Abstract
Keywords
Introduction
With the progressive increase of mining depth, most Chinese mines have the characteristics of high gas pressure, low permeability, and high in-situ stress (Cao et al., 2017; Chen et al., 2017). Therefore, these characteristics seriously restricted to exploit coal resources and gas resources. In China, extraction of protective coal layer and pre-mining coalbed methane (CBM) are used to prevent coal and gas outbursts. Some practices have proved that the extraction of protective coal layer is an economical and effective mean to prevent coal and gas outburst (Li, 2014; Wang et al., 2015; Yao et al., 2016; Yang et al., 2011). However, many mines do not have the condition of the extraction of protective coal layer, which limits the application of the extraction of protective coal layer. Pre-mining CBM has the technical features of wide application, low cost, easy construction and so on (Wang et al., 2016). However, the measure of pre-mining CBM needs a long time to achieve drainage reach standard for the low permeability coal seam (Wang et al., 2014; Zhang et al., 2017). In order to improve the effect of gas drainage for a single coal seam, water invasion, which includes water injection (Li et al., 2005; Song, 2004), hydraulic fracturing (Huang et al., 2017; Wang et al., 2014, 2017) and hydraulic slotting (Lin et al., 2015; Shen et al., 2012), is taken to increase the permeability of coal seam. After water invades into coal matrix, the influence of water on the gas desorption exists a disagreement. There are three insights about the influence of water on the gas desorption. The first insight is that high pressure water can increase the permeability of the coal seam, displace the free gas in the coal body and enhance the gas production for CBM (Cheng and Chen, 2014; Teng et al., 2016; Wei and Zhang, 2005). The second insight is that after liquid water invades into the coal matrix pores under the action of the capillary force, water and methane exist competitive absorption. Water is a polar molecule which can more easily get an absorption position in coal (Jiang et al., 2007; Qin et al., 2013; Zhang, 2011). The third insight is that water invades into the coal matrix pores under the action of the capillary force, and water can inhibit gas emission and reduce the methane diffusion rate (Xiao and Wang, 2011; Zhao et al., 2011). The above studies show that the addition of water affecting gas desorption during implementation of the hydraulic technology can include multiple effects such as increasing permeability, displacement, and inhibition. The three aspects of effects blend with each other. Therefore, the mechanism of controlling gas by hydraulic technology is unclear. In order to reveal the mechanism of controlling gas by hydraulic technology, the complex comprehensive function process of water affecting gas should be separated to research the mechanism and contribution degree of the single factor. Spontaneous imbibition is an important mechanism of oil production from fractured unconventional reservoirs (Tong et al., 2015; Wang et al., 2017). In porous media, the process of replacing a wetting phase fluid with a non-wetting fluid under the action of capillary force is called imbibition (Gao and Hu, 2016; Morrow and Mason, 2001; Zhou et al., 2002). However, coal has a double pore structure, which can form a complex capillary network (Naveen et al., 2017; Sharon et al., 2015). Therefore, the water displacement by the methane in coal is similar to oil displacement by imbibition in low permeability oil reservoir. Wang et al. (2015) use an imbibition test device of isobaric pressure adding water, which can eliminate displacement effect of high pressure injection, to separately study the imbibition effect of adding water in coal. The results show that water can displace the adsorbed gas by imbibition effect. However, the gas pressure in the coal sample tank rises after water invades into the coal, which results in a part of the free gas reabsorbing. The adsorbed gas will increase the gas pressure in the pores and prevent the movement of water in the coal. It cannot accurately simulate the imbibition effect of water in CBM. Therefore, laboratory studies on the dynamic response characteristics of water and methane during isobaric imbibition process remain to be reported.
In this paper, a self-designed experimental device of isobaric imbibition is used to analyze the dynamic response characteristics of water and methane during isobaric imbibition process in remolded coal containing methane. A soft coal seam has characteristics of low permeability and a small influence zone of corresponding to a single bore hole (Gao et al., 2015; Li, 2016; Yao et al., 2011). Soft coal is more likely to experience an outburst, whereas outbursts rarely occur in hard coal. This presents difficulty in removing a coal core from a soft coal seam. Therefore, the remolded coals are used in this study. Therefore, this study focuses on what response characteristics of water and methane during isobaric imbibition process and the isobaric imbibition can lay a theoretical foundation for revealing the mechanism of controlling gas by hydraulic technology, and provide a new method to control gas in deep coal seams.
