Abstract
Introduction
Carbon dioxide (CO2) is the primary greenhouse gas responsible for global climate change. Additionally, CO2 contributes to atmospheric pollution and acid rain. Reducing CO2 emissions can alleviate these environmental issues, enhance air quality, and improve ecological systems (Lei et al., 2023). Moreover, CO2 mitigation supports the development of a low-carbon economy, promotes the use of sustainable energy, and fosters clean production, thereby conserving energy resources and promoting their sustainable use (Godin et al., 2021). Current methods for capturing CO2 include absorption, adsorption, membrane separation (Vorotyntsev et al., 2012), and biological fixation (Saini et al., 2009, Gong et al., 2018). Membrane separation is typically applied to gases with low CO2 concentrations and offers a simple process that can be operated continuously. However, challenges such as membrane durability and contamination must be addressed. Biological fixation, suitable for low CO2 concentrations, has low operational costs but requires high initial construction and operational expenses. Adsorption, by contrast, is a mainstream method for treating low-concentration CO2, offering lower investment and operational costs (Al Wahedi et al., 2015). In addition, adsorbents used in adsorption processes can be regenerated, extending their service life. Among these, the absorption method is effective for gases with high CO2 concentrations, but the limited lifespan of absorbents, along with the need for frequent replacement or regeneration, results in relatively high operational costs. The key to adsorption efficiency lies in the selection of adsorption equipment and adsorbents (Chouikhi et al., 2020), as high-performance equipment and adsorbents improve efficiency, stability, and cost-effectiveness.
Common CO2 adsorption systems include fixed beds (Chouikhi et al., 2020; Jurkiewicz et al., 2023) and fluidized bed (Chen et al., 2015, Lasek et al., 2023). Fixed beds are widely used due to their simple design and ease of operation, but they face drawbacks such as high mass transfer resistance and long regeneration times for adsorbents. Fluidized beds, on the other hand, offer high mass transfer efficiency and shorter regeneration times, but they require complex equipment and precise control of gas velocity, leading to higher energy consumption. To avoid issues such as back-mixing, which can lower adsorption efficiency, careful control of gas flow rates and pressures is necessary in fluidized beds (Bérard et al., 2021). The high-gravity rotating adsorption bed offers a potential solution by rotating the adsorbent to prevent particle mixing, thereby enhancing mass transfer and significantly increasing both adsorption rates and capacities. Gao found that solid polyamine adsorbents in a rotating bed exhibited superior adsorption performance compared to a fixed bed (Gao et al., 2023). Compared with fluidized beds, high-gravity rotating adsorption beds have distinct advantages, especially in reducing back-mixing. The centrifugal force in high-gravity beds keeps the adsorbent particles fixed and creates a plug-flow-like environment, effectively minimizing axial and radial mixing that commonly occurs in fluidized beds. This leads to higher adsorption efficiency, more uniform breakthrough behavior, and improved mass transfer, as well as stable operation without the need for precise gas velocity control. Therefore, high-gravity beds are better suited for efficient and scalable CO2 adsorption applications. The rotating bed also improves CO2 recovery by creating more uniform airflow and temperature distribution, reducing clogging and wear, and maintaining long-term adsorbent performance. Rotation enhances diffusion between gas and solid phases, facilitating deep pore adsorption.
Adsorbent selection is another critical factor influencing adsorption performance. Common CO2 adsorbents include activated carbon (Jang and Park 2011, 2012), molecular sieve (MS) (Vaduva and Stanciu 2007; Heck et al., 2018; Wu 2018), and silica gel (Zhu et al., 2010). An ideal adsorbent should have a well-developed surface area and pore structure. MSs, known for their thermal stability and regeneration capabilities, are widely used due to their selective adsorption based on molecular size and polarity (Matsuda et al., 2010) (Dai et al., 2020). However, the physical adsorption capacity of MSs is limited. Modifying MSs can enhance their adsorption properties, with amines (Hu et al., 2017), silicon amine (Popa et al., 2018) and polymer (Wang et al., 2016) being commonly used modifiers. Amines, in particular, contain amine groups (-NH2, -NH) that react with CO2 to form carbamate or bicarbonate species through chemical adsorption mechanisms (Elwakeel and Guibal 2015; Elwakeel et al., 2016; Elwakeel and Guibal 2016; Alghamdi et al., 2024). These functional groups significantly enhance the selectivity and adsorption capacity of MSs for CO2, making amine-modified adsorbents a focal point of research. The traditional method of modifying MSs through impregnation relies on molecular diffusion forces to achieve loading, but inappropriate concentrations of amine solutions can block pores, reducing adsorption performance. Therefore, developing new modification processes that address these limitations is crucial. As an intensified process, high gravity has been proven to improve production efficiency and product quality in various applications (Zhao et al., 2016; Guo et al., 2018), including absorption (Lin and Chu 2015a, 2015b), distillation (Chu et al., 2013; Garcia et al., 2017), and extraction(Modak et al., 2016, Chang et al., 2019). Recent studies have shown that high-gravity liquid–solid adsorption technology can increase liquid component loading and ensure better dispersion (Guo et al., 2019a, 2019b). Applying this technology to modify MSs can improve amine utilization while preserving the porous structure of the sieves, combining physical and chemical adsorption to enhance CO2 adsorption capacity.
In this study, high-efficiency amine-modified MSs were prepared using high-gravity modification technology, and CO2 was adsorbed using a high-gravity rotating adsorption bed. The study delves into the modification performance and adsorption mechanisms, optimizing the adsorption process. The findings provide new insights into CO2 treatment technology and demonstrate promising potential for innovative applications.
Experimental
Materials
Diethylenetriamine (DETA, ≥ 99%) was purchased from Tianjin Damao Chemical Reagent Factory, China. 13X MS (particle size 2 mm, analytical reagent grade) was supplied by Henan Zhisheng Environmental Protection Technology Co., Ltd CO2 (99.99%) and N2 (99.99%) gases were obtained from Shanxi Taineng Gas Co., Ltd
Experiment process
The high-gravity rotating adsorption bed is used for amine-modified 13X MS. The high-gravity rotating adsorption bed is used for modified 13X MS to adsorb CO2. Figure 1 is a schematic diagram of the equipment structure.

