Abstract
Keywords
Introduction
Coal and gas outburst is a complex dynamic phenomenon in underground coal mines that has occurred frequently over the past 150 years, and coal and gas outbursts is the main factor affecting safe coal mine production (Du et al., 2020). The coal-bearing strata in China are mainly Carboniferous Permian and Jurassic and mostly occur in the form of coal seam groups, which are produced by complex geological tectonic movements. Under the action of tectonic stress, a composite coal seam with soft and hard coals has formed, and is widely distributed throughout the country, such as in the provinces of Henan, Shanxi, Guizhou, and so on (Hao, 2020). Soft and hard composite coal seams exhibit uneven changes in softness and hardness, and poor stability. The total pore volume, macropore and medium-pore volume, and pore connectivity of soft coal are larger than those of hard coal (Tian et al., 2018); therefore, the gas diffusion rate of soft coal is faster (Gu et al., 2016). Owing to poor air permeability, poor mechanical properties, easy collapse of drainage boreholes, and poor drainage effect, the effect of composite coal seam gas control is poor, and coal and gas outbursts are prone to occur.
In recent years, the technology of coal seam liquid injection and freezing to prevent coal and gas outburst has been applied (Xie, 2010). Freezing holes are arranged in the coal seam, and the coal seam’s heat conduction allows the coal seam temperature to reach the freezing temperature, reducing gas pressure (Wang et al., 2015,) and improving the mechanical properties of the coal seam (Qin et al., 2022), and reducing the risk of coal and gas outburst (Zhou and Zhai, 2021) analyzed the influence of liquid nitrogen freezing on the mechanical properties of coal and determined the elastic modulus, compressive strength, and tensile strength of frozen coal. Wang (2018) found that the moisture content had a significant impact on the strength of coal under the action of low temperature. Zhu et al., (2021) conducted a series of direct shear tests on the frozen coal-rock interface under various test temperatures, coal moisture content and normal stress. Low-temperature freezing coal can inhibit gas desorption (Wang et al., 2018), and reduce coal gas pressure and the risk of coal and gas outbursts. Wang (Wang et al., 2021) found that, as the temperature decreases, the dynamic viscosity of the molecule and the mean free path of the gas molecules also decreases, and the methane molecules cannot easily separate from the surface of the coal matrix, which manifests as an increase in the adsorption amount. Wang et al.(2011, 2018) found that the coal gas desorption rate under low-temperature conditions is slower than that under normal temperature and variable temperature conditions. Yue et al. (2014) investigated the change law of the coal gas diffusion coefficient in a low-temperature environment. Yue et al. (2019) compared the adsorption capacity of coal at different temperatures and found that low temperatures inhibit gas desorption. (Lou et al., 2015) and (Ma et al., 2019) investigated the coal gas adsorption at low temperatures and found that, as the temperature decreased, the amount of gas adsorption increased. (Qin et al., 2020) found that the amount of coal gas adsorption gradually increased as the liquid nitrogen freezing and thawing time increased.
At present, liquid injection freezing and outburst prevention measures are mainly applied to uncover coal in cross-cut (Xie et al., 2011 and 2012), and previous studies focused on investigating the gas desorption/adsorption evolution law of coal samples with the same hardness under different low-temperature environments. However, these studies that on freezing outburst prevention in composite coal seams are rare. Therefore,In the process of low-temperature freezing, the evolution of the temperature field and gas pressure field of gas-bearing coals with different hardnesses urgently need to be further studied. Based on the independent design of the experimental device for identifying the freezing response characteristics of gas-containing coal, this study measured the evolution of gas pressure under different freezing temperatures and different adsorption equilibrium pressures for coal samples with different hardness in composite coal seams. The findings of this study have important theoretical guidance for the prevention and control of coal and gas outbursts in composite coal seams.