Materials and methodology
Physical parameters of coal
The anthracite samples used in this study are obtained from the Yonghong coal mine, Jincheng, located in the Shanxi province of China. The physical parameters of coal are evaluated using Chinese national standards (Table 1): ash content (
Physical parameters of coal.

Production of experimental coal samples
The remolded coal with hollow center is compressed with an electro-hydraulic servo universal machine controlled by an EHC-3100 microcomputer. The process of forming the remolded coal with hollow center is as follows:
The coal specimen is then ground and sieved with three different sizes (0.25–0.5 mm, 0.2–0.25 mm, less than 0.25 mm). Three different sizes of coal particles ((0.25–0.5) mm: (0.2–0.25) mm: (<0.2) mm = 2:2:1) are mixed with 20% distilled water, and the wet coals are stirred thoroughly. The wet coals are placed in a mold, and the electro-hydraulic servo universal machine controlled by an EHC-3100 microcomputer is used to compress the remolded coal with hollow center under a load of 200 kN for 20 min. The EHC-3100 microcomputer controlling the electro-hydraulic servo universal machine drops out the remolded coal with hollow center. The resulting remolded coal with hollow center sample is shown in Figure 1. As shown in Figure 1, the remolded coal with hollow center is 50 mm in diameter and 80 mm in height and the hollow center is 22 mm in diameter and 80 mm in height. The purpose of the remolded coal with hollow center is to test the imbibition effect. The hollow center is designed to hold the water container for the imbibition test.
Experimental remolded coal sample with hollow center sample.

Experimental device
The experimental device includes a vacuum degassing system, a thermostatic system, an inflation system, an adsorption equilibrium system, an isobaric pressure relief system, an isobaric pressure adding water system, an desorption system, and a data acquisition system. The vacuum degassing system consists of a vacuum pump and a compound vacuum gauge. The range of the compound vacuum gauge is between 1 × 10−2 and 1 × 105 Pa. The thermostatic system controls the adsorption equilibrium system with a thermostatic water bath and simulates the CBM reservoir temperature. The inflation system includes a high pressure methane cylinder with 99.99% purity. The adsorption equilibrium system includes a special coal sample tank, a reference tank, and some pressure sensors. The isobaric pressure relief system is shown in Figure 2.
Isobaric pressure relief device.
As shown in Figure 2, the isobaric pressure relief system is to achieve constant pressure regulator by opening and closing the built-in rubber hose. The wall thickness of the rubber hose is thin, and it can rapidly achieve opening and closing. The isobaric pressure relief device overcomes the problem of insensitive pressure relief of one-way valve. The rubber hose is covered by a casing with some holes. The principle of the isobaric pressure relief system is described as follows. A certain amount of water is added to the isobaric pressure relief system until the water can submerge the rubber hose. A certain amount of high pressure gas is filled into the isobaric pressure relief system until the number of pressure sensor achieves
Isobaric pressure adding water system includes a coal sample tank, remolded coal with hollow center, and a glass bottle of water storage. The glass bottle of water storage is pre placed inside the remolded coal with hollow center. When water needs to be added to the coal sample, the needle valve should be rotated and the glass bottle of water storage is broken under the action of cylinder block of micro protrusion. Therefore, water is added to the coal sample tank under the condition of isobaric pressure. The volume of free space in adsorption system will not change until water is added to the coal sample tank. The isobaric pressure adding water system simulates the imbibition effect of water-based measures, which is different from traditional hydraulic measures. The desorption system is a desorption instrument. The data acquisition system includes a high accuracy pressure sensor, data acquisition module, and computer host and screen, which are used to test the methane pressure of each system. The experimental system mechanism of isobaric imbibition is shown in Figure 3.
Schematic setup for isobaric imbibition. (1) High pressure methane cylinder; (2) data acquisition system; (3) reference tank; (4) compound vacuum gauge; (6), (7) and (18) pressure sensors; (8) rubber hose; (9) isobaric pressure relief system; (10) needle valve; (11) sealing ring; (12) tank; (13) remolded coal; (14) cylinder block of micro protrusion; (15) water; (16) glass bottle of water storage; (17) cover; (19) isobaric pressure adding water system; (20) desorption instrument; (21) thermostatic water bath; a–i–valves.