Schematic of the high-gravity rotating adsorption bed and its process flow using modified 13X molecular sieve.
The 13X MS was washed with deionized water and dried in an oven at 80°C. Figure 1 is a process flow chart of high-gravity modified 13X MS (Labeled as 13X MS-RPB). In order to compare the modification effect, the stirred tank was used to modify 13X MS under the same process conditions (labeled 13X MS-ST). 10 g of pretreated 13X MS was loaded into the packing area (diameter 45 mm, height 20 mm) of RPB. The rotor speed was set to 0–1200 rpm; DETA aqueous solution (1.0 mol/L) was pumped from a storage tank by a peristaltic pump at a flow rate of 40∼160 mL/min. The DETA solution was recirculated to maximize contact. For comparison, conventional impregnation was performed in a 500 mL glass reactor under identical conditions (DETA: 1.0 mol/L).
Figure 2 is a flow chart of the high-gravity adsorption process. The modified 13X MS (mass 30 g) was packed into the RPB (height 350 mm, diameter 10 mm). A CO2/N2 gas mixture (CO2 1% vol.) was introduced at a total flow rate of 1.5 m³/h (measured with a mass flow controller), at ambient temperature and atmospheric pressure. Inlet and outlet CO2 concentrations were continuously monitored by an infrared gas analyzer (model 3000GHG). Each adsorption test was repeated at least three times.

The process flow chart of modified 13X MS adsorbing CO2 in high-gravity environment. MS: molecular sieve; CO2: carbon dioxide.
Equation 1 is used to calculate the CO2 adsorption capacity of 13X MS.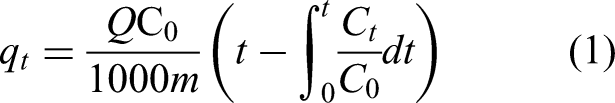
The high-gravity factor describes the high-gravity field strength during the adsorption and modification process and is calculated using Equation 2.
Sample characterization
The specific surface area and pore size structure of the sample were analyzed by Brunauer–Emmett–Teller (BET) (Autosorb-iQ2-MP, a micropore physical adsorption instrument of Quanta Corporation in the United States). The crystal structure of the sample was determined using X-ray diffraction (XRD) (SmartLab Intelligent X-ray Diffractometer from Rigaku Corporation, Japan). XRD uses a copper target for measurement and wide-angle diffraction of rice seedlings with an angle of 5–85°. Thermal stability and decomposition properties of the samples were determined by thermogravimetric analysis (TGA) (NETZSCH analyzer, Germany). The microscopic morphology and features of the sample surfaces were determined by scanning electron microscope (SEM) (Thermo Scientific Apreo 2S). The functional group structure and chemical bond characteristics of the samples were detected by infrared spectrometer (PE, USA) and the modification conditions of the samples were analyzed.
Results and discussion
Selection of diethylenetriamine concentration
The aim of this section is to determine the optimal concentration of DETA for modifying 13X MSs (13X MS) in order to balance pore structure preservation and CO2 adsorption efficiency.
Figure 3 shows that as the DETA concentration increases, the specific surface area of the 13X MS gradually decreases, while the pore size increases. This is due to the adsorption of the DETA functional groups on the surface of the zeolite. As the concentration increases, more functional groups enter the pore structure, occupying the surface active sites originally available for physical adsorption, resulting in partial pore blockage or deformation. This affects both the specific surface area and pore size.

