and mood changes. As with traditional 21/7 regimens, having a variety of options for contraception will allow women to select contraceptive regimens that they find most acceptable, balancing the bleeding patterns of lower dose formulations and extended cycles with the estrogen related side effects of the higher hormone doses.
As with other extended-cycle regimens, dosing in greater than monthly cycles may increase patient adherence to a regimen secondary to convenience of fewer pharmacy visits, which prevents delays in starting the next pill cycle, and a preference for decreased scheduled bleeding. When choosing among the dozens of contraceptive options currently available for women, patient preference and satisfaction are critical in selecting a regimen with the greatest chance of continued adherence, and hence decreased chance of unintended pregnancy or other morbidity, as possible.
The 84/7 regimen of 0.02 mg EE and 0.1 mg LNG plus 0.01 mg EE also is an option for women seeking the non contraceptive benefits of an extended COC regimen. These benefits primarily include those related to menstrual cycle control. Extended regimens have potential social and economic impact as treatment for medical conditions that are exacerbated by monthly menstrual cycling. Menstrual disorders are a leading cause of missed work among women. Each year, women with heavy menstrual bleeding are estimated to work 6.9% less than women with light or moderate bleeding, with an average estimated loss of wages of $1692.00 per woman annually. Women with heavy menstruation are also 1.45 times more likely to use health care and 72% more likely to miss work than women with lighter menstrual periods. Decrease in menstrual flow with less frequent, lighter menstrual periods may reduce some of the economic burden of menstrual disorders.22,23 With counseling that users should expect some increase in unscheduled bleeding that resolves over time, this regimen, along with other extended-cycle contraceptive products, may have great long term benefit both for women who suffer from heavy menstruation as well as for the people who rely up on them. Other non contraceptive benefits of this regimen, although not yet directly evaluated, include therapy for other menstruation related problems such as dysmenorrhea, chronic pelvic pain, as well as non gynecologic catamenial syndromes such as menstrual migraines or epilepsy.
Concluding Remarks
A 91-day COC regimen with 84 days of 0.1 mg LNG/0.02 mg EE plus 7 days of 0.01 mg EE is a safe, acceptable, and effective option for contraception and menstrual regulation. This is the first dedicated approved 91-day cycle COC formulated with 20 mcg EE. As this is a newer product in the panel of available COCs, the only published data regarding this regimen has been related to its efficacy and safety. From the pivotal trial, pregnancy rates, side effects, and bleeding patterns appear to be comparable to those found in trials evaluating similar regimens formulated with higher doses of the same hormone components. This study was not powered to specifically evaluate the incidence of rare side effects or compare the regimen to other COCs. Interpretation of this product's likely place in therapy, therefore, is based upon theory and findings in the contraceptive research literature. Theoretic concerns with lower dose (0.02 mg EE versus higher dose EE) of estrogen include greater breakthrough bleeding or decreased efficacy. Bleeding patterns for an extended-cycle regimen with 0.02 mg EE appear to be similar to those of a 0.03 mg EE extended regimen, 17 however direct comparisons have yet to be evaluated. Potential benefits of a lower dose EE regimen include theoretic decrease in adverse estrogen-related side effects due to lower estrogen exposure. Ultimately, however, patient satisfaction and continuation with this formulation will likely be directly affected by the woman's expectations and the counseling she receives from her health care provider regarding side effects.
Another salient finding in the pivotal trial of this regimen was no significant difference in effectiveness based upon weight or BMI. Concern exists for effectiveness of lower-dose COCs among obese and morbidly obese women. While two retrospective studies have demonstrated a higher failure rate of COCs among obese women, other retrospective cohort studies have not found the same difference when controlling for other patient characteristics.24–26 Although the evaluation and analysis of the impact of weight on contraceptive effectiveness was post hoc and based upon crude pregnancy rates, it suggests that this 0.02 mg EE 91-day regimen is appropriate for use in overweight and obese women. 21
Further evaluation of this regimen should focus on the questions that will help providers tailor appropriate counseling for women interested in an extended combined hormonal contraceptive regimen. These studies may include evaluation of follicular development and direct comparisons to other extended hormonal contraception regimens focusing on side effects and patient satisfaction and adherence.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