Experimental methods
The isobaric imbibition experiment is conducted by a self-designed experimental device (Figure 3). The general procedure for the isobaric imbibition process is shown below.
Drying experimental coal samples: To remove water from the remolded coal with hollow center so that it does not affect the test results, the remolded coals with hollow center are placed in an oven at a constant temperature of 378.15 K. During drying, the remolded coals are weighed every 30 min until their weight achieve stabilization, after which they are stored in a dry container for later use. Adding water to the glass bottle of water storage: The quality of water is obtained by the quality of dried remolded coal with hollow center multiplying water content. After water is added to the glass, if the glass bottle of water storage has remaining space, it can be filled with liquid paraffin. After paraffin wax is solidified, the upper surface of solid paraffin wax and water storage glass bottle cap will be sealed with AB glue. Vacuum degassing: The remolded coal (13) is placed into the coal tank (12). The cylinder block of micro protrusion (14) is placed in the bottom of the remolded coal with a center hole. The glass bottle of water storage (16) is placed on cylinder block of micro protrusion (14). Calibrate the sample cell volume and double-check the tightness of the whole test system. When the number of compound vacuum gauge is below 20 Pa, the degassing process is stopped. Adsorption equilibrium: The thermostatic system is adjusted to 303.15 K and maintained a constant temperature during the entire test. All the valves (a–i) are closed in the experimental device of isobaric imbibition. The valve (a) is opened and the reference tank (3) is inflated by high pressure methane cylinder (1). When the pressure of the reference tank is stable which can be obtained by the pressure sensor (6), the valve (a) can be closed. The valves (b, e and g) are opened, and the coal sample tank (19) is inflated by the reference tank (3). Then, the valves (b, e and g) are closed. In order to reach adsorption equilibrium at a predefined pressure, the process of inflating usually goes through multiple times. The equilibrium process typically takes around 10 to 15 h. Adding water to the isobaric pressure relief system: A certain amount of water is added to the isobaric pressure relief system until the water can submerge the rubber hose. The valve (a) is opened, and the reference tank (3) is inflated by high pressure methane cylinder (1). When the pressure of the reference tank (3) achieves stabilization, which can be monitored by the pressure sensor (6), the valve (a) should be closed. The valves (b, d and i) are opened, and the isobaric pressure relief system (9) is inflated by the reference tank (3) until the pressure of the isobaric pressure relief system is the same as the absorption equilibrium pressure. Adding water under isobaric pressure: The needle valve (10) is rotated and the glass bottle of water storage (16) is broken under the action of cylinder block of micro protrusion (14). Therefore, water is added to the coal sample tank under the condition of isobaric pressure. When water invades into the remolded coal containing methane, the valves (g and f) should be opened. If the pressure of coal sample tank is greater than the adsorption equilibrium pressure, the rubber hose (8) will be opened, and the free gas will be released to the outside of the coal sample tank. If the pressure of coal sample tank is equal to the adsorption equilibrium pressure, the rubber hose (8) will be closed. The pressure of the coal sample tank is guaranteed to be a relatively stable value by the isobaric pressure relief system. Data acquisition: The amount of desorption gas is collected by a desorption instrument which is recorded at regular intervals. If the imbibition quantity is less than 0.007 mL/g within 2 h, the testing process is deemed complete.
Results and discussion
Dynamic response characteristics of water and methane during isobaric imbibition process
This experiment tested the isobaric imbibition process of remolded coal samples with the adsorption equilibrium pressures of 1.5 MPa and 2.5 MPa and water contents of 2%, 4%, 6%, 8%, and 10%. As shown in Figure 4, after water is added to the coal sample tank, the imbibition quantity and free gas pressure initially increase rapidly with increasing time, after which the growth rate gradually decreases. If free gas pressure in the coal sample tank is greater than initial adsorption equilibrium pressure, the rubber hose will be opened. The free gas is released through the isobaric pressure relief system, and the rubber hose will be closed until the free gas pressure in the coal sample tank is equal to initial adsorption equilibrium pressure. Under the same adsorption equilibrium pressure, the larger imbibition quantity increases with increasing water content. Finally, the imbibition quantity reaches a maximum value. When the adsorption equilibrium pressure is 2.5 MPa, the maximum imbibition quantity increase by 0.65, 0.73, 0.9, 0.42 and 0.46 mL⋅g−1 at the water content of 2%, 4%, 6%, 8%, and 10%, respectively, compared to the adsorption equilibrium pressure of 1.5 MPa.