Effects of modified DETA concentrations on surface area, pore size, and adsorption capacity. DETA: diethylenetriamine.
In terms of adsorption performance, increasing the DETA concentration enhances the adsorption capacity of the 13X zeolite, particularly at low concentrations. At these concentrations, the improvement in adsorption capacity primarily depends on physical adsorption. When the concentration exceeds 1.0 mol/L, the increase in adsorption capacity becomes more gradual (as shown in Figure 3), indicating that the contribution of physical adsorption diminishes, and the adsorption performance is increasingly dependent on the chemical adsorption of the functional groups. Excessive DETA may lead to pore blockage or the aggregation of functional groups, thereby limiting further improvements in adsorption performance.
Overall, when the DETA concentration is 1.0 mol/L, the 13X MS achieves a balance between physical and chemical adsorption, maintaining a good pore structure while providing sufficient functional groups for efficient CO2 adsorption. Therefore, the recommended optimal modification concentration is 1.0 mol/L.
Therefore, a DETA concentration of 1.0 mol/L is recommended as the optimal condition for modifying 13X MS. This concentration provides the best compromise between maximizing adsorption performance and maintaining structural integrity.
Brunauer–Emmett–Teller characterization
To investigate the effect of DETA modification on the specific surface area, pore volume, and pore size of 13X MS, BET analysis was conducted on both unmodified and modified samples.
Figure 4 shows the nitrogen adsorption–desorption isotherms and pore size distribution plots for different samples. The nitrogen adsorption–desorption isotherms exhibit a typical type III isotherm, indicating multilayer adsorption. The nitrogen adsorption–desorption curve for DETA-modified 13X MS suggests multilayer adsorption. Table 1 lists the specific surface area, pore size, and pore volume for the different samples. According to the data in the table, the specific surface area and pore volume of the modified 13X MS are reduced. After DETA loading, the reduction in specific surface area was moderate for both samples (3.8% for RPB and 11.5% for ST), but the average pore size increased by only 9.9% for RPB and by 75.1% for ST. This substantial difference suggests a distinct mechanism of DETA distribution between the two modification methods. In the ST sample, the static loading process likely leads to non-uniform DETA deposition and local aggregation, as evidenced by the formation of DETA clusters observed in the SEM images (Figure 5(b)). These aggregates may partially block some pores while simultaneously opening or merging others, resulting in a pronounced increase in average pore diameter. In contrast, the RPB sample benefits from enhanced mixing and centrifugal force, which facilitate the uniform dispersion of DETA molecules on the MS surface. This results in a thin and continuous DETA coating, minimizing aggregation and preserving the original pore network (Figure 6(c)). Thus, the difference in pore size change can be explained by the tendency for DETA aggregation and partial pore blockage in the ST sample, versus uniform pore widening in the RPB sample.

Nitrogen adsorption–desorption isotherms and pore size distribution of different samples.

The SEM image of sample ((a) 13X MS; (b) 13X MS-ST-1 mol/L DETA; (c) 13X MS-RPB-1 mol/L DETA; (d) Surface local magnification of 13X MS-RPB-1 mol/L DETA). MS: molecular sieve; DETA: diethylenetriamine; SEM: scanning electron microscope.

Characterization results of different samples((a) EDS diagrams; (b) XRD patterns; (c) Fourier transform infrared spectra (FT-IR); (d) thermogravimetric (TG) diagrams); therefore, the TGA results confirm the improved DETA incorporation and excellent thermal resistance of the modified adsorbents, which is critical for their reliable application in CO2 adsorption processes. DETA: diethylenetriamine; XRD: X-ray diffraction; TGA: thermogravimetric analysis.
Specific surface area, pore volume, and pore size of different modified 13X MS.