Coal sample preparation and test process
Coal sample preparation and test device
Coal sample preparation
Coal samples with different firmness coefficients (f < 0.5, f > 0.5) from the No. 3 composite seam of Yuxi Coal Mine in Jincheng, Shanxi Province, were selected as the experimental research objects. After their collection, the samples were sealed, immediately stored, and sent to the laboratory. The basic parameters of the coal samples are listed in Table 1. The raw coal sample was crushed by a pulverizer and sieved to 0.18–0.25 mm. An electronic scale was used to weigh the quantity of coal powder, a certain proportion of distilled water was added for complete mixing, stirring was carried out to evenly distribute the water, and the coal sample was placed in a self-made abrasive tool. Load control was adopted on a universal servo machine to achieve the stable pressure of 15 MPa (equal to the ground stress of the coal seam). After the pressure was maintained for 6 h, the coal was pressed into a briquette with a size of Φ50 mm × 100 mm. And place the briquette in a drying box at 105°C for drying until the quality of the coal sample no longer changes.
Basic parameters of coal samples.

Experimental device
The test used a self-made gas-containing coal freezing response characteristic experimental device (Figure 1). The main components, vacuum tubes, and vacuum gauges are mainly responsible for the vacuum degassing of gas-filled tanks, coal sample tanks, and experimental pipelines. During the simulation experiment, the volume calibration of the experimental pipelines, gas-filled tanks, and coal sample tanks was carried out by the system, and the main function of the pressure-reducing valve and cylinder gas source (CH4) was to fill the coal sample tank with methane gas to ensure that the coal and methane in the coal sample tank were adsorbed and balanced at a certain pressure. The main components of the data acquisition system are the temperature sensor and pressure gauge. The function of the refrigeration system was to use absolute ethanol (99% concentration) to provide a cold source for the coal sample tank.

Self-made experimental device for freezing response characteristics of gas-containing coal.
Experimental process
Vacuum degassing: The dry coal sample was placed into the coal sample tank, all valves of the device were closed, the vacuum pump started, the coal sample tank outlet and pipeline valves were slowly opened, and the coal sample and pipeline were vacuum degassed. When the vacuum gauge was less than 10 Pa, the outlet valve of the coal sample tank and the vacuum pump were closed to finish the degassing procedure.
Inflatable adsorption balance: The constant-temperature water bath was adjusted to 28 °C. The coal sample tank was placed in a constant temperature water bath and left undisturbed until the temperature of the coal sample increased to 28 °C and remained unchanged. Then, a gas source and the pressure reducing valve in the steel cylinder of the experimental device were used to obtain the freezing response characteristics of the gas-containing coal (Figure 1). The tank was filled with gas such that the equilibrium pressure of gas adsorption in the coal sample tank reached 1.0 MPa and remained unchanged for 24 h.
Monitoring of temperature and gas pressure changes at different temperature measurement points: Absolute ethanol (concentration of 99%) was added to the low-temperature freezing tank, the refrigeration system was turned on, the refrigeration temperature was adjusted to −10 °C, the coal sample tank with the briquette was placed into the freezing tank, and the temperature of the coal sample was monitored after 28 °C. The gas pressure of the briquette in the coal sample tank was measured at different times until the temperature reached −10 °C.
According to the previous steps, different freezing temperatures (-15 °C, −20 °C, −25 °C, −30 °C), different gas adsorption equilibrium pressures (1.5 MPa, 2.0 MPa), and different hardness (hard coal, soft coal) were considered, and the response characteristics of gas pressure were obtained during freezing.
Changes in gas pressure of gas-containing coal at different freezing temperatures
To investigate the characteristics of the influence of the freezing temperature on the coal sample gas pressure during the freezing process, the adsorption equilibrium pressure of hard coal samples was tested at 1.0 MPa and different freezing temperatures (-10 °C, −15 °C, −20 °C, −25 °C, −30 °C), and the gas pressure evolution characteristics were obtained. The measurement results are shown in Figure 2.

Variation curve of gas pressure at different freezing temperatures during freezing.