The isobaric imbibition quantity under the conditions of different water contents. (a) Adsorption equilibrium pressure 1.5 MPa; (b) adsorption equilibrium pressure 2.5 MPa.
This experiment also tested the isobaric imbibition process of remolded coal samples with water contents of 4% and 10% and the adsorption equilibrium pressures of 1.0, 1.5, 2.0, 2.5, and 3.0 MPa. As shown in Figure 5, the imbibition quantity initially also increases rapidly with increasing time, after which the growth rate gradually decreases. Under the same water content, the imbibition quantity increases with increasing adsorption equilibrium pressure. Finally, the imbibition quantity reaches a maximum value. When water content is 10%, the maximum imbibition quantity increase by 2.12, 2.14, 1.92, 1.87, and 1.91 mL⋅g−1 at the adsorption equilibrium pressures of 1.0, 1.5, 2.0, 2.5 and 3.0 MPa, respectively, compared to water content 4%.
The isobaric imbibition quantity under the conditions of different adsorption equilibrium pressures. (a) Water content 4%; (b) water content 10%.
During this experiment (Figures 4 and 5), water enters the coal sample only by spontaneous imbibition rather than the external injection pressure. The water can permeate the coal, improve the water saturation, and enhance the gas recovery in the coal matrix. Furthermore, the void volume in the coal sample tank does not change after breaking the glass bottle of water storage because the water is kept in coal sample tank in advance.
Changes in maximum imbibition quantity and rate with different water contents. (a) Adsorption equilibrium pressure 1.5 MPa; (b) adsorption equilibrium pressure 2.5 MPa.
Quantification of the maximum imbibition quantity and maximum imbibition rate
The maximum imbibition quantity, defined as the maximum imbibition gas quantity per gram of coal, means that maximum imbibition quantity after adding water to coal sample tank (mL·g−1) is

In order to evaluate the imbibition intensity under the condition of different water contents, the imbibition rate is introduced. The imbibition rate (%) can be expressed as follow

Based on equations (1) and (2), the maximum imbibition quantity and the maximum imbibition rate with different water contents are shown in Figure 6. The maximum imbibition quantity and maximum imbibition rate with different adsorption equilibrium pressures are shown in Figure 7. As shown in Figures 6 and 7, X axis, Y axis, and Z axis represent water content, maximum imbibition quantity, and maximum imbibition rate, respectively. The scatter points in the XOY plane and the XOZ plane are obtained by the scatter point projection in the three-dimensional coordinate system space.
Changes in maximum imbibition quantity and rate with different adsorption equilibrium pressures. (a) Water content 4%; (b) water content 10%.
As shown in Figure 6, the maximum imbibition quantity and the maximum imbibition rate increase with increasing water content under the same adsorption equilibrium pressure, but the increasing extent decreases. The remolded coal consists of various pores, which can be considered capillary tubes. However, some capillary tubes are not continuous. In these capillary tubes, the added water stays in a discontinuous position and will not continue to flow. The imbibition height of water has a limit value because of gravity and gas pressure. Therefore, the imbibition process will stop when the imbibition height of water reaches a limit value. In order to quantitatively evaluate the dynamic response characteristics of water and methane and forecast the change trend, the relationship between water content and maximum imbibition quantity can be described by a Langmuir function given in equation (3). The relationship between water content and imbibition rate also can be described by a Langmuir function given in equation (4). The fitting curves are shown in Figure 8, and the fitting parameters are shown in Table 2


Fitting parameters.

Fitting parameters of equations (5) and (6).

As shown in Figure 7, the maximum imbibition quantity increases with increasing adsorption equilibrium pressure under the same water content, but the increasing extent decreases. The relationship between the maximum imbibition quantity and adsorption equilibrium pressure can be described by a Langmuir function given in equation (5). However, the maximum imbibition rate decreases with increasing adsorption equilibrium pressure under the same water content. For a water content of 4%, the maximum imbibition rate is 14.02%, 13.94%, 13.76%, 13.63%, and 13.55% when the adsorption equilibrium pressure is 1.0, 1.5, 2.0, 2.5, and 3.0 MPa, respectively. For a water content of 10%, the maximum imbibition rate is 24.94%, 24.15%, 22.58%, 21.51%, and 20.68%, respectively. The difference indicates that the maximum imbibition rate is more sensitive to the water content. The relationship between the maximum imbibition rate and adsorption equilibrium pressure can be described by an exponential function given in equation (6). The fitting parameters of equations (5) and (6) are shown in Table 3. Based on equations (3) and (5), the maximum imbibition quantity has a limit value under the same water content or the adsorption equilibrium pressure
Schematic diagram of water molecule interacting with adsorbed methane.