MS: molecular sieve; DETA: diethylenetriamine.
DETA has a highly microfine characteristic, enabling it to be uniformly loaded onto the surface and within the pores of 13X MS without local overaccumulation in the RPB. As a result, the channel space is neither occupied nor blocked by DETA. This indicates that RPB-modified 13X MS effectively retains its original structure while its pore size is moderately expanded, providing enhanced pore structure stability and good adsorption performance.
Overall, the RPB-modified 13X MS maintains a better pore structure and higher surface area, which is beneficial for subsequent CO2 adsorption performance. The moderate increase in pore size and good retention of pore volume provide a structural basis for the superior adsorption capability of the RPB-modified material.
Scanning electron microscope characterization
To investigate the morphological changes and DETA distribution on 13X MS after modification, SEM analysis was performed on unmodified and modified samples. This approach allows for the visualization of surface structure changes, pore characteristics, and the dispersion of the modifying agent under different preparation methods.
SEM characterization of different samples (Figure 5) shows that 13X MS (Figure 5(a)) has distinct porous cavities. The modified 13X MS retains its porous structure, but some cavities are covered, indicating that it still possesses physical adsorption capabilities. The surface of 13X MS presents rough crystalline material, which effectively increases the specific surface area of the support. In the modified 13X MS, the crystalline material slightly disappears, with more of it disappearing in 13X MS-ST-1 mol/L DETA compared to 13X MS-RPB-1 mol/L DETA. The 13X MS-RPB-1 mol/L DETA maintains the well-developed pore structure and specific surface area of the original support. The modified 13X MS tends to undergo surface aggregation and accumulation of DETA in the stirring tank, which damages the crystalline structure of the support and reduces its specific surface area. In contrast, RPB-modified 13X MS is dispersed more uniformly on the surface and within the pores of 13X MS, preserving its original developed pore structure.
The EDS results of different samples (Figure 6(a)) indicate that 13X MS is primarily composed of calcium (Ca), aluminum (Al), and silicon (Si) elements. The proportion of calcium decreases and that of silicon increases in the 13X MS after DETA modification. This is because the DETA solution reacts with calcium ions to form organic amine-calcium ion complexes, which replace calcium during the modification process; as 13X MS has a porous structure mainly composed of silicates and aluminates linked by oxygen bridges, the removal of calcium ions effectively increases the DETA loading in the pores, thereby enhancing the modification effect.
Therefore, SEM analysis reveals that high-gravity RPB modification provides a more favorable surface morphology and DETA distribution, which helps preserve the structural advantages of 13X MS and enhances its potential for CO2 adsorption.
X-ray diffraction characterization
The aim of this section is to evaluate the effect of DETA modification on the crystallinity and structural integrity of 13X MS, using XRD analysis. By comparing the XRD patterns before and after modification, we can assess whether the modification process alters the zeolite framework and investigate the dispersion of DETA within the samples.
The XRD of different samples (Figure 6(b)) shows that the peaks of 13x MS and modified 13x MS are sharp and highly crystalline. The characteristic peaks of 13x MS appear at 7.9°, 16.3°, 23.9°, 27.6°, 31.2 and the secondary peaks at 15.9°, 17.4°, 34.4°, 37.2°. The modified 13x MS maintains the structure of the original 13X MS because higher peaks are detected at the same position. However, the modified 13x MS peak shape has attenuated. This is because DETA is loaded on 13x MS and covers the crystal form, causing clogging of the pores and destruction of crystallinity. The sharpness of 13x MS-RPB-1 mol/L DETA is higher than that of 13x MS-ST-1 mol/L DETA. The results show that the modified DETA in stirred tank has poor dispersibility and is easy to accumulate on the carrier. The high-gravity rotating adsorption bed evenly distributes DETA on the carrier and is not easy to accumulate on the crystal. The XRD characterization results show that the high-gravity rotating adsorption bed is a good modification device.
The reduction in peak intensity after DETA modification indicates that some crystallinity is lost due to DETA covering the crystal surface. The effect is more pronounced in the ST-modified sample, suggesting less uniform DETA dispersion and greater structural disruption. In contrast, the RPB-modified sample retains higher crystallinity, reflecting more uniform DETA distribution and better preservation of the crystal structure.
Fourier transform infrared spectra characterization
Fourier transform infrared spectra (FT-IR) spectroscopy was employed to investigate the chemical interactions between DETA and the 13X MS after modification. The goal was to identify changes in functional groups and chemical bonds that might result from DETA loading.