As shown in Figure 2, at the same adsorption equilibrium pressure, the change trend of the coal sample gas pressure at different freezing temperatures is similar. As the temperature of the coal sample gradually decreased to the refrigeration temperature, the gas pressure underwent three stages: rapid decline, steady decline, and slow decline. Let us consider the change of gas pressure in a coal sample tank at the freezing temperature of −10 °C as an example: 0∼13min, the gas pressure drops rapidly from 1.0MPa to 0.6MPa, and the pressure drop is 0.4MPa, accounting for 70.18% of the total pressure reduction; 13∼23min, the gas pressure drops steadily to 0.5MPa, and the pressure drop is 0.1MPa, accounting for the entire pressure reduction. The pressure range is 17.54%; 23∼45.5min, the gas pressure slowly drops to 0.43MPa, and the pressure reduction range is 0.07MPa, accounting for 12.28% of the total pressure reduction range. As the temperature decreased, the amount of gas adsorption increased, which manifested as a decrease in the gas pressure of the coal sample tank. At the beginning of freezing, the coal sample tank contained a large number of free gas molecules. Owing to the large temperature difference of the coal sample, the heat transfer rate of the coal matrix was high. The gas molecules were adsorbed because the molecular kinetic energy was reduced to less than the adsorption potential well on the surface of the coal matrix, and a large amount of gas was adsorbed. The gas pressure in the coal sample tank rapidly decreased. As the cooling process progressed, the free gas molecules decreased, the coal sample gradually reached the saturation adsorption, and the gas pressure adsorption gradually decreased, which manifested as a slow decrease in gas pressure.
As the freezing temperature decreased, less time was required to drop to the same gas pressure, and the gas pressure of the coal sample tank was lower at the same time. According to the rules and regulations of coal and gas outburst, the coal seam gas pressure of Chinese mines is greater than 0.74 MPa, which means that these mines are outburst mines. Different freezing temperatures can reduce the gas pressure to below 0.74 MPa, which effectively prevents coal and gas outbursts. The freezing temperature is −10 °C to −30 °C, and the time required to decrease to 0.74 MPa is 6.5 min, 6 min, 5 min, 4 min, and 3.5 min. When freezing for 30 min, the gas pressure is 0.53 MPa, 0.48 MPa, 0.43 MPa, 0.38 MPa, and 0.33 MPa, respectively. As the freezing temperature decreased, the difference between the initial temperature of the coal sample and the freezing temperature became greater, the freezing effect during the cooling process became stronger, and the gas adsorption rate and gas pressure reduction rate become faster. Moreover, the coal matrix shrank, and the porosity and the pore surface area increased. As the gas adsorption capacity increased, the saturated adsorption capacity of the coal sample became stronger. Therefore, during the freezing process, the gas pressure of the coal sample tank will be lower with a lower freezing temperature.
When the gas-containing coal sample had the same adsorption equilibrium pressure at different freezing temperatures (-10 °C, −15 °C, −20 °C, −25 °C, −30 °C), the gas pressure of the coal sample tank changed as a monotonously decreasing function. The relationship between the gas pressure and freezing time is a power function, and its expression is given by Equation (1); all degrees of fit are above 0.99. The relevant fitting parameters are presented in Table 2.
Gas pressure-freezing time fitting parameters of coal sample tanks with different freezing temperatures.

As presented in Table 2, as the freezing temperature decreased, the gas pressure attenuation coefficient of the coal sample tank gradually increased by 0.089, 0.092, 0.095, 0.109, and 0.124, respectively; that is, the gas pressure decreased faster. When the frozen coal sample was saturated to adsorb gas, the gas pressure of the coal sample tank gradually decreased. With a lower freezing temperature, the freezing effect became stronger and the gas adsorption rate and the pressure reduction rate of the coal sample tank became faster. The coal matrix shrank, the porosity increased, and the pore surface area also increased. The adsorption capacity of the coal sample increased with the gas adsorption capacity.
Change of gas pressure of gas-bearing coal under different adsorption equilibrium pressures
To investigate the effect of the adsorption equilibrium pressure on the gas pressure of the coal sample tank during the freezing process, the hard coal samples were tested at the freezing temperature of −25 °C under different adsorption equilibrium pressures (1.0 MPa, 1.5 MPa, 2.0 MPa). The evolution characteristics of the tank gas pressure and the results are shown in Figure 3.

Changes of gas pressure in coal sample tanks with different adsorption equilibrium pressures during freezing.