Mechanisms of the dynamic response characteristics of water and methane
Based on the experimental results in Figures 4–7, the following mechanisms are proposed to interpret the dynamic response characteristics of water and methane.
The CBM has absorbed form and free form. The interaction force between coal and methane is Van der Waals' force, and the methane molecules are nonpolar molecules. Therefore, the adsorption of methane in the pores of the coal is physical adsorption. Water molecules are polar molecules. The interaction force between coal and methane is Van der Waals' force and hydrogen bond, and the hydrogen bond is greater than Van der Waals' force (Hu et al., 2018; Joubert et al., 1974). The rich specific surface area of coal can provide a space for adsorption of water molecules and methane molecules. After water is added to the coal containing gas, one part of water replace the free methane, but the pressure of the coal sample tank will not increase. Since the total volume of free gas has not changed, the other part of water will be sucked into nano pores under the action of the capillary force. Water molecule and methane molecule will produce competitive adsorption. Some adsorbed gas will be displaced by adding water, which will be released by the isobaric pressure relief system. Owing to the oxygen-containing functional groups in coal and the hydrogen bond of water, water can easily occupy high-energy sites and only the low-energy sites are available for methane (Gensterblum et al., 2014; Mahajan and Walker, 1971; Nishino, 2001; Ozdemir and Schroeder, 2009; Song et al., 2017). Water is adsorbed on the surface of coal as a continuous water molecule layer, and further reacts with coal molecules at a microscopic level. Therefore, the dynamic equilibrium status of methane adsorption will be changed with the imbibition of water; water interacting with adsorbed methane in nano pore is shown in Figure 8. If water content is increased under the same adsorption pressure, it is difficult to adsorb more and more water molecules in the pores of the coal. Since the adsorption of water molecule in the pore of the coal gradually decreases. Therefore, the increasing extent of imbibition quantity with increasing water content under the same adsorption equilibrium pressure gradually decreases. The gas adsorption quantity is the same under different water content. Hence, the increasing extent of maximum imbibition rate with increasing water content under the same adsorption equilibrium pressure also gradually decreases.
A schematic diagram of imbibition in a single capillary tube.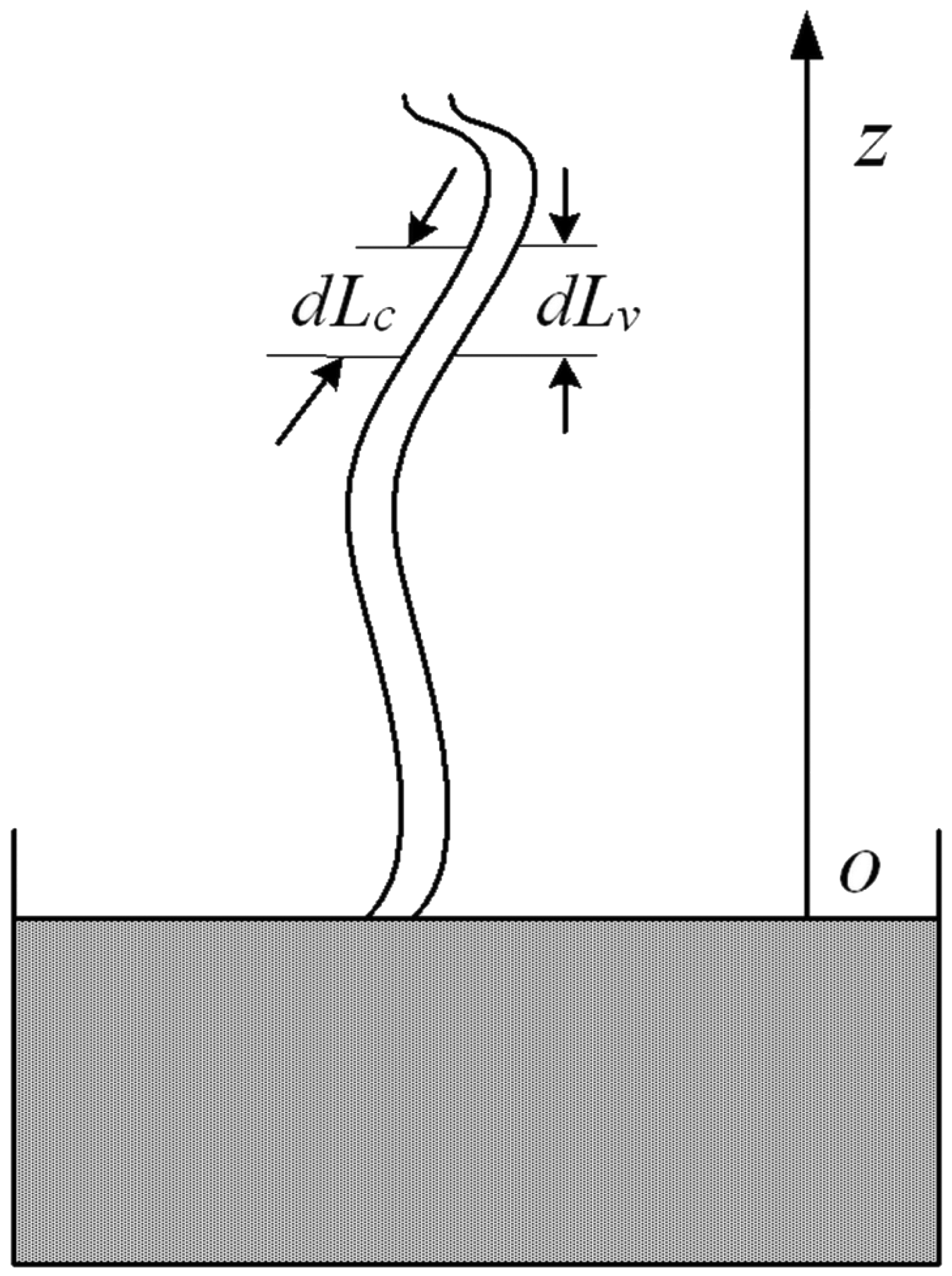