The modified 13X MS did not exhibit any new characteristic peaks of functional groups in the FT-IR spectrum (Figure 6(c)). However, changes in both the intensity and position of the original characteristic absorption peaks indicate an interaction between DETA molecules and the 13X MS surface. The bending vibration peak of the Si-O-Si framework was observed at 556 cm−1, while the peak at 670 cm−1 corresponds to the characteristic vibration of the Si-O or Si-O-Al framework. The stretching vibration peak of Si-O-Si at 997 cm−1 confirms the presence of the MS framework. The peak at 1638 cm−1 is attributed to the bending vibration of H-O-H, whereas the peak at 3437 cm−1 corresponds to the stretching vibrations of O-H and N-H. Notably, the increased peak intensity at this position for the modified 13X MS compared to the unmodified sample suggests an enhancement of N-H vibrations, further confirming the success of DETA modification. Additionally, the increased intensity of the Si-O-Si stretching vibration peak at 997 cm−1 after modification suggests that the introduction of DETA enhances the interaction within the 13X MS framework. Although no new functional group characteristic peaks were observed in the FT-IR spectrum, the shifts and intensity changes of the characteristic peaks indicate the presence of physical interactions or weak chemical interactions between DETA and the 13X MS surface.
The FT-IR spectra for unmodified and DETA-modified 13X MS show no new functional group peaks, but notable changes in the intensity of characteristic absorptions confirm the occurrence of interactions between DETA and the 13X MS surface. Specifically, the O–H/N–H stretching vibration at 3437 cm−1 exhibited a marked increase in peak area after DETA modification. Quantitatively, the peak area at 3437 cm−1 increased by 42.3% for the ST-modified sample and by 78.6% for the RPB-modified sample compared to unmodified 13X MS. The ratio of the peak area at 3437 cm−1 to that at 997 cm−1 (Si–O–Si stretching) also increased by 0.24 for ST modification and 0.47 for RPB modification, respectively, relative to the original sample. These quantitative results demonstrate that RPB modification introduces and retains more N–H functional groups on the 13X MS surface than ST modification, evidencing higher DETA loading and better dispersion. Therefore, even though no new functional group peaks appeared, the substantial increase in the characteristic N–H vibration region provides direct evidence of successful DETA modification, especially using high-gravity technology.
Overall, the FT-IR analysis confirms the successful modification of 13X MS by DETA, primarily through physical adsorption or weak chemical interactions, which is essential for enhancing CO2 adsorption capacity.
Tg characterization
The aim of this section is to evaluate the thermal stability and DETA loading of the modified 13X MS samples by TGA.
Figure 6(d) shows that the thermal weight loss mainly occurs before 200°C, which is primarily attributed to the evaporation of adsorbed moisture and gases. Compared to unmodified 13X MS, the modified samples (13X MS-ST-1 mol/L DETA and 13X MS-RPB-1 mol/L DETA) exhibit greater weight loss. This phenomenon can be attributed to the increased mesoporous structures and higher DETA loading after modification, leading to enhanced volatilization of DETA. Specifically, after 350°C, the weight loss of 13X MS-RPB-1 mol/L DETA is higher than that of 13X MS-ST-1 mol/L DETA, indicating that the RPB-modified adsorbent has a higher DETA loading. Furthermore, the TG diagram indicates that within the temperature range up to 150°C, the mass retention for all three adsorbents is above 90%, showing good thermal stability.
Modified 13X molecular sieve for carbon dioxide adsorption
To compare the CO2 adsorption performance of 13X MS modified by different methods and to evaluate the effect of high-gravity technology, breakthrough adsorption experiments were conducted using both fixed beds and high-gravity rotating adsorption beds under identical operating conditions.
The CO2 adsorption performance of modified 13X MS prepared by two different methods was compared under identical conditions to illustrate the adsorption performance enhancement. Additionally, the adsorption behavior of the modified 13X MS in fixed bed and high-gravity rotating adsorption bed was compared.
Figure 7(a) presents the breakthrough curves of CO2 adsorption for different 13X MS samples in a fixed bed, and Table 2 summarizes their corresponding saturated adsorption capacities. Results show that the adsorption performance of DETA-modified 13X MS significantly improved compared to unmodified 13X MS. The breakthrough times of 13X MS-ST-1 mol/L DETA and 13X MS-RPB-1 mol/L DETA were approximately increased by 1.6 times and 2.3 times, respectively, compared with 13X MS, indicating enhanced adsorption performance. The breakthrough curves shifted to the right for the modified samples, and the adsorption equilibrium times of all samples were similar. Additionally, the slopes of breakthrough curves gradually decreased over time as adsorption approached equilibrium. According to Table 2, the saturated adsorption capacities of 13X MS-ST-1 mol/L DETA and 13X MS-RPB-1 mol/L DETA increased by 17.78% and 23.81%, respectively.