As shown in Figure 3, the freezing temperature was −25 °C, the coal had the same hardness, and the change trend of the gas pressure of the coal sample under different adsorption equilibrium pressures was similar. As the temperature of the coal sample gradually decreased to the freezing temperature, the gas pressure underwent three phases: a rapid drop, a steady drop, and a slow drop. Take the change of gas pressure in a coal sample tank with an adsorption equilibrium pressure of 1.5MPa as an example: 0∼13.5min, the gas pressure drops rapidly from 1.5MPa to 0.85MPa, and the pressure drop is 0.65MPa, accounting for 65% of the entire pressure drop; 13.5∼31min, the gas The pressure drops steadily to 0.6 MPa, with a pressure drop of 0.25 MPa, accounting for 25% of the total pressure drop; 31∼57.5 min, the gas pressure slowly drops to 0.5 MPa, with a pressure drop of 0.1 MPa, accounting for 10% of the total pressure drop.
With the greater equilibrium pressure of gas adsorption, the gas pressure of the coal sample tank took longer to drop to 0.74 MPa. At the end of freezing, the gas pressure in the coal sample tank was greater. The gas adsorption equilibrium pressure was 1.0 MPa, 1.5 MPa, and 2.0 MPa, and the time required for the gas pressure to decrease to 0.74 MPa was 4 min, 19 min, and 49 min, respectively. Therefore, when the adsorption equilibrium pressure was larger, a lower freezing temperature was required to quickly reduce it to 0.74 MPa. At the end of freezing, the gas pressure in the coal sample tank was 0.27 MPa, 0.5 MPa, and 0.72 MPa, respectively. Because the freezing temperature was the same and the coal matrix has a certain adsorption capacity, the adsorption equilibrium pressure and the gas pressure of the coal sample tank increased.
Under the same freezing temperature and different adsorption equilibrium pressures (1.0 MPa, 1.5 MPa, 2.0 MPa) for the gas-containing coal sample, the gas pressure of the coal sample tank changed as a monotonously decreasing function and its relationship with the freezing time can be described by a power function. Equation (1) shows that all degrees of fit are above 0.99, and the relevant fitting parameters are listed in Table 3.
Gas pressure-freezing time fitting parameters of coal sample tanks with different adsorption equilibrium pressures.