Equation (7) is Hagen–Poiseuille equation which represents the movement of liquid under the action of capillary force and gravity. Coal has a double pore structure, which can form a complex capillary network. Therefore, equation (7) can be used to describe the movement of water in coal without gas. As shown in Figure 9, when a curved capillary is in contact with a wetting phase liquid, the liquid rises under the action of the capillary force and its own gravity. Capillary force is a driving force. Since the capillary is curved, the imbibition height is not equal to water absorption length. For a vertical capillary, imbibition height is equal to water absorption length. When the capillary force is equal to the gravity given in equation (8), the imbibition height reaches the maximum value
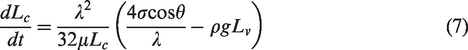

The movement of water is affected by gravity, capillary force, and pore gas pressure in the coal containing methane. Equation (9) can be used to describe the movement of water in the coal containing methane. When capillary force, gravity, and pore gas pressure reach a balance state, the imbibition height reaches the maximum value. The water stops moving in the pores of the coal, that is, it can no longer displace the adsorbed gas, and the imbibition quantity will reach a maximum value
A schematic diagram for drainage borehole and water inlet borehole in deep coal seam.

The maximum imbibition quantity increases with increasing the adsorption equilibrium pressure under the same water content. This is principally because there is more methane absorption and closer distributions on the same surface. A water molecule in the same space can displace more methane molecules. If the adsorption equilibrium pressure is increased under the same water content, the gas pressure in the pore will increase. Therefore, the adsorption of water into the nano pore becomes difficult, and the increasing extent of maximum imbibition quantity with increasing adsorption equilibrium pressure under the same water content gradually decreases. If all the water molecules occupy the adsorption sites of the coal body, the increasing adsorption equilibrium pressure will no longer affect the maximum quantity. The maximum imbibition quantity and methane absorption quantity both increase with equilibrium pressure. However, the increasing extent of methane absorption quantity is greater than that of the maximum imbibition quantity. Therefore, the maximum imbibition rate decreases with increasing the adsorption equilibrium pressure under the same water content. Compared to the adsorption equilibrium pressure, the water content has a greater effect on the imbibition quantity and imbibition rate.