The adsorption breakthrough curves of CO2 under different conditions ((a) different 13X molecular sieves (fixed bed); (b) different adsorption equipment).
The adsorption capacity of different 13X MS.

Working conditions: the gas volumetric flow is 1.5 m3/h, and the initial CO2 concentration is 198214 mg/m3.
MS: molecular sieve; DETA: diethylenetriamine; CO2: carbon dioxide.
Modification with DETA introduced additional active sites on the surface of 13X MS, enhancing its ability to selectively adsorb CO2 and weakening competitive adsorption of other components. The improved performance of 13X MS-RPB-1 mol/L DETA compared to 13X MS-ST-1 mol/L DETA is attributed to the intensified mass transfer and better DETA dispersion achieved in the high-gravity rotating adsorption bed. Under high-speed rotation conditions, DETA was atomized effectively, enhancing its loading and distribution on the 13X MS surface. The high-gravity environment also improved pore surface renewal rates, creating a concentration gradient driving force within the pores, thereby increasing DETA coverage and enhancing intermolecular chemical interactions between CO2 and the adsorbent surface, resulting in superior adsorption performance.
In order to further verify the adsorption performance of the high-gravity rotating adsorption bed, the adsorption performance of 13X MS-RPB-1 mol/L DETA for CO2 in the fixed bed and the high-gravity rotating adsorption bed was measured under the same process conditions. Figure 7(b) shows the adsorption breakthrough curves of different adsorption equipment. Table 3 shows the adsorption capacity of different adsorption devices for CO2. The adsorption capacity of the high-gravity rotating adsorption bed is increased by 16.48% compared with that of the fixed bed.
The adsorption capacity of different adsorption device.

Working conditions: the gas volumetric flow is 1.5 m3/h, and the initial CO2 concentration is 198214 mg/m3.
MS: molecular sieve; DETA: diethylenetriamine; CO2: carbon dioxide.
Therefore, DETA modification and high-gravity technology work synergistically to enhance the CO2 adsorption performance of 13X MS. The use of a high-gravity rotating adsorption bed not only increases adsorption capacity but also prolongs the effective adsorption period, offering significant advantages for practical CO2 capture applications.
Adsorption performance of unmodified 13X molecular sieve in high-gravity rotating adsorption bed
To further clarify whether the performance enhancement arises from high-gravity rotating adsorption bed or the DETA modification, control experiments using unmodified 13X MS in the high-gravity rotating adsorption bed were conducted under the same conditions. The corresponding adsorption capacity is shown in Table 4.
Co2 adsorption capacities of different adsorbents and beds.

Working conditions: gas volumetric flow 1.5 m³/h, initial CO2 concentration 198214 mg/m³.
MS: molecular sieve; DETA: diethylenetriamine.
The data in Table 4 clearly show that the high-gravity rotating adsorption bed itself can improve the adsorption performance of unmodified 13X MS by 17.36% compared to the fixed bed, due to intensified mass transfer and improved gas–solid contact. Furthermore, DETA modification (13X MS-RPB-1 mol/L DETA) enhances the adsorption capacity both in fixed and high-gravity beds. The combination of DETA modification and high-gravity adsorption results in the highest capacity (21.54 mg/g), representing a 48.93% increase over unmodified 13X MS in a fixed bed.
Therefore, both high-gravity operation and adsorbent modification contribute synergistically to the observed performance improvement. The effect of high gravity on unmodified 13X MS demonstrates that the bed design itself plays a significant role, but the additional benefit from DETA modification is even greater under high-gravity conditions.
To further elucidate the adsorption mechanism, the experimental breakthrough curves were analyzed using the Yoon-Nelson model. The Yoon-Nelson model is described by the following equation: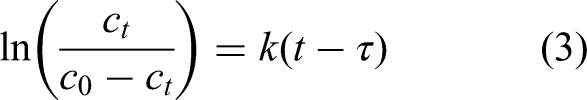
where C0 and Ct are the inlet and outlet concentrations at time t, respectively, k is the rateconstant (min−1), and r is the time required for 50% breakthrough (min).
Nonlinear regression was used to fit the Yoon-Nelson model to the breakthrough data for four adsorbent systems: 13X MS, 13X MS-ST-1 mol/L DETA, 13X MS-RPB-1 mol/L DETA, and 13X MS-RPB-1 mol/L DETA. The fits were performed separately for the high-gravity rotating adsorption bed and the conventional fixed bed. The resulting model parameters are summarized in Table 5.
Yoon-Nelson model fitting parameters for different adsorbents and beds.