As presented in Table 3, as the adsorption equilibrium pressure increased, the gas pressure attenuation coefficient of the coal sample tank gradually decreased by 0.109, 0.075, and 0.072; that is, the gas pressure decreased slower. Because the freezing temperature is the same, the coal matrix has a certain ability to absorb gas, and with higher adsorption equilibrium pressure, the gas molecules in the coal sample tank increased, the gas adsorption speed became slower.
Changes in gas pressure of gas-bearing coals with different hardness
To investigate the effect of coal hardness on the gas pressure of the coal sample tanks during freezing, the gas pressure evolution characteristics of the coal sample tanks were investigated by testing at the freezing temperature of −20 °C under the adsorption equilibrium pressure of 1.0 MPa for coal samples with different hardness. The results are shown in Figure 4.
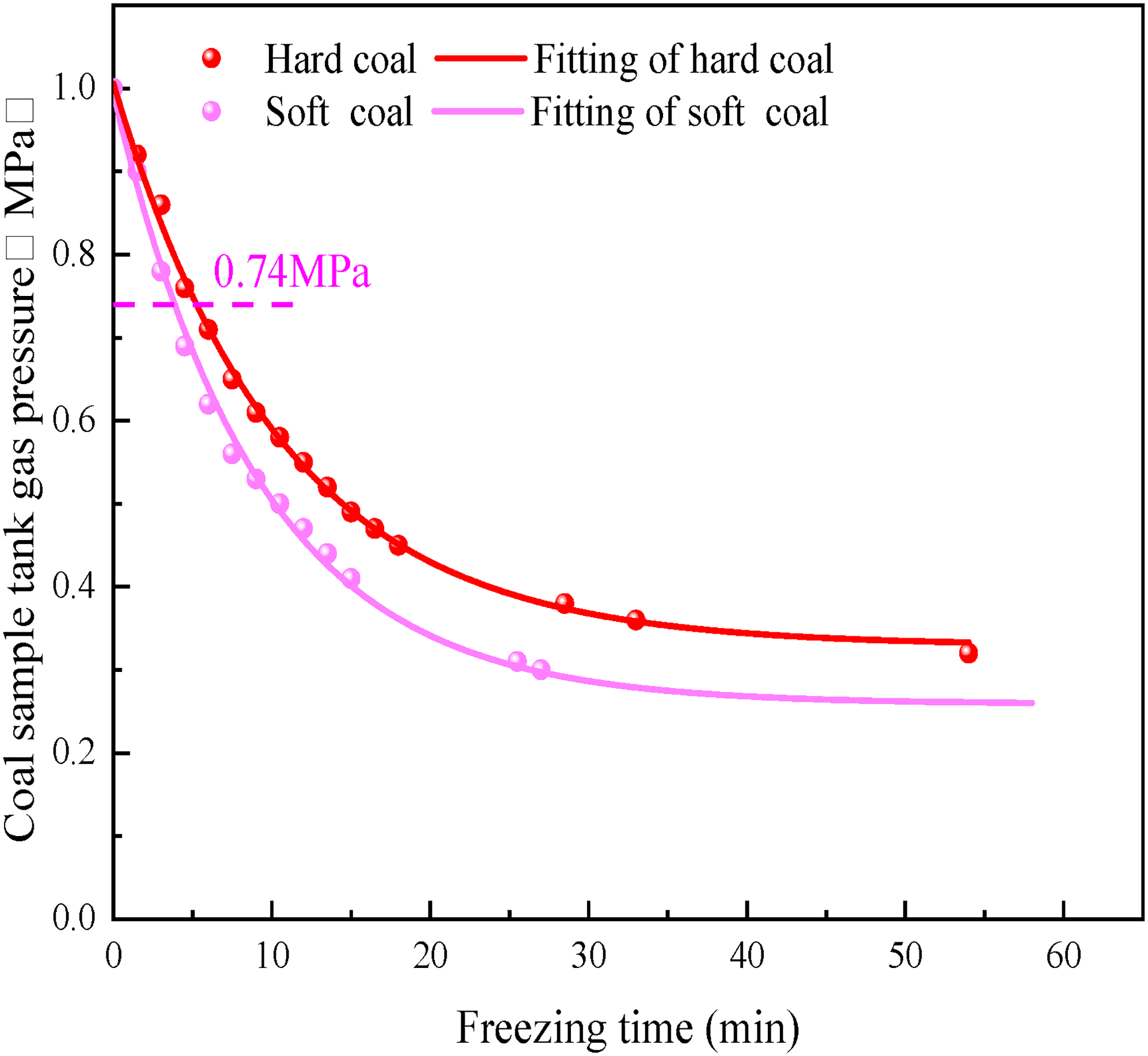
Change of Gas pressure in coal sample tanks of different hardness coal during freezing process.
As can be seen, the freezing temperature was −20 °C, the adsorption equilibrium pressure was 1 MPa, and the gas pressure of the coal sample tank underwent three stages: rapid decline, steady decline, and slow decline during the freezing process of coal-containing gas with different hardness. At 0–8.5 min, the gas pressure of the hard coal sample tank rapidly dropped from 1.0 MPa to 0.63 MPa with an average rate of decrease of 0.043 MPa/min, while that of the soft coal sample tank quickly dropped to 0.54 MPa with an average rate of decrease of 0.054 MPa/min. At 8.5–15 min, the gas pressure of the hard coal sample tank dropped to 0.49 MPa, and the average rate of decrease was 0.021 MPa/min; the gas pressure of the soft coal sample tank dropped to 0.41 MPa, and the average rate of decrease was 0.02 MPa/min. At 15–54 min (the end of freezing), the gas pressure of the hard coal sample tank slowly dropped to 0.32 MPa, and the average decrease rate was 0.0040 MPa/min. At 15–58 min (the end of freezing), the gas pressure of the soft coal tank slowly dropped to 0.23 MPa, and the average rate of decrease was 0.0042 MPa/min. By comparison, it was found that the gas pressure of soft coal was lower than that of hard coal at the same freezing time. The reason for this is the fact that, coal matrix gas adsorption is physical adsorption, it was mainly concentrated on the surface of the micropores and transition pores in the coal body, while the surface of the micropores and transition pores in the coal body of soft coal was larger than that of hard coal, the soft coal adsorption capacity was large, and the low temperature was conducive to gas adsorption; therefore, the gas pressure of the soft coal sample tank was lower.