Engineering application of isobaric imbibition
The isobaric imbibition process in the remolded coal containing methane really simulates the process of adding water invasion into the coal. The experimental results mentioned above have proved that the addition of water can displace the absorbed methane. Therefore, the water invasion into the coal can be used to prevent and control CBM. The deep coal seam has characteristics of high situ stress, high gas pressure, and low permeability. The applicability of increasing permeability measures is poor for deep coal seam. Since, high situ stress causes the fracture to be closed again. Therefore, single borehole drainage method is not suitable for the deep coal seam, and the water imbibition and borehole drainage method together are an effective method to the deep coal seam. However, some problems need to be paid attention to.
Coal seams are heterogeneous, and the permeability of some areas is slightly good and the permeability of some areas is poor (Moore et al., 2015; Peng et al., 2017; Yin et al., 2018). After addition of water invades into the coal seam, the displaced methane can easily flow out through coal seam fractures to a region with a slightly good permeability. Therefore, the gas content can decrease and the borehole drainage method is not needed. However, the displaced gas is hard to flow out to the area of poor permeability, and a high gas pressure region will be formed around the adding water area, which is not conducive to CBM control. Water imbibition and borehole drainage method together are needed to be considered. On the one hand, the borehole drainage method can make the free gas flow out of the coal seam, which can reduce the gas pressure and gas content within the influence range of gas drainage borehole. On the other hand, water imbibition can promote desorption. The reasonable arrangement distance of the borehole is important to the borehole drainage method which can reduce the CBM content. However, the blank zone of borehole gas drainage is a common problem in CBM control. If the borehole spacing is too large, the number of boreholes is too low, leading to unsatisfactory gas drainage. Some accidents may happen in the high gas pressure area; if the borehole spacing is too small, too many boreholes are needed, causing waste of manpower and material resources (Chen et al., 2018; Ma et al., 2009; Si et al., 2018; Wang et al., 2012). A schematic diagram for drainage borehole and adding water borehole in deep coal seam is shown in Figure 10. As shown in Figure 10, the water inlet borehole should be within the influence range of gas drainage borehole. After water is added to the borehole, some absorbed gas will be displaced. The free gas begins to spread around. If the water inlet borehole is not within the influence range of gas drainage borehole, some free gas cannot flow out, which will form a high gas pressure zone around the water inlet region. Therefore, a suitable borehole spacing is important to reduce CBM content. Investigation of the dynamic response characteristics of water and methane during isobaric imbibition process in remolded coal containing methane can present a theoretical foundation for the application of water imbibition to control gas in deep coal seams.
Conclusions
The dynamic response characteristics of water and methane are tested by a self-designed experimental system for isobaric imbibition. After the water is added to the remolded coal, it can displace the adsorbed methane and increase the free gas. The imbibition quantity increases with increasing water content under the same adsorption equilibrium pressure, and the imbibition quantity also increases with increasing adsorption equilibrium pressure under the same water content. However, the increasing extent of imbibition quantity is decreasing. Finally, the imbibition quantity reaches a maximum value.
The maximum imbibition quantity and the maximum imbibition rate increase with increasing water content under the same adsorption equilibrium pressure, but the increasing extent decreases. The relationship between water content and maximum imbibition quantity or the maximum imbibition rate can be described by a Langmuir function.
The maximum imbibition quantity increases with increasing adsorption equilibrium pressure under the same water content, but the increasing extent decreases. The relationship between maximum imbibition quantity and adsorption equilibrium pressure can be described by a Langmuir function. However, the maximum imbibition rate decreases with increasing adsorption equilibrium pressures under the same water content. The relationship between maximum imbibition rate and adsorption equilibrium pressure can be described by an exponential function. Compared to the adsorption equilibrium pressure, the water content has a greater effect on the imbibition quantity and imbibition rate.
Based on the transformation form of Hagen–Poiseuille equation, adsorption force of coal and gas and adsorption force of coal and water, the dynamic response characteristics of water and methane during isobaric imbibition process were revealed. Water imbibition can displace the absorbed methane. The borehole drainage method can make the free gas flow out of the coal seam, which can reduce the gas pressure and gas content within the influence range of gas drainage borehole. Water imbibition and borehole drainage method together can be used to control gas in deep coal seams.