MS: molecular sieve; DETA: diethylenetriamine.
The higher rate constant (k) for 13X MS-RPB-1 mol/L DETA in the high-gravity rotating adsorption bed demonstrate significantly enhanced adsorption kinetics and adsorbent utilization, confirming intensified mass transfer and improved process efficiency. Moreover, the Yoon-Nelson model fit the experimental data very well (R² > 0.99), providing reliable mechanistic insight and a quantitative basis for comparing adsorbent performance and process configurations.
Mechanism of adsorption process
To further explore the adsorption mechanism of the high-gravity rotating packed adsorption bed, the adsorption diffusion process was studied in this work. The Weber-Morris intraparticle diffusion model was employed to describe the adsorption process, allowing the identification of the rate-controlling steps at different adsorption stages. The mathematical expression of this model is as follows: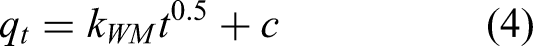
where kWM is intraparticle diffusion rate constant, mg/(g·min0.5);
The Weber-Morris particle diffusion model was applied to describe the adsorption behaviors in different adsorption devices. The adsorption process includes three stages: initial rapid surface adsorption, subsequent slower intraparticle diffusion, and finally the adsorption equilibrium. As shown in Figure 8, the fitting lines did not pass through the origin, indicating that adsorption was jointly controlled by intraparticle diffusion and film diffusion rather than a single mechanism. The intraparticle diffusion rate constants (kWM) of the high-gravity rotating adsorption bed and the fixed bed were 989.05 mg·g−1·min−0.5 and 897.32 mg·g−1·min−0.5, respectively, clearly indicating enhanced mass transfer and adsorption performance in the high-gravity rotating adsorption bed. Furthermore, Table 6 demonstrates that the surface coverage area and coverage ratio of CO2 in the high-gravity rotating adsorption bed are significantly higher than in the fixed bed, attributed to intensified gas–solid contact and improved mass transfer, thus providing more accessible adsorption sites within the inner pores of 13X MS.

The diffusion model of adsorbent particles in high-gravity rotating adsorption bed and fixed bed.
The coverage area and surface coverage of CO2 in high-gravity rotating adsorption bed and fixed bed on 13X MS

MS: molecular sieve; CO2: carbon dioxide.
The results indicate that the high-gravity rotating adsorption bed significantly outperforms the fixed bed in terms of adsorption performance. Specifically, the adsorption penetration time of the high-gravity rotating adsorption bed is 1.35 times that of the fixed bed. The entire penetration curve shifts to the right, clearly demonstrating a delayed breakthrough and prolonged effective adsorption phase. Additionally, after the adsorption saturation point, the slope of the penetration curve slightly increases, indicating faster penetration post-breakthrough. This improvement primarily stems from the enhanced mass transfer properties achieved through mechanical rotation. In contrast to the static fixed bed, adsorbent particles in the rotating bed continuously rotate and tumble, promoting rapid surface renewal and intensifying gas–solid interaction. This dynamic movement effectively improves gas renewal rates at the adsorbent surface and facilitates deep pore adsorption through concentration gradients between the surface and inner pores. Thus, adsorption dead zones are minimized, significantly improving adsorbent utilization. This conclusion is also quantitatively supported by the fact that the coverage area is increased by approximately 26% (461.05 m²/g compared to 365.34 m²/g), and the surface coverage ratio is enhanced by 3.46 percentage points (from 13.15% to 16.61%), clearly demonstrating the superior adsorption capability of the high-gravity rotating adsorption bed. The three-stage adsorption process involves external film diffusion, intraparticle diffusion, and the final equilibrium stage. In the high-gravity rotating adsorption bed, the intense centrifugal force greatly reduces the thickness of the liquid film on the adsorbent surface, significantly enhancing the rate of external film diffusion. This rapid renewal of the boundary layer facilitates faster mass transfer of adsorbate molecules from the bulk phase to the adsorbent surface. Subsequently, the high-gravity field also intensifies intraparticle diffusion by promoting turbulence and micro-mixing within the pores, reducing diffusion resistance and allowing adsorbate molecules to penetrate deeper into the adsorbent structure. Finally, during the equilibrium stage, the more uniform concentration profiles achieved under high-gravity conditions enable the adsorbent to approach equilibrium more rapidly and effectively compared to fixed-bed systems.
Although high-gravity adsorption requires more energy to drive rotation, it greatly improves mass transfer and adsorption rates. As a result, it can achieve higher throughput and better adsorbent utilization, even though their energy consumption (0.12 kWh/m³ gas) is somewhat higher than that of fixed beds (<0.1 kWh/m³ gas). With proper optimization, the benefits in performance can outweigh the extra energy cost, making HGRPBs attractive for industrial use.
Therefore, the mechanistic analysis confirms that the high-gravity rotating adsorption bed not only increases the rate of intraparticle diffusion but also improves the overall adsorption efficiency of 13X MS for CO2, owing to intensified gas–solid contact and better utilization of internal pore structure.
Influence of key parameters on carbon dioxide removal efficiency in high-gravity rotating adsorption bed
To further investigate the CO2 removal performance of DETA-modified 13X MS in a high-gravity rotating adsorption bed, we analyzed the effect of key operational parameters. These include temperature, gas flow rate, gravity factor, and adsorption time, each evaluated in terms of their influence on CO2 removal efficiency.
As shown in Figure 9(a), the CO2 removal efficiency increases with temperature up to an optimal point (approximately 60 °C), after which it declines. This trend reflects the dual nature of the adsorption process. At moderate temperatures, enhanced molecular mobility promotes both physical adsorption and chemical interaction between CO2 and amine groups. However, at higher temperatures, physisorption weakens and partial desorption of CO2 may occur, resulting in reduced removal efficiency.