For the gas-containing coal sample at the same freezing temperature (-20 °C) and adsorption equilibrium pressure (1.0 MPa), the gas pressure of different coal types (soft and hard coal) in the coal sample tank changed as a monotonously decreasing function. The gas pressure of the coal sample tank is proportional to the freezing time as a power function relationship, and its expression is given by Equation (1); all degrees of fit are above 0.99. The relevant fitting parameters are listed in Table 4.
Gas pressure-freezing time fitting parameters of different hardness coal sample tanks.

As presented in Table 4, during the freezing process, the gas pressure attenuation coefficient of the soft coal sample tank was 0.109, while that of the hard coal was 0.095. The gas pressure attenuation coefficient of the soft coal sample tank was larger; that is, the gas pressure decreased faster. Because the coal matrix gas adsorption is physical adsorption, it is mainly concentrated on the surface of the micropores and transition pores in the coal body. Because the surface of the micropores and transition pores of soft coal is larger than that of hard coal, the adsorption capacity of soft coal is higher than that of hard coal. Therefore, under the same freezing temperature, soft coal absorbed gas faster, and the gas pressure of the coal sample tank decreased faster. When the freezing ended, the gas pressure value of the tank was less compared with that of hard coal.
Conclusion
Through the independent design of an experimental device for determining the freezing response characteristics of gas-containing coal, the gas pressure evolution law was measured for coal samples with different hardness in composite coal seams under different freezing temperatures and adsorption equilibrium pressures, and the following conclusions were drawn.
During the freezing process of the gas-containing coal sample, the gas pressure in the coal sample tank changed monotonously in three stages: rapid decline, steady decline, and slow decline. The gas pressure of the coal sample tank has a power-function relationship with the freezing time: When the adsorption equilibrium pressure and hardness of the gas-containing coal was the same at different freezing temperatures (-10 °C, −15 °C, −20 °C, −25 °C, −30 °C), the adsorption capacity of the coal sample increased as the temperature decreased. As the temperature decreased, the greater the difference between the initial temperature of the coal sample and the freezing temperature, the stronger the freezing effect during the cooling process, and the faster the gas pressure reduction rate,the gas pressure attenuation coefficient increased (0.089, 0.092, 0.095, 0.109, 0.124), the gas pressure of the coal sample tank decreased faster, and the effect was more obvious in the slow decrease stage. When the adsorption equilibrium gas pressure was 1 MPa and the freezing ended, the gas pressure values decreased to 0.43 MPa, 0.38 MPa, 0.32 MPa, 0.27 MPa, and 0.21 MPa, respectively. When the gas-containing coal had the same freezing temperature and hardness, and the gas adsorption equilibrium pressure was 1.0 MPa, 1.5 MPa, and 2.0 MPa, the adsorption equilibrium pressure was larger, the amount of gas molecules increased, as the gas pressure attenuation coefficient decreased (0.109, 0.075, 0.072;). the gas pressure decreased faster, the gas pressure of the coal sample tank decreased to 0.74 MPa, and the duration increased to 4 min, 19 min, and 49 min, respectively. During the freezing process with the same freezing temperature and adsorption equilibrium pressure and different gas-containing coal hardness, the surface of the micropores and transition pores in the soft coal was larger compared with hard coal, and the adsorption capacity of the soft coal increased. The gas pressure of the soft coal sample tank decreased. The gas pressure attenuation coefficient of the soft coal sample tank was 0.109, and that of the hard coal sample tank was 0.095. The gas pressure attenuation coefficient of the soft coal sample tank was large, and the rapid decrease in the gas pressure was mainly affected by freezing. The gas pressure of the soft and hard coal sample tanks was relatively small. When the adsorption equilibrium gas pressure was 1 MPa and the freezing ended, the gas pressure of soft coal was 0.23 MPa and that of hard coal was 0.32 MPa. After the gas-containing composite coal seam underwent freezing, the gas pressure dropped below 0.74 MPa, which reduced the risk of coal and gas outburst The findings of this study have important theoretical guiding significance for the prevention and control of coal and gas outbursts caused by freezing in composite coal seams.