Influence of key parameters on CO2 removal efficiency in the high-gravity rotating adsorption bed: (a) effect of temperature, (b) effect of gas flow rate, (c) effect of gravity factor, (d) effect of adsorption time.
Figure 9(b) illustrates the effect of gas flow rate. As the flow rate increases from 1.0 m³/h to 2.0 m³/h, the removal efficiency steadily decreases. This is primarily due to shorter contact time between the gas and the adsorbent, which limits mass transfer and leads to incomplete CO2 capture. Higher flow rates also increase the likelihood of breakthrough before equilibrium is reached.
In contrast, the effect of gravity factor, shown in Figure 9(c), exhibits a clear positive correlation. As the gravity factor increases, the centrifugal field intensifies. This promotes rapid gas–solid mixing and enhances CO2 penetration into the porous structure of 13X MS. The resulting increase in mass transfer leads to continuous improvement in removal efficiency, which exceeds 90% at a gravity factor of 400.
Figure 9(d) presents the relationship between adsorption time and removal efficiency. Initially, the efficiency increases rapidly as abundant active sites are available for CO2 binding. Over time, the adsorption rate slows and eventually plateaus, indicating that the system approaches equilibrium and the adsorbent becomes saturated. Optimal efficiency is observed around 30 min.
Consequently, Figure 9(a–(d)) demonstrates that optimal temperature, lower gas flow rates, higher gravity factors, and sufficient adsorption time are all critical for maximizing CO2 removal efficiency in high-gravity rotating adsorption beds. These factors synergistically enhance gas–solid contact, improve amine utilization, and promote deep pore diffusion, resulting in more effective CO2 capture.
The results demonstrate that each operational parameter plays a distinct and significant role in CO2 removal efficiency. Moderate temperatures favor both physisorption and chemisorption; slower gas flow rates provide sufficient residence time for mass transfer; higher gravity factors amplify gas–solid contact and facilitate deep pore diffusion; and appropriate adsorption time ensures that the adsorbent capacity is fully utilized. These factors synergistically enhance CO2 capture in high-gravity rotating beds.
Conclusions
In this study, high-gravity technology was successfully applied to modify 13X MS with DETA, resulting in more uniform dispersion and higher loading of active components, as verified by multiple characterizations. The CO2 adsorption capacity of the high-gravity modified 13X MS was increased by 31.24% compared to the unmodified material and by 7.9% over conventional impregnation methods. Furthermore, application of the high-gravity rotating adsorption bed led to an additional 16.48% improvement in CO2 adsorption performance compared to a fixed bed. Mechanism analysis showed that the high-gravity rotating bed not only strengthened external mass transfer but also enhanced internal pore utilization, significantly improving adsorption efficiency and material utilization. Key operational parameters were systematically investigated, revealing that optimal conditions could achieve CO2 removal efficiencies above 90%. Overall, our findings demonstrate that high-gravity technology offers an innovative and highly efficient approach for CO2 capture, with strong potential for industrial application.
